Abstract
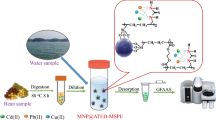
We describe a simple and efficient method for solid phase extraction and speciation of trace quantities of arsenic. It is based on the use of functionalized aluminum oxide nanoparticles and does not require any oxidation or reduction steps. The experimental parameters affecting extraction and quantitation were optimized using fractional factorial design methods. Adsorbed arsenic was eluted from the sorbent with 1 M hydrochloric acid and determined by graphite furnace atomic absorption spectrometry. Preconcentration factors up to 750 were achieved depending on the sample volume. Studies on potential interferences by various anions and cations showed the method to be highly selective. Under optimum conditions, the calibration plots are linear in the 5.0 to 280 ng L−1 and 8.0 to 260 ng L−1 concentration ranges for As(III) and total arsenic, respectively. The detection limits (calculated for S/N ratios of 3) are 1.81 and 1.97 ng L−1 for As(III) and total arsenic, respectively. The method was successfully applied to the determination and speciation of arsenic in (spiked) environmental, food and biological samples and gave good recoveries. The method was validated using a certified geological reference material.

Novel functionalized Al2O3 nanoparticles were synthesized and used for speciation and determination of arsenic in different samples. The experimental variables were optimized using fractional factorial design that can save time and operational costs.


Similar content being viewed by others
References
Pizarro I, Gómez M, Cámara C, Palacios MA (2003) Arsenic speciation in environmental and biological samples. Extraction and stability studies. Anal Chim Acta 495:85–98. doi:10.1016/j.aca.2003.08.009
Rabieh S, Bagheri M, Planer-Friedrich B (2013) Speciation of arsenite and arsenate by electrothermal AAS following ionic liquid dispersive liquid-liquid microextraction. Microchim Acta 180:415–421. doi:10.1007/s00604-013-0946-2
Khayatian G, Hassanpoor S (2013) Development of ultrasound-assisted emulsification solidified floating organic drop microextraction for determination of trace amounts of iron and copper in water, food and rock samples. J Iran Chem Soc 10:113–121. doi:10.1007/s13738-012-0131-2
Uluozlu OD, Tuzen M, Mendil D, Soylak M (2010) Determination of As(III) and As(V) species in some natural water and food samples by solid-phase extraction on streptococcus pyogenesimmobilized on Sepabeads SP 70 and hydride generation atomic absorption spectrometry. Food Chem Toxicol 48:1393–1398. doi:10.1016/j.fct.2010.03.007
Rezvani M, Asgharinezhad AA, Ebrahimzadeh H, Shekari N (2014) A polyaniline-magnetite nanocomposite as an anion exchange sorbent for solid-phase extraction of chromium (VI) ions. Microchim Acta 181:1887–1895. doi:10.1007/s00604-014-1262-1
Leufroy A, Noël L, Dufailly V, Beauchemin D, Guérin T (2011) Determination of seven arsenic species in seafood by ion exchange chromatography coupled to inductively coupled plasma-mass spectrometry following microwave assisted extraction: method validation and occurrence data. Talanta 83:770–779. doi:10.1016/j.talanta.2010.10.050
Pineiro AM, Moreda-Pineiro J, Alonso-Rodrıguez E, nLopez-Mahıa P, Muniategui-Lorenzo S, Prada-Rodrıguez D (2013) Arsenic species determination in human scalp hair by pressurized hot water extraction and high performance liquid chromatography-inductively coupled plasma-mass spectrometry. Talanta 105:422–428. doi:10.1016/j.talanta.2012.10.070
