Abstract
Background
Environmental factors are suggested to affect the pathogenesis of several diseases, including inflammatory bowel disease (IBD). The seasonality of disease onset and exacerbation in IBD, however, are not well established. We herein aimed to clarify the disease seasonality and to investigate the underlying characteristics in IBD patients exhibiting seasonality of the disease course.
Methods
This was a multicenter observational study comprising 20 institutions (Osaka Gut Forum) in Japan. Data were collected from November 2013 to August 2014 using survey forms for physicians and questionnaires for patients. Multivariate analysis was performed to clarify the independent factors affecting disease seasonality.
Results
A total of 1055 patients, including 298 patients with Crohn’s disease (CD) and 757 patients with ulcerative colitis (UC), were enrolled. The proportion of CD patients with disease onset in the summer was significantly larger than that in the other seasons, while UC patients exhibited no seasonality of disease onset. More than half of the IBD patients (51.1%) experienced seasonal exacerbation of IBD, and winter was the most common season for disease exacerbation in both CD and UC patients. Seasonality of disease onset and exacerbation was observed in young-onset patients (≤40 years old), but not in elderly-onset patients. Age at onset was independently associated with the seasonality of both disease onset and exacerbation.
Conclusions
Seasonality of disease onset and exacerbation was observed especially in young-onset IBD patients. Underlying pathophysiologic triggers for disease initiation and exacerbation may be influenced by age at disease onset.




Similar content being viewed by others
References
Fuyuno Y, Yamazaki K, Takahashi A, et al. Genetic characteristics of inflammatory bowel disease in a Japanese population. J Gastroenterol. 2016;51:672–81.
Ananthakrishnan AN. Epidemiology and risk factors for IBD. Nat Rev Gastroenterol Hepatol. 2015;12:205–17.
Rogler G, Vavricka S. Exposome in IBD: recent insights in environmental factors that influence the onset and course of IBD. Inflamm Bowel Dis. 2015;21:400–8.
Boctor FN, Charmy RA, Cooper EL. Seasonal differences in the rhythmicity of human male and female lymphocyte blastogenic responses. Immunol Invest. 1989;18:775–84.
Nelson RJ. Seasonal immune function and sickness responses. Trends Immunol. 2004;25:187–92.
Kokolus KM, Capitano ML, Lee CT, et al. Baseline tumor growth and immune control in laboratory mice are significantly influenced by subthermoneutral housing temperature. Proc Natl Acad Sci. 2013;110:20176–81.
Eng JW, Reed CB, Kokolus KM, et al. Housing temperature-induced stress drives therapeutic resistance in murine tumour models through β2-adrenergic receptor activation. Nat Commun. 2015;6:6426.
Shirai T, Magara KK, Motohashi S, et al. TH1-biased immunity induced by exposure to Antarctic winter. J Allergy Clin Immunol. 2003;111:1353–60.
Limketkai BN, Bayless TM, Brant SR, et al. Lower regional and temporal ultraviolet exposure is associated with increased rates and severity of inflammatory bowel disease hospitalisation. Aliment Pharmacol Ther. 2014;40:508–17.
Khot A, Burn R, Evans N, et al. Seasonal variation and time trends in childhood asthma in England and Wales 1975–81. Br Med J (Clin Res Ed). 1984;289:235–7.
Stewart S, McIntyre K, Capewell S, et al. Heart failure in a cold climate. Seasonal variation in heart failure-related morbidity and mortality. J Am Coll Cardiol. 2002;39:760–6.
Nomura T, Ohkusa T, Araki A, et al. Influence of climatic factors in the incidence of upper gastrointestinal bleeding. J Gastroenterol Hepatol. 2001;16:619–23.
Fink AM, Mayer W, Steiner A. Seasonal variations of deep vein thrombosis and its influence on the location of the thrombus. Thromb Res. 2002;106:97–100.
Moum B, Aadland E, Ekbom A, et al. Seasonal variations in the onset of ulcerative colitis. Gut. 1996;38:376–8.
Aratari A, Papi C, Galletti B, et al. Seasonal variations in onset of symptoms in Crohn’s disease. Dig Liver Dis. 2006;38:319–23.
Romberg-Camps MJ, Hesselink-van de Kruijs MA, Schouten LJ, et al. Inflammatory Bowel Disease in South Limburg (the Netherlands) 1991–2002: incidence, diagnostic delay, and seasonal variations in onset of symptoms. J Crohns Colitis. 2009;3:115–24.
