Abstract
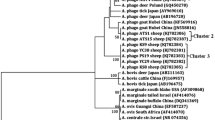
Rickettsia from the genus Anaplasma is obligated intracellular microorganism of animal host and human. The aim of this study was to investigate the prevalence and distribution of Anaplasma phagocytophilum in domesticated small ruminant of Iran. Three hundred seventy blood samples of sheep and goat were collected from different regions in Hamedan province, Iran. The DNA was extracted from blood samples and subsequently, 16S rRNA and MSP4 genes were analyzed by Nested-PCR method and overall 1.08% (4/370) of domesticated small ruminants were positive for A. phagocytophilum infection. The data recorded were analyzed by the Chi-square test and did not show any significant relation between infection and variables such as location, tick infestion, age, and sex of the host (p > 0.05). No significant correlation was found between the altitude and the Anaplasma species infection (Mann-Whitney test: p > 0.05). The present research is the first report of A. phagocytophilum infection in Iranian domesticated small ruminants and their zoonotic potential as a carrier of A. phagocytophilum.





Similar content being viewed by others
References
Ahmadi-Hamedani M, Khaki Z, Rahbari S, Kazemi B, Bandehpour M (2009) Molecular identification of anaplasmosis in goats using a new PCR-RFLP method. Iran J Vet Res 10(4):367–372
Alberdi MP, Walker AR, Urquhart KA (2000) Field evidence that roe deer (Capreolus capreolus) are a natural host for Ehrlichia phagocytophila. Epidemiol Infect 124:315–323
Bashiribod H, Kazemi B, Eslami G, Bigdeli E, Bandehpour M, Rahbarian N, Ramezani Z (2004) First molecular detection of Anaplasma phagocytophilum in Ixodes ricinus ticks in Iran. J Med Sci 4(4):282–286
Chapman AS, Bakken JS, Folk SM, Paddock CD, Bloch KC, Krusell A, Sexton DJ, Buckingham SC, Marshall GS, Storch GA, Dasch GA, McQuiston JH, Swerdlow DL, Dumler SJ, Nicholson WL, Walker DH, Eremeeva ME, Ohl CA (2006) Tickborne rickettsial diseases working group; CDC. Diagnosis and management of tickborne rickettsial diseases: Rocky Mountain spotted fever, ehrlichioses, and anaplasmosis—United States: a practical guide for physicians and other health-care and public health professionals. MMWR Recomm Rep 55(RR-4):1–27
Chen SM, Dumler JS, Bakken JS, Walker DH (1994) Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J Clin Microbiol 32:589–595
Dikmans G (1950) The transmission of anaplasmosis. Am J Vet Res 11:5–16
Dumler JS, Barbe AF, Bekker CP, Dasch GH, Ray SC, Rikihisa Y, Rurangirwa FR (2001) Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and 'HGE agent' as subjective synonyms of Ehrlichia phagocytophila. Int J Syst Evol Microbiol 51:2145–2165
Dumler JS, Choi KS, Garcia-Garcia JC, Barat NS, Scorpio DG, Garyu JW, Grab DJ, Bakken JS (2005) Human granulocytic anaplasmosis and Anaplasma phagocytophilum. Emerg Infect Dis 11:1828–1834
Ewing SA (1981) Transmission of Anaplasma marginale by arthropods. In: Proceedings of the 7th National Anaplasmosis Conference, (Mississippi State University, MS. USA) 395–423
Foil LD (1989) Tabanids as vectors of disease agents. Parasitol Today 5:88–96
Gharekhani J, Gerami-Sadeghian A, Sadeghi-Dehkordi Z, Youssefi MR (2015) Determination of hard tick species (Acarina: Ixodidae) on sheep and cattle in Hamedan Province, Iran. J Coast Life Med 3:612–615
Hosseini-Vasoukolaei N, Oshaghi MA, Shayan P, Vatandoost H, Babamahmoudi F, Yaghoobi-Ershadi MR, Telmadarraiy Z, Mohtarami F (2014) Anaplasma infection in ticks, livestock and human in Ghaemshahr, Mazandaran Province, Iran. Iran J Arthropod-Borne Dis 8(2):204–211
