Abstract
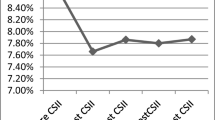
Continuous subcutaneous insulin infusion (CSII) is effective and safe in children and adults with type 1 diabetes. Notwithstanding, some patients decide to discontinue using CSII. We evaluated the discontinuation rate, and its related factors, in a large group of children and adolescents with type 1 diabetes using CSII in Italy. Data on all patients with type 1 diabetes younger than 18 years were collected by 28 Pediatric Diabetologic referral Centers located throughout Italy. The primary endpoint was to measure the discontinuation rate using CSII. Among the study population (n = 6,644), 985 (14.8%) were using CSII. Sixty patients discontinued using CSII, representing the 6.1%. The discontinuation rate significantly increased (P = 0.002) with age: 0–6 years, 1/84 (1.2%), 7–11 years, 8/262 (3.1%), 12–18 years, 51/579 (8.8%). The average time to discontinuation was 1.8 ± 1.4 years. The average age of patients who discontinued using CSII was higher than in patients still on CSII (12.1 ± 3.2 vs. 10.3 ± 3.8, P = 0.0001), while their diabetes duration was significantly shorter (8.6 ± 2.7 vs. 10.2 ± 3.7, P = 0.0001). HbA1c decreased only in patients still on CSII (8.7 ± 1.3% vs. 7.8 ± 1.3%, P = 0.02), but not in patients who discontinued using CSII (8.5 ± 1.6% vs. 8.2 ± 1.3%, P = 0.213). HbA1c might be one important indicator helpful to identify patients at higher risk discontinuing using CSII.


Similar content being viewed by others
References
Weissberg-Benchell J, Antisdel-Lomaglio J, Seshadri R (2003) Insulin pump therapy: a meta-analysis. Diabetes Care 26:1079–1087
Plotnick LP, Clark LM, Brancati FL, Erlinger T (2003) Safety and effectiveness of insulin pump therapy in children and adolescents with type 1 diabetes. Diabetes Care 26:1142–1146
Nahata L (2006) Insulin therapy in pediatric patients with type I diabetes: continuous subcutaneous insulin infusion versus multiple daily injections. Clin Pediatr 45:765–774
Ludvigsson J, Samuelsson U (2007) Continuous insulin infusion (CSII) or modern type of multiple daily injections (MDI) in diabetic children and adolescents: a critical review on a controversial issue. Pediatr Endocrinol Rev 5:666–678
Jeitler K, Horvath K, Berghold A, Gratzer TW, Neeser K, Pieber TR, Siebenhofer A (2008) Continuous subcutaneous insulin infusion versus multiple daily insulin injections in patients with diabetes mellitus: systematic review and meta-analysis. Diabetologia 51:941–951
Pańkowska E, Błazik M, Dziechciarz P, Szypowska A, Szajewska H (2009) Continuous subcutaneous insulin infusion vs multiple daily injections in children with type 1 diabetes: a systematic review and meta-analysis of randomized control trials. Pediatr Diabetes 10:52–58
Bruttomesso D, Costa S, Baritusso A (2009) Continuous subcutaneous insulin infusion (CSII) 30 years later: still the best option for insulin therapy. Diabetes Metab Res Rev 25:99–111
Pickup JC, Sutton AJ (2008) Severe hypoglycaemia and glycaemic control in type 1 diabetes: meta-analysis of multiple daily insulin injections compared with continuous subcutaneous insulin infusion. Diabet Med 25:765–774
Monami M, Lamanna C, Marchionni N, Mannucci E (2010) Continuous subcutaneous insulin infusion versus multiple daily insulin injections in type 1 diabetes: a meta-analysis. Acta Diabetol 47(suppl 1):S77–S81
Colino E, Alvarez MA, Carcavilla A, Alonso M, Ros P, Barrio R (2010) Insulin dose adjustment when changing from multiple daily injections to continuous subcutaneous insulin infusion in the pediatric age group. Acta Diabetol 47(suppl 1):S1–S6
Phillip M, Battelino T, Rodriguez H, Danne T, Kaufman F; European Society for Paediatric Endocrinology; Lawson Wilkins Pediatric Endocrine Society; International Society for Pediatric and Adolescent Diabetes; American Diabetes Association (2007) European Association for the study of diabetes: use of insulin pump therapy in the pediatric age-group: consensus statement from the European Society for Paediatric Endocrinology, the Lawson Wilkins Pediatric Endocrine Society, and the International Society for Pediatric and Adolescent Diabetes, endorsed by the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 30:1653–1662
Wood JR, Moreland EC, Volkening LK, Svoren BM, Butler DA, Laffel LBM (2006) Durability of insulin pump use in pediatric patients with type 1 diabetes. Diabetes Care 29:2355–2360
Ronsin O, Jannot-Lamotte MF, Vague P, Lassman-Vague V (2005) Factors related to CSII compliance. Diabetes Metab 31:90–95
Schifferdecker E, Schmidt K, Boehm BO, Schatz H (1994) Long-term compliance of intensified insulin therapy. Diabetes Res Clin Pract 23:17–23
Floyd JC Jr, Cornell RG, Jacober SJ, Griffith LE, Funnell MM, Wolf LL, Wolf FM (1993) A prospective study identifying risk factors for discontinuance of insulin pump therapy. Diabetes Care 16:1470–1478
