Abstract
Background
Milk fat globule-epidermal growth factor 8 (MFG-E8) promotes phagocytic clearance of apoptotic cells to maintain normal tissue homeostasis. However, its functions in intestinal inflammation and carcinogenesis are unknown.
Methods
Experimental colitis was induced in MFG-E8 knockout (KO) and wild-type (WT) mice by dextran sodium sulfate (DSS) administration. Colon tissues were used for assessments of colitis activity and epithelial proliferation. A mouse colitis-associated cancer (CAC) model was induced by intraperitoneal injection of azoxymethane (AOM) and then the animals were given a single administration of DSS. A sporadic colon cancer model was established by repeated intraperitoneal injections of AOM. The role of MFG-E8 in epithelial proliferation with or without treatment of siRNA targeting αv-integrin was examined in vitro using a WST-1 assay.
Results
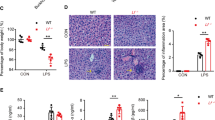
The severity of colitis in KO mice was greater than that in WT mice, while the proliferative potential of colonic epithelial cells in KO mice was lower during the regenerative phase. In both CAC and sporadic models, tumor size in KO was lower as compared to WT mice, while decreased tumor incidence was only found in the CAC model. In vitro findings showed that MFG-E8 promotes epithelial cell proliferation, and treatment with a siRNA targeting αv-integrin reduced the proliferation of Colon-26 cells stimulated with recombinant MFG-E8.
Conclusions
MFG-E8 promotes tumor growth regardless of the presence or absence of colonic inflammation, whereas colon tumor development is initiated by MFG-E8 under inflammatory conditions. These MFG-E8 functions may be dependent on integrin-mediated cellular signaling.







Similar content being viewed by others
References
Krysko DV, D`Herde K, Vandenabeele P. Clearance of apoptotic and necrotic cells and its immunological consequences. Apoptosis. 2006;11:1709–26.
Nagata S, Hanayama R, Kawane K. Autoimmunity and the clearance of dead cells. Cell. 2010;140:619–30.
Gregory CD, Devitt A. The macrophage and the apoptotic cell: an innate immune interaction viewed simplistically. Immunology. 2004;113:1–14.
Lauber K, Blumenthal SG, Waibel M, Wesselborg S. Clearance of apoptotic cells: getting rid of the corpses. Mol Cell. 2004;14:277–87.
Savill J, Fadok V. Corpse clearance defines the meaning of cell death. Nature. 2000;407:784–8.
Miyasaka K, Hanayama R, Tanaka M, Nagata S. Expression of milk fat globule epidermal growth factor 8 in immature dendritic cells for engulfment of apoptotic cells. Eur J Immunol. 2004;34:1414–22.
Hanayama R, Tanaka M, Miwa K, Shinohara A, Iwamatsu A, Nagata S. Identification of a factor that links apoptotic cells to phagocytes. Nature. 2002;417:182–7.
Asano K, Miwa M, Miwa K, Hanayama R, Nagase H, Nagata S, et al. Masking of phosphatidylserine inhibits apoptotic cell engulfment and induces autoantibody production in mice. J Exp Med. 2004;200:459–67.
Hanayama R, Tanaka M, Miyasaka K, Aozasa K, Koike M, Uchiyama Y, et al. Autoimmune disease and impaired uptake of apoptotic cells in MFG-E8-deficient mice. Science. 2004;304:1147–50.
Ait-Oufella H, Kinugawa K, Zoll J, Simon T, Boddaert J, Heeneman S, et al. Lactadherin deficiency leads to apoptotic cell accumulation and accelerated atherosclerosis in mice. Circulation. 2007;115:2168–77.
Thorp E, Tabas I. Mechanisms and consequences of efferocytosis in advanced atherosclerosis. J Leukoc Biol. 2009;86:1089–95.
Fuller AD, Van Eldik LJ. MFG-E8 regulates microglial phagocytosis of apoptotic neurons. J Neuroimmune Pharmacol. 2008;3:246–56.
Kusunoki R, Ishihara S, Aziz M, Oka A, Tada Y, Kinoshita Y. Roles of milk fat globule-epidermal growth factor 8 in intestinal inflammation. Digestion. 2012;85:103–7.