Doker S, Uzun L, Denizli A (2013) Arsenic speciation in water and snow samples by adsorption onto PHEMA in a micro-pipette-tip and GFAAS detection applying large-volume injection. Talanta 103:123–129. doi:10.1016/j.talanta.2012.10.019
Khayatian G, Hassanpoor S, Nasiri F, Zolali A (2012) Preconcentration, determination and speciation of iron using dimethyl (E)-2-[(Z)-1-acetyl)-2-hydroxy-1-propenyl]-2-butendioate. Quim Nov. 35:535–540. doi:10.1590/S0100-40422012000300018
Jain VK, Handa A, Sait SS, Shrivastava P, Agrawal YK (2001) Preconcentration, separation and trace determination of lanthanum (III), cerium (III), thorium (IV) and uranium (VI) on polymer supported o-vanillinsemicarbazone. Anal Chim Acta 429:237–246. doi:10.1016/S0003-2670(00)01299-X
Khayatian G, Hassanpoor S (2012) Simultaneous separation and extraction of Ag (I), Pb (II) and Pd (II) ions by solid phase method and determination of these ions by flame atomic absorption spectrometry. J Incl Phenom Macrocycl Chem 73:151–159. doi:10.1007/s10847-011-0038-z
Starvin AM, Rao TP (2004) Solid phase extractive preconcentration of uranium (VI) onto diarylazobisphenol modified activated carbon. Talanta 63:225–232. doi:10.1016/j.talanta.2003.11.001
Puri BK, Wasey A, Katyal M (1989) Spectrophotometric determination of iron after separation of its 9,10-phenanthrenequinone monoximate on microcrystalline naphthalene. Microchim Acta 99:17–23. doi:10.1007/BF01242785
Tuzen M, Melek E, Soylak M (2008) Solid-phase extraction of copper, iron and zinc ions on Bacillus thuringiensis israelensis loaded on Dowex optipore V-493. J Hazard Mater 159:335–341. doi:10.1016/j.jhazmat.2008.02.021
Pourreza N, Parham H, Kiasat AR, Ghanemi K, Abdollahi N (2009) Solid phase extraction of mercury on sulfur loaded with N-(2-chloro benzoyl)-N-phenylthiourea as a new adsorbent and determination by cold vapor atomic absorption spectrometry. Talanta 78:1293–1297. doi:10.1016/j.talanta.2009.01.056
Özdemir S, Okumus V, Dündar A, Kilinç E (2013) Preconcentration of metal ions using microbacteria. Microchim Acta 180:719–739. doi:10.1007/s00604-013-0991-x
Khayatian G, Hassanpoor S, Azar ARJ, Mohebbi S (2013) Spectrophotometric determination of trace amounts of uranium (VI) using modified magnetic iron oxide nanoparticles in environmental and biological samples. J Braz Chem Soc 24:1808–1817. doi:10.5935/0103-5053.20130226
Erdoğan H, Yalçınkaya Ö, Türker AR (2011) Determination of inorganic arsenic species by hydride generation atomic absorption spectrometry in water samples after preconcentration/separation on nano ZrO2/B2O3 by solid phase extraction. Desalination 280:391–396. doi:10.1016/j.desal.2011.07.029
Lucena R, Simonet BM, Cárdenas S, Valcárcel M (2011) Potential of nanoparticles in sample preparation. J Chromatogr A 1218:620–637. doi:10.1016/j.chroma.2010.10.069
Hua M, Zhang S, Pan B, Zhang W, Lv L, Zhang Q (2012) Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J Hazard Mater 211:317–331. doi:10.1016/j.jhazmat.2011.10.016
Nasiri F, Zolali A, Asadbegi S (2015) Solvent-free one-pot synthesis of 2,2′-Dithioxo-[5,5′] bithiazolidinylidene-4,4′-diones. J Heterocyclic Chem
Bahadur NM, Furusawa T, Sato M, Kurayama F, Siddiquey IA (2011) Fast and facile synthesis of silica coated silver nanoparticles by microwave irradiation. J Colloid Interf Sci 355:312–320. doi:10.1016/j.jcis.2010.12.016
Fan J, Qin Y, Ye C, Peng P, Wu C (2008) Preparation of the diphenylcarbazone-functionalized silica gel and its application to on-line selective solid-phase extraction and determination of mercury by flow-injection spectrophotometry. J Hazard Mater 150:343–350. doi:10.1016/j.jhazmat.2007.04.111
