Abstract
Background
Efficacy of available therapies for patients with HCV who have previously failed treatment is limited. Two Phase III, open-label trials in Japan investigated efficacy and safety of simeprevir and peginterferon-α-2a/ribavirin (PR) combination therapy in treatment-experienced patients with genotype 1 HCV infection.
Methods
In CONCERTO-2, prior non-responders to IFN-based therapy (N = 106) received simeprevir (TMC435) 100 mg QD with PR for 12 (SMV12, n = 53) or 24 weeks (SMV24, n = 53) followed by response-guided therapy (RGT) with PR for 12/36 (SMV12) or 0/24 (SMV24) weeks. In CONCERTO-3, relapsers after IFN-based therapy (N = 49) received simeprevir 100 mg QD with PR for 12 weeks followed by RGT with PR for 12/36 weeks. Primary endpoints were the rates of sustained virologic response 12 weeks after treatment end (SVR12).
Results
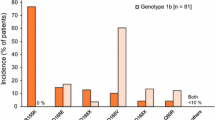
SVR12 rates were 52.8 % (SMV12) and 35.8 % (SMV24) for prior non-responders, and 95.9 % for prior relapsers (SMV12; p ≤ 0.0001 vs null hypothesis, respectively). Most prior non-responders (SMV12: 81.1 %; SMV24: 73.6 %) and prior relapsers (95.9 %) met RGT criteria and completed PR to Week 24. Of these, 60.5 %, 48.7 %, and 95.7 %, respectively, achieved SVR12. Viral breakthrough occurred in 13.2 % (SMV12) and 11.3 % (SMV24) of prior non-responders; no viral breakthrough occurred in prior relapsers. Viral relapse occurred in 38.6 % (SMV12) and 51.1 % (SMV24) of prior non-responders and 8.2 % of prior relapsers. Simeprevir with PR was generally well tolerated in both studies.
Conclusion
Re-treatment with 12 weeks of simeprevir QD with PR provided high SVR in treatment-experienced patients with chronic HCV genotype 1 infection, and allowed most patients to complete treatment in 24 weeks.


Similar content being viewed by others
References
Lavanchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect. 2011;17:107–15.
Chung H, Ueda T, Kudo M. Changing trends in hepatitis C infection over the past 50 years in Japan. Intervirology. 2010;53:39–43.
Umemura T, Ichijo T, Yoshizawa K, Tanaka E, Kiyosawa K. Epidemiology of hepatocellular carcinoma in Japan. J Gastroenterol. 2009;44(Suppl 19):102–7.
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol. 2011;55:245–64.
Izumi N. Diagnostic and treatment algorithm of the Japanese society of hepatology: a consensus-based practice guideline. Oncology. 2010;78(Suppl 1):78–86.
Fried MW, Shiffman ML, Reddy KR, Smith C, Marinos G, Gonçales FL Jr, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347:975–82.
Manns MP, McHutchison JG, Gordon SC, Rustgi VK, Shiffman M, Reindollar R, et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet. 2001;358:958–65.
Iino S, Okita K, Omata M, Kumada H, Hayashi N, Tanigawa H. Clinical efficacy of PEG-interferon alpha-2b and ribavirin combination therapy for 48 weeks in chronic hepatitis C patients with genotype 1 and high viral load: retrospective comparison with interferon alpha-2b and ribavirin combination therapy for 24 weeks. Kantansui. 2004;49:1099–121.
Kuboki M, Iino S, Okuno T, Omata M, Kiyosawa K, Kumada H, et al. Peginterferon alpha-2a (40 KD) plus ribavirin for the treatment of chronic hepatitis C in Japanese patients. J Gastroenterol Hepatol. 2007;22:645–52.
Jacobson IM, Gonzalez SA, Ahmed F, Lebovics E, Min AD, Bodenheimer HC Jr, et al. A randomized trial of pegylated interferon alpha-2b plus ribavirin in the retreatment of chronic hepatitis C. Am J Gastroenterol. 2005;100:2453–62.