Aziz MM, Ishihara S, Mishima Y, Oshima N, Moriyama I, Yuki T, et al. MFG-E8 attenuates intestinal inflammation in murine experimental colitis by modulating osteopontin-dependent alphavbeta3 integrin signaling. J Immunol. 2009;182:7222–32.
Otani A, Ishihara S, Aziz MM, Oshima N, Mishima Y, Moriyama I, et al. Intrarectal administration of milk fat globule epidermal growth factor-8 protein ameliorates murine experimental colitis. Int J Mol Med. 2012;29:349–56.
Mishiro T, Kusunoki R, Otani A, Ansary MM, Tongu M, Harashima N, et al. Butyric acid attenuates intestinal inflammation in murine DSS-induced colitis model via milk fat globule-EGF factor 8. Lab Invest. 2013;93:834–43.
Chogle A, Bu HF, Wang X, Brown JB, Chou PM, Tan XD. Milk fat globule-EGF factor 8 is a critical protein for healing of dextran sodium sulfate-induced acute colitis in mice. Mol Med. 2011;17:502–7.
Komura H, Miksa M, Wu R, Goyert SM, Wang P. Milk fat globule epidermal growth factor-factor VIII is down-regulated in sepsis via the lipopolysaccharide-CD14 pathway. J Immunol. 2009;182:581–7.
Cui T, Miksa M, Wu R, Komura H, Zhou M, Dong W, et al. Milk fat globule epidermal growth factor 8 attenuates acute lung injury in mice after intestinal ischemia and reperfusion. Am J Respir Crit Care Med. 2010;181:238–46.
Bu HF, Zuo XL, Wang X, Ensslin MA, Koti V, Hsueh W, et al. Milk fat globule-EGF factor 8/lactadherin plays a crucial role in maintenance and repair of murine intestinal epithelium. J Clin Invest. 2007;117:3673–83.
Wu R, Dong W, Wang Z, Jacob A, Cui T, Wang P. Enhancing apoptotic cell clearance mitigates bacterial translocation and promotes tissue repair after gut ischemia–reperfusion injury. Int J Mol Med. 2012;30:593–8.
Zhao QJ, Yu YB, Zuo XL, Dong YY, Li YQ. Milk fat globule-epidermal growth factor 8 is decreased in intestinal epithelium of ulcerative colitis patients and thereby causes increased apoptosis and impaired wound healing. Mol Med. 2012;18:497–506.
Ajakaiye MA, Jacob A, Wu R, Yang WL, Nicastro J, Coppa GF, et al. Recombinant human MFG-E8 attenuates intestinal injury and mortality in severe whole body irradiation in rats. PLoS One. 2012;7:e46540.
Silvestre JS, Théry C, Hamard G, Boddaert J, Aguilar B, Delcayre A, et al. Lactadherin promotes VEGF-dependent neovascularization. Nat Med. 2005;11:499–506.
Carrascosa C, Obula RG, Missiaglia E, Lehr HA, Delorenzi M, Frattini M, et al. MFG-E8/lactadherin regulates cyclins D1/D3 expression and enhances the tumorigenetic potential of mammary epithelial cells. Oncogene. 2012;31:1521–32.
Sugano G, Bernard-Pierrot I, Laé M, Battail C, Allory Y, Stransky N, et al. Milk fat globule-epidermal growth factor-factor VIII (MFGE8)/lactadherin promotes bladder tumor development. Oncogene. 2011;30:642–53.
Jinushi M, Nakazaki Y, Carrasco DR, Draganov D, Souders N, Johnson M, et al. Milk fat globule EGF-8 promotes melanoma progression through coordinated Akt and twist signaling in the tumor microenvironment. Cancer Res. 2008;68:8889–98.
Neutzner M, Lopez T, Feng X, Bergmann-Leitner ES, Leitner WW, Udey MC. MFG-E8/lactadherin promotes tumor growth in an angiogenesis-dependent transgenic mouse model of multistage carcinogenesis. Cancer Res. 2007;67:6777–85.
Jinushi M, Sato M, Kanamoto A, Itoh A, Nagai S, Koyasu S, et al. Milk fat globule epidermal growth factor-8 blockade triggers tumor destruction through coordinated cell-autonomous and immune-mediated mechanisms. J Exp Med. 2009;206:1317–26.
