Abstract
Background
The optimal retreatment regimen for patients with hepatitis C virus (HCV) infection who failed interferon-free, direct-acting antiviral (DAA) therapy is undetermined. In this study, we aimed to evaluate the efficacy and safety of 12-week retreatment with ledipasvir (LDV) and sofosbuvir (SOF) with add-on ribavirin (RBV) for patients who previously failed to respond to HCV-NS5A inhibitor, daclatasvir (DCV), and HCV-NS3 inhibitor, asunaprevir (ASV), therapy.
Methods
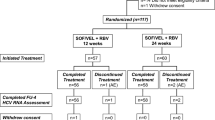
This multicenter, prospective study enrolled 15 patients with genotype-1 HCV infection who failed DCV/ASV combination therapy. They were retreated with SOF, LDV, and RBV for 12 weeks and underwent physical examinations and blood tests at baseline, during treatment, and after therapy. At baseline and relapse, NS3/NS5A and NS5B resistance-associated variants (RAVs) were evaluated.
Results
Of the 15 enrolled patients, 73.3% (11/15), 86.7% (13/15), and 0% (0/15) had RAVs in NS3 D168A/V/T/E, NS5A L31I/M/F/V plus Y93H, and NS5B S282T, respectively. Overall, 86.7% (13/15) of patients achieved a sustained viral response, and all patients completed therapy. No patients experienced severe adverse events. Two patients who failed to respond to SOF, LDV, and RBV combination therapy were elderly women, had the IL28B non-TT genotype, and NS5A RAVs in L31I/Y93H or NS5A A92 K at baseline.
Conclusions
This study revealed that SOF, LDV, and RBV combination therapy was effective and well-tolerated for patients with genotype-1 HCV infection who failed DCV and ASV combination therapy. Thus, RBV added to DAA therapy for difficult-to-treat patients might improve treatment outcomes.

Similar content being viewed by others
Abbreviations
- DAAs:
-
Direct-acting antivirals
- HCV:
-
Hepatitis C virus
- PI:
-
Protease inhibitor
- RAVs:
-
Resistance-associated variants
- SVR:
-
Sustained virological response
References
Suda G, Kudo M, Nagasaka A, et al. Efficacy and safety of daclatasvir and asunaprevir combination therapy in chronic hemodialysis patients with chronic hepatitis C. J Gastroenterol. 2016;1(7):733–40.
Mizokami M, Yokosuka O, Takehara T, et al. Ledipasvir and sofosbuvir fixed-dose combination with and without ribavirin for 12 weeks in treatment-naive and previously treated Japanese patients with genotype 1 hepatitis C: an open-label, randomised, phase 3 trial. Lancet Infect Dis. 2015;15(6):645–53.
Kumada H, Suzuki Y, Ikeda K, et al. Daclatasvir plus asunaprevir for chronic HCV genotype 1b infection. Hepatology. 2014;59(6):2083–91.
Ito J, Suda G, Yamamoto Y, et al. Prevalence and characteristics of naturally occurring sofosbuvir resistance-associated variants in patients with hepatitis C virus genotype 1b infection. Hepatol Res. 2016;46(13):1294–303.
Suzuki Y, Ikeda K, Suzuki F, et al. Dual oral therapy with daclatasvir and asunaprevir for patients with HCV genotype 1b infection and limited treatment options. J Hepatol. 2013;58(4):655–62 Epub 2012 Nov 23.
McCown MF, Rajyaguru S, Le Pogam S, et al. The hepatitis C virus replicon presents a higher barrier to resistance to nucleoside analogs than to nonnucleoside polymerase or protease inhibitors. Antimicrob Agents Chemother. 2008;52(5):1604–12.
European Association for the Study of the Liver. Electronic address eee. EASL Recommendations on Treatment of Hepatitis C 2016. J Hepatol. 2017;66(1):153–94.
Akuta N, Sezaki H, Suzuki F, et al. Retreatment efficacy and predictors of ledipasvir plus sofosbuvir to HCV genotype 1 in Japan. J Med Virol. 2017;89(2):284–90.
Curry MP, O’Leary JG, Bzowej N, et al. Sofosbuvir and Velpatasvir for HCV in patients with decompensated cirrhosis. N Engl J Med. 2015;373(27):2618–28.
Suda G, Yamamoto Y, Nagasaka A, et al. Serum granulysin levels as a predictor of serious telaprevir-induced dermatological reactions. Hepatol Res. 2015;45(8):837–45.
Tanaka T, Tsukiyama-Kohara K, Yamaguchi K, et al. Significance of specific antibody assay for genotyping of hepatitis C virus. Hepatology. 1994;19(6):1347–53.
Vallet-Pichard A, Mallet V, Nalpas B, et al. FIB-4: an inexpensive and accurate marker of fibrosis in HCV infection. Comparison with liver biopsy and fibrotest. Hepatology. 2007;46(1):32–6.
Ohnishi Y, Tanaka T, Ozaki K, et al. A high-throughput SNP typing system for genome-wide association studies. J Hum Genet. 2001;46(8):471–7.
Ochi H, Maekawa T, Abe H, et al. ITPA polymorphism affects ribavirin-induced anemia and outcomes of therapy—a genome-wide study of Japanese HCV virus patients. Gastroenterology. 2010;139(4):1190–7.