Welsch C, Jesudian A, Zeuzem S, Jacobson I. New direct-acting antiviral agents for the treatment of hepatitis C virus infection and perspectives. Gut. 2012;61(Suppl 1):i36–46.
Chayama K, Hayes CN, Ohishi W, Kawakami Y. Treatment of chronic hepatitis C virus infection in Japan: update on therapy and guidelines. J Gastroenterol. 2013;48:1–12.
Hayashi N, Okanoue T, Tsubouchi H, Toyota J, Chayama K, Kumada H. Efficacy and safety of telaprevir, a new protease inhibitor, for difficult-to-treat patients with genotype 1 chronic hepatitis C. J Viral Hepat. 2012;19:e134–42.
Kumada H, Toyota J, Okanoue T, Chayama K, Tsubouchi H, Hayashi N. Telaprevir with peginterferon and ribavirin for treatment-naive patients chronically infected with HCV of genotype 1 in Japan. J Hepatol. 2012;56:78–84.
Sherman KE, Flamm SL, Afdhal NH, Nelson DR, Sulkowski MS, Everson GT, et al. Response-guided telaprevir combination treatment for hepatitis C virus infection. N Engl J Med. 2011;365:1014–24.
Jacobson IM, McHutchison JG, Dusheiko G, Di Bisceglie AM, Reddy KR, Bzowej NH, et al. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med. 2011;364:2405–16.
Bacon BR, Gordon SC, Lawitz E, Marcellin P, Vierling JM, Zeuzem S, et al. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1207–17.
Hezode C. Boceprevir and telaprevir for the treatment of chronic hepatitis C: safety management in clinical practice. Liver Int. 2012;32(Suppl 1):32–8.
Poordad F, McCone J Jr, Bacon BR, Bruno S, Manns MP, Sulkowski MS, et al. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1195–206.
Hayashi N, Seto C, Kato M, Komada Y, Goto S. Once-daily simeprevir (TMC435) with peginterferon/ribavirin for treatment-naïve hepatitis C genotype 1-infected patients in Japan: the DRAGON study. J Gastroenterol. 2014;49:138–47.
Kaiser S, Lutze B, Hass HG, Werner CR. High sustained virologic response rates in HCV genotype 1 relapser patients retreated with peginterferon alfa-2a (40KD) plus ribavirin for 72 weeks. Hepatology. 2008;48:1140A (abstract).
Jensen DM, Marcellin P, Freilich B, Andreone P, Di BA, Brandao-Mello CE, et al. Re-treatment of patients with chronic hepatitis C who do not respond to peginterferon-alpha2b: a randomized trial. Ann Intern Med. 2009;150:528–40.
Zeuzem S, Berg T, Gane E, Ferenci P, Foster GR, Fried MW, et al. Simeprevir increases rate of sustained virologic response among treatment-experienced patients with HCV genotype-1 infection: a phase IIb trial. Gastroenterology. 2014;146:430–41.e6.
Manns M, Reesink H, Berg T, Dusheiko G, Flisiak R, Marcellin P, et al. Rapid viral response of once-daily TMC435 plus pegylated interferon/ribavirin in hepatitis C genotype-1 patients: a randomized trial. Antivir Ther. 2011;16:1021–33.
Zeuzem S, Andreone P, Pol S, Lawitz E, Diago M, Roberts S, et al. Telaprevir for retreatment of HCV infection. N Engl J Med. 2011;364:2417–28.
Martinot-Peignoux M, Stern C, Maylin S, Ripault MP, Boyer N, Leclere L, et al. Twelve weeks posttreatment follow-up is as relevant as 24 weeks to determine the sustained virologic response in patients with hepatitis C virus receiving pegylated interferon and ribavirin. Hepatology. 2010;51:1122–6.
Fried MW, Buti M, Dore GJ, Flisiak R, Ferenci P, Jacobsen I, et al. Once-daily simeprevir (TMC435) with pegylated interferon and ribavirin in treatment-naïve genotype 1 hepatitis C: the randomized PILLAR study. Hepatology. 2013;58:1918–29.