Guinn TS, Bailey GJ, Mecklenburg RS (1988) Factors related to discontinuation of continuous subcutaneous insulin-infusion therapy. Diabetes Care 11:46–51
Rabbone I, Iafusco D, Toni S, Lombardo F, Lorini R, Italian Study Group on Diabetes of Italian Society Pediatric of Endocrinology and Diabetology (ISPED) (2010) Italian (retrospective) survey on continuous subcutaneous insulin infusion (CSII) therapy in children and adolescents with type 1 diabetes mellitus. Infusystem International 9:9–13
Hofer SE, Heidtmann B, Raile K, Fröhlich-Reiterer E, Lilienthal E, Berghaeuser MA, Holl RW, DPV-Science-Initiative and the German working group for insulin pump treatment in pediatric patients (2010) Discontinuation of insulin pump treatment in children, adolescents, and young adults. A multicenter analysis based on the DPV database in Germany and Austria. Pediatr Diabetes 11:116–121
Bell DS, Ackerson C, Cutter G, Clements RS Jr (1988) Factors associated with discontinuation of continuous subcutaneous insulin infusion. Am J Med Sci 295:23–28
Knight G, Boulton AJM, Ward JD (1986) Experience of continuous subcutaneous insulin infusion in the outpatient management of diabetic teenagers. Diabetes Care 3:82–84
Brink SJ, Stewart C (1986) Insulin pump treatment in insulin-dependent diabetes mellitus. Children, adolescents, and young adults. JAMA 255:617–622
de Vries L, Grushka Y, Lebenthal Y, Shalitin S, Phillip M (2010) Factors associated with increased risk of insulin pump discontinuation in pediatric patients with type 1 diabetes. Pediatr Diabetes Aug 15 [Epub ahead of print]
Scaramuzza A, Iafusco D, Lombardo F, Rabbone I, Toni S, Italian Society of Paediatric Endocrinology and Diabetology (2008) Adolescent use of insulin and patient-controlled analgesia pump technology: a 10-year food and drug administration retrospective study of adverse events. Pediatrics 122:473–474
Pinelli L, Rabbone I, Salardi S, Toni S, Scaramuzza A, Bonfanti R, Cherubini V, Franzese A, Frongia AP, Lafusco D, Sulli N, Tumini S, Curto O, Miassimelli M, Diabetes Study Group of the Italian Society of Paediatric Endocrinology and Diabetology (2008) Insulin pump therapy in children and adolescents with type 1 diabetes: the Italian viewpoint. Acta Biomed 79:57–64
Havelka M, Lucanin JD, Lucanin D (2009) Biopsychosocial model—the integrated approach to health and disease. Coll Antropol 33:303–310
Malik JA, Koot HM (2009) Explaining the adjustment of adolescents with type 1 diabetes: role of diabetes-specific and psychosocial factors. Diabetes Care 32:774–779
Hasken J, Kresl L, Nydegger T, Temme M (2010) Diabulimia and the role of school health personnel. J Sch Health 80:465–469
Iafusco D, Vanelli M, Gugliotta M, Iovane B, Chiari G, Prisco F (2004) Prevalence of eating disorders in young patients with type 1 diabetes from two different Italian cities. Diabetes Care 27:2278
Iafusco D, Confetto S, Prisco F, Lombardo F, Salzano G, De Luca F (2006) The egg or the chicken? Should good compliance to multi-injection insulin therapy be a criterion for insulin pump therapy, or does insulin pump therapy improve compliance? J Pediatr 148:421
Conflict of interest
None of the authors (neither the one listed in the title page nor the one listed in the “Appendix”) has any conflict of interest to declare. The study was not sponsored.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study is conducted for the Diabetes Study Group of the Italian Society of Paediatric Endocrinology and Diabetology (ISPED). All authors are listed in the “Appendix”.
Appendix
Appendix
R. Lera, Alessandria
V. Cherubini, Ancona
C. Zecchino, Department of Pediatrics, Bari
E. Frezza, Policlinico Giovanni XXIII, Bari
S. Zucchini, S. Salardi, Bologna
P. Frongia, Brotzu Hospital, Cagliari
D. Lo Presti, Catania
F. Citriniti, Catanzaro
S. Tumini, F. Chiarelli, Chieti
L. Spallino, Como
N. Lazzaro, Crotone
S. Toni, Firenze
G. d’Annunzio, R. Lorini, Genova
M. Bruzzese, Locri
F. Lombardo, G. Salzano, F. De Luca, Messina
R. Bonfanti, F. Meschi, San Rafael Institute, Milano
A. Scaramuzza, G. V. Zuccotti, University of Milano, Luigi Sacco Hospital, Milano
D. Iafusco, F. Prisco, Second University, Napoli
P. Buono, A. Franzese, Federico II University, Napoli
F. Cadario, Novara
V. Calcaterra, Pavia
L. Iughetti, Reggio Emilia
R. Schiaffini, M. Cappa, Bambin Gesù Hospital, Roma
M. Delvecchio, San Giovanni Rotondo
I. Rabbone, F. Cerutti, Torino
F. Fontana, Tortona
L. Guearraggio, Tradate
A. Salvatoni, Varese
Rights and permissions
About this article
Cite this article
Lombardo, F., Scaramuzza, A.E. & Iafusco, D. Failure of glycated hemoglobin drop after continuous subcutaneous insulin infusion initiation may indicate patients who discontinue: a 4-year follow-up study in children and adolescents with type 1 diabetes. Acta Diabetol 49 (Suppl 1), 99–105 (2012). https://doi.org/10.1007/s00592-011-0344-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-011-0344-3