Gilead Sciences HARVONI combination tablets® (sofosbuvir + ledipasvir), for oral use: PRESCRIBING INFORMATION 2015.
Osinusi A, Meissner EG, Lee YJ, et al. Sofosbuvir and ribavirin for hepatitis C genotype 1 in patients with unfavorable treatment characteristics: a randomized clinical trial. JAMA. 2013;310(8):804–11.
Gane EJ, Stedman CA, Hyland RH, et al. Nucleotide polymerase inhibitor sofosbuvir plus ribavirin for hepatitis C. N Engl J Med. 2013;368(1):34–44.
Wong KA, Worth A, Martin R, et al. Characterization of Hepatitis C virus resistance from a multiple-dose clinical trial of the novel NS5A inhibitor GS-5885. Antimicrob Agents Chemother. 2013;57(12):6333–40.
Friborg J, Zhou N, Han Z, et al. In vitro assessment of re-treatment options for patients with hepatitis C virus genotype 1b infection resistant to daclatasvir plus asunaprevir. Infect Dis Ther. 2014;4(1):137–44.
Suda G, Nagasaka A, Yamamoto Y, et al. Safety and efficacy of daclatasvir and asunaprevir in hepatitis C virus infected patients with renal impairment. Hepatol Res. 2016. doi: 10.1111/hepr.12851.
Wilson EM, Kattakuzhy S, Sidharthan S, et al. Successful retreatment of chronic HCV genotype-1 infection with ledipasvir and sofosbuvir after initial short course therapy with direct-acting antiviral regimens. Clin Infect Dis. 2016;62(3):280–8.
Hezode C, Chevaliez S, Scoazec G, et al. Retreatment with sofosbuvir and simeprevir of patients with hepatitis C virus genotype 1 or 4 who previously failed a daclatasvir-containing regimen. Hepatology. 2016;63(6):1809–16.
Kai Y, Hikita H, Tatsumi T, et al. Emergence of hepatitis C virus NS5A L31 V plus Y93H variant upon treatment failure of daclatasvir and asunaprevir is relatively resistant to ledipasvir and NS5B polymerase nucleotide inhibitor GS-558093 in human hepatocyte chimeric mice. J Gastroenterol. 2015;50(11):1145–51.
Uchida Y, Kouyama JI, Naiki K, et al. Development of rare resistance-associated variants that are extremely tolerant against NS5A inhibitors during daclatasvir/asunaprevir therapy by a two-hit mechanism. Hepatol Res. 2016;46(12):1234–46.
Iio E, Shimada N, Abe H, et al. Efficacy of daclatasvir/asunaprevir according to resistance-associated variants in chronic hepatitis C with genotype 1. J Gastroenterol. 2017;52(1):94–103.
Akuta N, Sezaki H, Suzuki F, et al. Ledipasvir plus sofosbuvir as salvage therapy for HCV genotype 1 failures to prior NS5A inhibitors regimens. J Med Virol. 2017. doi:10.1002/jmv.24767.
Morio K, Imamura M, Kawakami Y, et al. ITPA polymorphism effects on decrease of hemoglobin during sofosbuvir and ribavirin combination treatment for chronic hepatitis C. J Gastroenterol. 2016 [Epub ahead of print].
Acknowledgements
This study was supported in part by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan and the Japan Agency for Medical Research and development. The principal investigators of the NORTE study sites are listed below: Junichi Yoshida (JCHO Sapporo Hokushin Hospital), Akira Fuzinaga, (Abashiri-Kosei General Hospital), Keisuke Shinada (Keiwakai Ebetsu Hospital) Shuichi Muto (National Hospital Organization Hokkaido Medical Center), Takashi Meguro (Hokkaido Gastroenterology Hospital), Munenori Okamoto (Aiiku Hospital), Mineo Kudo (Sapporo Hokuyu Hospital), Nobuaki Akakura (NTT EAST Sapporo Hospital), Minoru Uebayashi (Japanese Red Cross Kitami Hospital), Kanji Katou (Iwamizawa Municipal General Hospital), Yasuyuki Kunieda (Wakkanai City Hospital), Miki Tateyama (Tomakomai Nissho Hospital), Atshuhiko Kawakami (Sapporo Century Hospital), and Izumi Tsunematsu (Touei hospital).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
N. Sakamoto received lecture fees from Bristol Myers Squibb and Pharmaceutical K.K., grants and endowments from MSD K.K. and Chugai Pharmaceutical Co., Ltd., and a research grant from Gilead Sciences, Inc. G. Suda received research grants from Bristol Myers Squibb and MSD K.K. Y. Tanaka received lecture fees from Bristol-Myers Squibb, MSD K.K., Chugai Pharmaceutical Co., Ltd., AbbVie Inc., Janssen Pharmaceutical K.K., Gilead Sciences, and a research grant from Bristol-Myers Squibb, Chugai Pharmaceutical Co., Ltd., and AbbVie Inc. The other authors have nothing to disclose.
Rights and permissions
About this article
Cite this article
Suda, G., Ogawa, K., Yamamoto, Y. et al. Retreatment with sofosbuvir, ledipasvir, and add-on ribavirin for patients who failed daclatasvir and asunaprevir combination therapy. J Gastroenterol 52, 1122–1129 (2017). https://doi.org/10.1007/s00535-017-1328-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-017-1328-z