Reddy KR, Lin F, Zoulim F. Response-guided and -unguided treatment of chronic hepatitis C. Liver Int. 2012;32(Suppl 1):64–73.
Pol S, Aerssens J, Zeuzem S, et al. Limited impact of IL28B genotype on response rates in telaprevir-treated patients with prior treatment failure. J Hepatol. 2013;58:883–9.
Lenz O, Verbinnen T, Lin TI, et al. In vitro resistance profile of the hepatitis C virus NS3/4A protease inhibitor TMC435. Antimicrob Agents Chemother. 2010;54:1878–87.
Halfon P, Locarnini S. Hepatitis C virus resistance to protease inhibitors. J Hepatol. 2011;55:192–206.
Huisman MT, Snoeys J, Monbaliu J, Martens M, Sekar V, Raoof A. In vitro studies investigating the mechanism of interaction between TMC435 and hepatic transporters. Poster 278 presented at the 61st Annual Meeting of the American Association for the Study of Liver Diseases (AASLD), Boston, USA, 29 Oct-2 Nov, 2010.
Acknowledgments
The authors would like to thank all patients, their families, principal investigators, and their staff at the following 28 study sites (in alphabetical order): Akio Ido (Kagoshima University Medical and Dental Hospital); Akito Sakai (Kanazawa University Hospital); Eiji Mita (National Hospital Organization Osaka National Hospital); Harumasa Yoshihara (Osaka Rosai Hospital); Hideki Hagiwara (Kansai Rosai Hospital); Hideyuki Nomura (Shin-Kokura Hospital); Hiromitsu Kumada (Toranomon Hospital); Hiroshi Yatsuhashi (National Hospital Organization Nagasaki Medical Center); Katsuaki Tanaka (Yokohama City University Medical Center); Kawakami Yoshiiku (Hiroshima University Hospital); Kiyomi Yasuda (Kiyokawa Hospital); Masashi Mizokami (Kohnodai Hospital, National Center for Global Health and Medicine); Masatoshi Kudo (Kinki University Hospital); Mina Nakagawa (Tokyo Medical And Dental University Hospital Faculty of Medicine); Kiyohide Kioka (Osaka City General Hospital); Namiki Izumi (Musashino Red Cross Hospital); Syuhei Nishiguchi (The Hospital of Hyogo College of Medicine); Takayoshi Ito (Showa University Hospital); Takeji Umemura (Shinshu University Hospital); Tatsuya Ide (Kurume University Hospital); Tetsuo Takehara (Osaka University Hospital); Toshifumi Ito (Osaka Koseinenkin Hospital); Toshihide Shima (Saiseikai Suita Hospital); Yoichi Hiasa (Ehime University Hospital); Yoshito Ito (University Hospital, Kyoto Prefectural University of Medicine); Yutaka Sasaki (Kumamoto University Hospital); Yoshiyasu Karino (Sapporo Kosei General Hospital). These studies were funded by Janssen Pharmaceutical K.K. Medical writing support was provided by Julie Adkins on behalf of Complete Medical Communications and was funded by Janssen Research & Development.
Conflict of interest
Namiki Izumi received honoraria for lectures from MSD Co., Chugai Co., Daiichi-Sankyo Co., and Bayer Co.. Norio Hayashi, Hiromitsu Kumada, Takeshi Okanoue, and Hiroshi Yatsuhashi have no conflict of interest. Hirohito Tsubouchi has an advisory relationship with Janssen Paharmacutical K.K. Mai Kato, Ki Ritou, Yuji Komada, Chiharu Seto, and Shoichiro Goto are employees of Janssen Pharmaceutical K.K.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Izumi, N., Hayashi, N., Kumada, H. et al. Once-daily simeprevir with peginterferon and ribavirin for treatment-experienced HCV genotype 1-infected patients in Japan: the CONCERTO-2 and CONCERTO-3 studies. J Gastroenterol 49, 941–953 (2014). https://doi.org/10.1007/s00535-014-0949-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-014-0949-8