Jalali SM, Khaki Z, Kazemi B, Bandehpour M, Rahbari S, Razi Jalali M, Yasini SP (2013) Molecular detection and identification of Anaplasma species in sheep from Ahvaz, Iran. Iran J Vet Res 14(1):50–56
Kocan KM, de la Fuente J, Guglielmone AA, Meléndez RD (2003) Antigens and alternatives for control of Anaplasma marginale infection in cattle. Clin Microbiol Rev 16:698–712
Milner JM, Beest FMV (2013) Ecological correlates of a tick-borne disease, Anaplasma phagocytophilum, in moose in southern Norway. Eur J Wildl Res 59(3):399–406
Noaman V, Shayan P (2009) Molecular detection of Anaplasma phagocytophilum in carrier cattle of Iran -first documented report. Iran J Microbiol 1(2):37–42
Noaman V, Shayan P, Shahmoradi AH (2009) Detection of Anaplasma ovis based on 16S rRNA gene by PCR-RFLP in sheep from central part of Iran. J Vet Med Lab 1:27–37 (In Persian)
Oter K, Cetinkaya H, Vurusaner C, Toparlak M, Ergunay K (2016) Molecular Detection and Typing of Anaplasma Species in Small Ruminants in Thrace Region of Turkey. doi: 10.9775/kvfld.2015.14075
Potgieter FT (1979) Epizootiology and control of anaplasmosis in South Africa. J S Afr Vet Assoc 50(4):367–372
Razmi GR, Dastjerdi K, Hosseini H, Naghibi A, Barati F, Aslani MR (2006) An epidemiological study on Anaplasma infection in cattle, sheep and goats in Mashhad suburb, Khorasan province, Iran. Ann N Y Acad Sci 1078:479–481
Rikihisa Y (1991) The tribe Ehrlichieae and ehrlichial diseases. Clin Microbiol Rev 4:286–308
Robinson MT, Shaw SE, Morgan ER (2009) Anaplasma phagocytophilum infection in a multi-species deer community in the New Forest, England. Eur J Wildl Res 55:439–442
Rymaszewska A, Grenda S (2008) Bacteria of the genus Anaplasma- characteristics of Anaplasma and their vectors: a review. Vet Med Czech 53(11):573–584
Silaghi C, Scheuerle MC, Friche Passos LM, Thiel C, Pfister K (2011) PCR detection of Anaplasma phagocytophilum in goat flocks in an area endemic for tick-borne fever in Switzerland. Parasite 18(1):57–62
Stuen S (2007) Anaplasma phagocytophilum—the most widespread tick-borne infection in animals in Europe. Vet Res Commun 31(suppl 1):79–84
Woldehiwet Z (2008) Immune evasion and immunosuppression by Anaplasma phagocytophilum, the causative agent of tick-borne fever of ruminants and human granulocytic anaplasmosis. Vet J 175:37–44
Yang J, Liu Z, Guan G, Liu Q, Li Y, Chen Z, Ma M, Liu A, Ren Q, Luo J, Yin H (2013) Prevalence of Anaplasma phagocytophilum in ruminants, rodents and ticks in Gansu, North-Western China. J Med Microbiol 62:254–258
Yang J, Li Y, Liu Z, Liu J, Niu Q, Ren Q, Chen Z, Guan G, Luo J, Yin H (2015) Molecular detection and characterization of Anaplasma spp. in sheep and cattle from Xinjiang, Northwest China. Parasit Vectors. doi:10.1186/s13071-015-0727-3
Acknowledgements
The authors would like to thank Mrs. Fatemeh Khademi, Mr. Abbas Yousefi, and Mr. Masoud Moradi for their help in collecting the samples and to Mis. Fariba Nouri for her constant encouragements, guidance, and support throughout the present study. This paper is a part of Ph.D thesis of the senior author and financially supported by Science and Research Branch of Islamic Azad University, Tehran, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study was funded by the Science and Research Branch of Islamic Azad University, Tehran.
Rights and permissions
About this article
Cite this article
Yousefi, A., Rahbari, S., Shayan, P. et al. Molecular evidence of Anaplasma phagocytophilum: an emerging tick-borne pathogen in domesticated small ruminant of Iran; first report. Comp Clin Pathol 26, 637–642 (2017). https://doi.org/10.1007/s00580-017-2429-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-017-2429-z