Riley SA, Mani V, Goodman MJ, et al. Why do patients with ulcerative colitis relapse? Gut. 1990;31:179–83.
Zeng L, Anderson FH. Seasonal change in the exacerbations of Crohn’s disease. Scand J Gastroenterol. 1996;31:79–82.
Lewis JD, Aberra FN, Lichtenstein GR, et al. Seasonal variation in flares of inflammatory bowel disease. Gastroenterology. 2004;126:665–73.
Koido S, Ohkusa T, Saito H, et al. Seasonal variations in the onset of ulcerative colitis in Japan. World J Gastroenterol. 2013;19:9063–8.
Bai A, Guo Y, Shen Y, et al. Seasonality in flares and months of births of patients with ulcerative colitis in a Chinese population. Dig Dis Sci. 2009;54:1094–8.
Jung YS, Song CS, Kim ER, et al. Seasonal variation in months of birth and symptom flares in Korean patients with inflammatory bowel disease. Gut Liver. 2013;7:661–7.
Ananthakrishnan AN, Shi HY, Tang W, et al. Systematic review and meta-analysis: phenotype and clinical outcomes of older-onset inflammatory bowel disease. J Crohns Colitis. 2016;10:1224–36.
Jeuring SF, van den Heuvel TR, Zeegers MP, et al. Epidemiology and long-term outcome of inflammatory bowel disease diagnosed at elderly age-an increasing distinct entity? Inflamm Bowel Dis. 2016;22:1425–34.
Shi HY, Chan FK, Leung WK, et al. Natural history of elderly-onset ulcerative colitis: results from a territory-wide inflammatory bowel disease registry. J Crohns Colitis. 2016;10:176–85.
Silverberg MS, Satsangi J, Ahmad T, et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol. 2005;19(Suppl A):5A–36A.
Van Assche G, Dignass A, Reinisch W, et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: special situations. J Crohns Colitis. 2010;4:63–101.
Jones A, McCurdy JD, Loftus EV, et al. Effects of antiviral therapy for patients with inflammatory bowel disease and a positive intestinal biopsy for cytomegalovirus. Clin Gastroenterol Hepatol. 2015;13:949–55.
Hintzpeter B, Mensink GB, Thierfelder W, et al. Vitamin D status and health correlates among German adults. Eur J Clin Nutr. 2008;62:1079–89.
Froicu M, Weaver V, Wynn TA, et al. A crucial role for the vitamin D receptor in experimental inflammatory bowel diseases. Mol Endocrinol. 2003;17:2386–92.
Froicu M, Zhu Y, Cantorna MT. Vitamin D receptor is required to control gastrointestinal immunity in IL-10 knockout mice. Immunology. 2006;117:310–8.
Froicu M, Cantorna MT. Vitamin D and the vitamin D receptor are critical for control of the innate immune response to colonic injury. BMC Immunol. 2007;8:5.
Cantorna MT, Munsick C, Bemiss C, et al. 1,25-Dihydroxycholecalciferol prevents and ameliorates symptoms of experimental murine inflammatory bowel disease. J Nutr. 2000;130:2648–52.
Ananthakrishnan AN, Cagan A, Gainer VS, et al. Normalization of plasma 25-hydroxy vitamin D is associated with reduced risk of surgery in Crohn’s disease. Inflamm Bowel Dis. 2013;19:1921–7.
Elson CO, Cong Y, McCracken VJ, et al. Experimental models of inflammatory bowel disease reveal innate, adaptive, and regulatory mechanisms of host dialogue with the microbiota. Immunol Rev. 2005;206:260–76.
Sartor RB. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics, and prebiotics. Gastroenterology. 2004;126:1620–33.
Faith JJ, Guruge JL, Charbonneau M, et al. The long-term stability of the human gut microbiota. Science. 2013;341:1237439.
Martinez C, Antolin M, Santos J, et al. Unstable composition of the fecal microbiota in ulcerative colitis during clinical remission. Am J Gastroenterol. 2008;103:643–8.
Dorshkind K, Montecino-Rodriguez E, Signer RA. The ageing immune system: is it ever too old to become young again? Nat Rev Immunol. 2009;9:57–62.
Stervbo U, Meier S, Mälzer JN, et al. Effects of aging on human leukocytes (part I): immunophenotyping of innate immune cells. Age (Dordr). 2015;37:9828.