Jinushi M, Nakazaki Y, Dougan M, Carrasco DR, Mihm M, Dranoff G. MFG-E8-mediated uptake of apoptotic cells by APCs links the pro- and antiinflammatory activities of GM-CSF. J Clin Invest. 2007;117:1902–13.
Moriyama I, Ishihara S, Rumi MA, Aziz MD, Mishima Y, Oshima N, et al. Decoy oligodeoxynucleotide targeting activator protein-1 (AP-1) attenuates intestinal inflammation in murine experimental colitis. Lab Invest. 2008;88:652–63.
Suzuki R, Kohno H, Sugie S, Nakagama H, Tanaka T. Strain differences in the susceptibility to azoxymethane and dextran sodium sulfate-induced colon carcinogenesis in mice. Carcinogenesis. 2006;27:162–9.
Tanaka T, Kohno H, Sakata K, Yamada Y, Hirose Y, Sugie S, et al. Modifying effects of dietary capsaicin and rotenone on 4-nitroquinoline 1-oxide-induced rat tongue carcinogenesis. Carcinogenesis. 2002;8:1361–7.
Neufert C, Becker C, Neurath MF. An inducible mouse model of colon carcinogenesis for the analysis of sporadic and inflammation-driven tumor progression. Nat Protoc. 2007;2:1998–2004.
Endo H, Hosono K, Uchiyama T, Sakai E, Sugiyama M, Takahashi H, et al. Leptin acts as a growth factor for colorectal tumours at stages subsequent to tumour initiation in murine colon carcinogenesis. Gut. 2011;60:1363–71.
Rosenberg DW, Giardina C, Tanaka T. Mouse models for the study of colon carcinogenesis. Carcinogenesis. 2009;30:183–96.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–35.
Ullman TA, Itzkowitz SH. Intestinal inflammation and cancer. Gastroenterology. 2011;140:1807–16.
Cooper HS, Everley L, Chang WC, Pfeiffer G, Lee B, Murthy S, et al. The role of mutant Apc in the development of dysplasia and cancer in the mouse model of dextran sulfate sodium-induced colitis. Gastroenterology. 2001;121:1407–16.
Greten FR, Eckmann L, Greten TF, Park JM, Li ZW, Egan LJ, et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. 2004;118:285–96.
Grivennikov S, Karin E, Terzic J, Mucida D, Yu GY, Vallabhapurapu S, et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell. 2009;15:103–13.
Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140:883–99.
Okuyama T, Kurata S, Tomimori Y, Fukunishi N, Sato S, Osada M, et al. p63(TP63) elicits strong trans-activation of the MFG-E8/lactadherin/BA46 gene through interactions between the TA and DeltaN isoforms. Oncogene. 2008;27:308–17.
Avraamides CJ, Garmy-Susini B, Varner JA. Integrins in angiogenesis and lymphangiogenesis. Nat Rev Cancer. 2008;8:604–17.
Conflict of interest
Kinoshita Y, Kusunoki R, Ishihara S, Oka A, Sonoyama H, Tada Y, Fukuba N, Oshima N, Moriyama I, Yuki T, and Kawashima K received research grants from Daiichi-Sankyo Pharm, Takeda Pharm, Astellas Pharm, AstraZeneca KK, Mitsubishi Tanabe Pharm and Eisai Pharm. Kinoshita Y received honoraria for lecture fees from Daiichi-Sankyo Pharm, Takeda Pharm, Astellas Pharm, AstraZeneca KK, and Eisai Pharm.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
535_2014_1036_MOESM2_ESM.tif
Supplementary material 2 Supplemental Figure 1. Colon epithelial cell proliferation was lower in MFG-E8 KO mice as compared to WT mice. a Representative images showing ki67 staining in histological sections during recovery phase of DSS colitis (3 weeks). b Average number of ki67-positive cells per colon crypt (3 weeks, blank bar WT, black bar KO) (WT, n = 4; KO, n = 4. *p < 0.05 vs. WT) (TIFF 2025 kb)
Rights and permissions
About this article
Cite this article
Kusunoki, R., Ishihara, S., Tada, Y. et al. Role of milk fat globule-epidermal growth factor 8 in colonic inflammation and carcinogenesis. J Gastroenterol 50, 862–875 (2015). https://doi.org/10.1007/s00535-014-1036-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-014-1036-x