Azizi H, Asahara Y, Mehrabi B, Chung SL (2011) Geochronological and geochemical constraints on the petrogenesis of high-K granite from the Suffi abad area, Sanandaj-Sirjan Zone, NW Iran. Chem Erde 71:363–376. doi:10.1016/j.chemer.2011.06.005
Imai N (1990) Multielement analysis of rocks with the use of geological certified reference material by inductively coupled plasma mass spectrometry. Anal Sci 6:389–395. doi:10.2116/analsci.6.389
Nalwa HS (2004) Encyclopedia for nanoscience and nanotechnology: American Scientific Publishers
Zeng Z, Yu J, Guo ZX (2005) Preparation of functionalized core-shell alumina/polystyrene composite nanoparticles 1: encapsulation of alumina via emulsion polymerization. Macromol Chem Phys 206:1558–1567. doi:10.1002/macp.200500060
Busca G, Lorenzelli V, Ramis G, Willey RJ (1993) Surface sites on spinel-type and corundum-type metal oxide powders. Langmuir 9:1492–1499. doi:10.1021/la00030a012
Khayatian G, Hosseini SS, Hassanpoor S (2013) Ionic liquid-based dispersive liquid–liquid microextraction for determination of trace amounts of iron in water, rock and human blood serum samples. J Iran Chem Soc 10:1167–1173. doi:10.1007/s13738-013-0258-9
Kokya TA, Farhadi K (2009) Optimization of dispersive liquid–liquid microextraction for the selective determination of trace amounts of palladium by flame atomic absorption spectroscopy. J Hazard Mater 169:726–733. doi:10.1016/j.jhazmat.2009.04.005
Ghaedi M, Niknam K, Larki SZHA, Roosta M, Soylak M (2013) Silica chemically bonded N-propyl kriptofix 21 and 22 with immobilized palladium nanoparticles for solid phase extraction and preconcentration of some metal ions. Mat Sci Eng C 33:3180–3189. doi:10.1016/j.msec.2013.03.045
Asadollahzadeha M, Tavakoli H, Torab-Mostaedi M, Hosseini G, Hemmati A (2014) Response surface methodology based on central composite design as a chemometric tool for optimization of dispersive-solidification liquid–liquid microextraction for speciation of inorganic arsenic in environmental water samples. Talanta 123:25–23. doi:10.1016/j.talanta.2013.11.071
Anthemidis AN, Martavaltzoglou EK (2006) Determination of arsenic (III) by flow injection solid phase extraction coupled with on-line hydride generation atomic absorption spectrometry using a PTFE turnings-packed micro-column. Anal Chim Acta 573–574:413–418. doi:10.1016/j.aca.2005.12.055
Rivas RE, López-García I, Hernández-Córdoba M (2009) Speciation of very low amounts of arsenic and antimony in waters using dispersive liquid–liquid microextraction and electrothermal atomic absorption spectrometry. Spectrochim Acta B 64:329–333. doi:10.1016/j.sab.2009.03.007
Jitmanee K, Oshima M, Motomizu S (2005) Speciation of arsenic (III) and arsenic (V) by inductively coupled plasma-atomic emission spectrometry coupled with preconcentration system. Talanta 66:529–533. doi:10.1016/j.talanta.2004.09.024
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 6121 kb)
Rights and permissions
About this article
Cite this article
Hassanpoor, S., Khayatian, G. & Azar, A.R.J. Ultra-trace determination of arsenic species in environmental waters, food and biological samples using a modified aluminum oxide nanoparticle sorbent and AAS detection after multivariate optimization. Microchim Acta 182, 1957–1965 (2015). https://doi.org/10.1007/s00604-015-1532-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-015-1532-6