Abstract
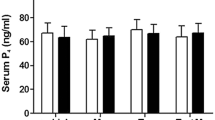
Release of prolactin, a milk-secreting hormone, is normally inhibited by the neurotransmitter dopamine. Numerous attempts have been made to explain the effects of environmental stress on the level of prolactin, proposing a basic role for cortisol in the modulation of the hormone secretion. The pharmacologic properties of antagonists in the release of prolactin, during the anovulatory early postpartum period, were examined in this research. This study included ten 3-year-old Mehraban ewes that gave birth to their third lambs, at least 2 weeks before the experiment. Each animal was randomly assigned to receive either a corresponding dose of either metoclopramide (0.3 mg/kg BW; im; n = 5) or haloperidol (0.1 mg/kg BW; im; n = 5). During the 2 weeks prior to the start of the study, all ewes were allowed to acclimatize to their environment with free access to food and tap water, in order to avoid stress-induced influences on hormone levels resulting from environmental factors. Blood samples were collected from each ewe through the jugular vein via intravenous catheter immediately at the time of study entry, 2 and 4 h before and after every injection, with a time interval of 30 min. The serum recovered was assayed for prolactin, cortisol, and progesterone. Animals were assumed to be anestrous if the mean serum level of progesterone was lower than 0.4 ng/mL and that of prolactin higher than 80 mIU/L. One-way ANOVA revealed that the mean serum concentrations of prolactin measured at 30 and 90 min after metoclopramide administration were statistically different compared with the control but showed no difference at other time points. The secretion of cortisol prior to the treatment and afterward was statistically similar; however, under haloperidol administration, it increased and the difference was statistically significant (p < 0.05). A corresponding cortisol-induced prolactin increase did not become evident under haloperidol medication, resulting in a non-significant difference between the two cortisol values found before and after haloperidol administration (p > 0.05). In conclusion, the pharmacologic properties of an antagonist to release prolactin, during the anovulatory early postpartum period, may depend on the presence of dopaminergic D2 receptor subtypes. In addition to its specificity for D2 receptors, the bioactivity of that antagonist may also play a role in the control of the secretion.


Similar content being viewed by others
References
Ahmadzadeh A, Barnes MA, Gwazdauskas FC, Akers RM (2006) Dopamine antagonist alters serum cortisol and prolactin secretion in lactating Holstein cows. J Dairy Sci 89:2051–2055
Angelopoulos EK, Markianos M, Daskalopoulou EG, Hatzimanolis J, Tzemos J (2002) Changes in central serotonergic function as a correlate of duration of illness in paranoid schizophrenia. Psychiatry Res 110:9–17
Boesgaard S, Hagen C, Andersen AN, Fenger M, Eldrup E (1990) Effect of dopamine, dopamine D-1 and D-2 receptor modulation on ACTH and cortisol levels in normal men and women. Acta Endocrinol 122:29–36
Bowman RE, MacLusky NJ, Sarmiento Y, Frankfurt M, Gordon M, Luine VN (2004) Sexually dimorphic effects of prenatal stress on cognition, hormonal responses, and central neurotransmitters. Endocrinology 145:3778–3787
Browning R, Gissendanner SJ, Wakefield T (2000) Ergotamine alters plasma concentrations of glucagon, insulin, cortisol, and triiodothyronine in cows. J Anim Sci 78:690–698
Cameron V, Espiner EA, Nicholls MG, Parratt S (1987) Effect of metoclopramide on adrenal secretion in sheep: influence of dexamethasone and sodium intake. J Steroid Biochem 26:717–721
Colthorpe KL, Anderson ST, Martin GB, Curlewis JD (1998) Hypothalamic dopamine D1 receptors are involved in the stimulation of prolactin secretion by high environmental temperature in the female sheep. J Neuroendocrinol 10:503–509
Debeljuk L, Lasaga M (2006) Tachykinins and the control of prolactin secretion. Peptides 27:3007–3019
Gentili S (2006) Regulation of SOCS-3 expression in fetal sheep tissues. The University of Adelaide, Adelaide
Horváth KM, Bánky Z, Tóth BE, Halász B, Nagy GM (2001) Effect of adrenalectomy and denamethasone treatment on prolactin secretion of lactating rats. Brain Res Bulletin 56:589–592