Stervbo U, Bozzetti C, Baron U, et al. Effects of aging on human leukocytes (part II): immunophenotyping of adaptive immune B and T cell subsets. Age (Dordr). 2015;37:9829.
Claesson MJ, Cusack S, O’Sullivan O, et al. Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc Natl Acad Sci. 2011;108(Suppl 1):4586–91.
Guergova S, Dufour A. Thermal sensitivity in the elderly: a review. Ageing Res Rev. 2011;10:80–92.
Blatteis CM. Age-dependent changes in temperature regulation—a mini review. Gerontology. 2012;58:289–95.
Pimentel M, Chang M, Chow EJ, et al. Identification of a prodromal period in Crohn’s disease but not ulcerative colitis. Am J Gastroenterol. 2000;95:3458–62.
Acknowledgements
This work was supported by a Grant-in-Aid from the Japan Society for the Promotion of Science (Grant No. 26460969). We are grateful to Dr. E. Mita (National Hospital Organization Osaka National Hospital), Dr. H. Ito (Kinshukai Infusion Clinic), Dr. M. Yamamoto (Kinki Central Hospital of Mutual Aid Association of Public School Teachers), Dr. K. Tominaga (Kaizuka City Hospital), Dr. T. Kitada (Kawanishi City Hospital), Dr. Y. Okuda (Saiseikai Senri Hospital), and Dr. S. Ishii (Osaka General Medical Center) in the Osaka Gut Forum for their contributions to the data collection. Writing assistance: The manuscript was edited by SciTechEdit International, LLC (7012 East Mountain Brush Circle Highlands Ranch CO 80130 USA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
535_2017_1313_MOESM1_ESM.docx
Supplementary material 1 (DOCX 15 kb). Supplementary Table 1. Questions regarding onset age and season of onset or exacerbation in the patient questionnaire
535_2017_1313_MOESM2_ESM.docx
Supplementary material 2 (DOCX 15 kb). Supplementary Table 2. Patient characteristics associated with exacerbation seasonality in IBD patients
535_2017_1313_MOESM3_ESM.tif
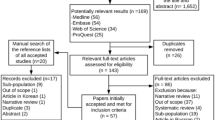
Supplementary material 3 (TIFF 2040 kb) Supplementary Fig. 1. Study flow chart. A total of 1055 patients were enrolled in the study, excluding 23 patients whose response regarding seasonality of onset or exacerbation was left vacant. We performed two analyses; one was an analysis of the seasonality of onset and the other was an analysis of the seasonality of exacerbation. We excluded 219 patients from the onset evaluation and 172 patients from the exacerbation evaluation because of inadequate responses (n = 836 and 883, respectively). Furthermore, analysis of the season of exacerbation was performed among patients who identified seasonal disease exacerbation [exacerbation seasonality (+)]. Prior to the analysis, we excluded 14 patients who did not respond with a definite season (e.g., turn of season)
535_2017_1313_MOESM4_ESM.tif
Supplementary material 4 (TIFF 942 kb) Supplementary Fig. 2. Ratio of IBD, CD, and UC patients who experienced seasonal exacerbation. More than half the CD and UC patients responded that disease activity was affected in a seasonal manner. A total of 883 IBD patients, comprising 257 CD patients and 626 UC patients, were analyzed
535_2017_1313_MOESM5_ESM.tif
Supplementary material 5 (TIFF 1123 kb) Supplementary Fig. 3. Ratio of IBD patients who experienced seasonal exacerbation subdivided by age at enrollment. One patient whose response to age at enrollment was left vacant was excluded from the exacerbation evaluation cohort, and a total of 882 patients were analyzed. Age at enrollment was not significantly different among the groups (p = 0.53)
535_2017_1313_MOESM6_ESM.tif
Supplementary material 6 (TIFF 3068 kb) Supplementary Fig. 4. Distribution of IBD patients who experienced seasonal exacerbation subdivided by age at onset. Eight patients whose response regarding age at onset was left vacant were excluded from the exacerbated season evaluation cohort, and a total of 429 patients were analyzed. Age at onset was not significantly different among the groups (p = 0.75)
Rights and permissions
About this article
Cite this article
Araki, M., Shinzaki, S., Yamada, T. et al. Age at onset is associated with the seasonal pattern of onset and exacerbation in inflammatory bowel disease. J Gastroenterol 52, 1149–1157 (2017). https://doi.org/10.1007/s00535-017-1313-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-017-1313-6