Iranmanesh A, Mulligan T, Veldhuis JD (1999) Mechanisms subserving the physiological nocturnal relative hypoprolactinemia of healthy older men: dual decline in prolactin secretory burst mass and basal release with preservation of pulse duration, frequency, and inter-pulse interval. A general clinical research study. J Clin Endocrinol Metab 84:1083–1090
Kan SF, Kau MM, Ho LLT, Wang PS (2003) Inhibitory effects of bromocriptine on corticosterone secretion in male rats. Eur J Pharmacol 468:141–149
Kapoor A (2008) Effects of acute periods of prenatal stress on behaviour and endocrine function in guinea pigs. The University of Toronto, Toronto
Koch A, Hoppen HO, Dieleman SJ, Kooistra HS, Günzel-Apel AR (2006) Effects of the dopamine agonist cabergoline on the pulsatile and TRH-induced secretion of prolactin, LH, and testosterone in male beagle dogs. Theriogenology 65:1666–1677
Korte SM (2001) Corticosteroids in relation to fear, anxiety and psychopathology. Neurosci Biobehav Rev 25:117–142
Matsuda M, Imaoka T, Vomachka AJ, Gudelsky GA, Hou Z, Mistry M, Bailey JP, Nieport KM, Walther DJ, Bader M, Horseman ND (2004) Serotonin regulates mammary gland development via an autocrine–paracrine loop. Dev Cell 6:193–203
Matteri RL, Carroll JA, Dyer CJ (2000) Neuroendocrine responses to stress. In: Moberg GP, Mench JA (eds) The biology of stress: basic principles and implications for animal welfare. CAB International, Wallingford, pp 43–76
Olanow CW, Koller WC (1998) An algorithm (decision tree) for the management of Parkinson’s disease: treatment guidelines. Neurology 50:1–57
Parncutt R (2006) Prenatal development. In: McPherson GE (ed) The child as musician. Oxford University Press, Oxford, pp 1–31
Parsons R, Stephenson M, Nicholson C (2005) Profitability of small ruminant farmstead dairy processing operations. Proceedings of the 11th annual Great Lakes Dairy Sheep Symposium Burlington, Vermont, USA; November 3–5
Patterson-Buckendahl P, Kubovcakova L, Krizanova O, Pohorecky LA, Kvetnansky R (2005) Ethanol consumption increases rat stress hormones and adrenomedullary gene expression. Alcohol 37:157–166
Rensis FD, Spattini G, Ballabio R, Scaramuzzi RJ (2006) The effect of administering a dopamine agonist (cabergoline) on follicular and luteal development during pro-estrus and estrus in the female greyhound. Theriogenology 66:887–895
Sarkar M, Prakash BS (2005) Circadian variations in plasma concentrations of melatonin and prolactin during breeding and non-breeding seasons in yak (Poephagus grunniens L.). Anim Reprod Sci 90:149–162
Segal M, Avital A, Rojas M, Hausvater N, Sandbank S, Liba D, Moguillansky L, Tal I, Weizman A (2004) Serum prolactin levels in unmedicated first-episode and recurrent schizophrenia patients: a possible marker for the disease’s subtypes. Psychiatry Res 127:227–235
Seki K, Kato T, Sekiya S (1997) Corticotrophin and vasopressin responses to metoclopramide in patients with hypothalamic amenorrhea. Clin Endocrinol 46:203–207
Sevi A, Casamassima D, Pulina G, Pazzona A (2009) Factors of welfare reduction in dairy sheep and goats. Ital J Anim Sci 8:81–101
Silanikove N, Shamay A, Shinder D, Moran A (2000) Stress down regulates milk yield in cows by plasmin induced β-casein product that blocks K+ channels on the apical membranes. Life Sci 67:2201–2212
Sowers JR, Beck FW, Stern N, Asp N (1983) Effects of metoclopramide on plasma corticosteroid levels in sheep. Endocrinology 113:903–906
Van den Bergh BRH (1992) Maternal emotions during pregnancy and fetal and neonatal behaviour. In: Nijhuis JG (ed) Fetal behaviour. Oxford University Press, Oxford, pp 157–208
Welberg LAM, Seckl JR (2001) Prenatal stress, glucocorticoids and the programming of the brain. J Neuroendocrinol 13:113–128
Acknowledgment
Authors would like to express their thanks and gratitude to Mr. J. Jalaie for his technical assistance in the clinical experiments and Dr. M. Ansari for her help and advice in the statistical analysis of the study records. The financial support by Shiraz University is also appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajaian, H., Nazifi, S., Bidadkosh, A. et al. The modulatory role of cortisol on prolactin secretion in anestrus Iranian fat-tailed ewes. Comp Clin Pathol 21, 565–570 (2012). https://doi.org/10.1007/s00580-010-1131-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-010-1131-1