Summary
Background
Pulmonary embolism (PE) is the third cardiovascular cause of hospital admission, following acute coronary syndrome and stroke. Despite high-tech diagnostic methods and new treatment modalities, PEs continue to have a high mortality rate within the first 3 months. This study was designed to assess the additional prognostic value of a complete blood cell count, renal function markers, C-reactive protein, and simplified pulmonary embolism severity index (sPESI) scoring system in PE 100-day mortality.
Materials and methods
The study retrospectively enrolled 208 consecutive patients who were hospitalized with the diagnosis of an acute PE. The patients’ demographic characteristics and clinical and laboratory parameters were recorded from the hospital electronic database and patient’s case notes. The primary end point of the study was an adverse 100-day outcome, defined as death from any cause.
Results
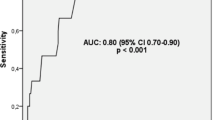
The all-cause mortality in the first 100 days was 14.42 %. The mean age was 57.87 ± 18.17 (range: 16–93) years. We included 79 (38 %) male and 129 (62 %) female individuals. Red cell distribution width (RDW) and sPESI were found to be statistically significant predictors of PE mortality by multivariate regression analysis. On multivariate regression analysis, RDW was associated with a 4.08-fold (95 % confidence interval: 1.229–13.335, P = 0.021) increase in PE mortality.
Conclusion
The results of this study demonstrated that RDW and sPESI may be a useful guide in predicting 100-day mortality. The elevated RDW may alert physicians to possible poor prognosis.
Zusammenfassung
Grundlagen
Die Pulmonalembolie (PE) ist nach dem akuten Koronarsyndrom und dem Schlaganfall der dritthäufigste kardiovaskuläre Grund für eine Aufnahme ins Spital. Trotz hochtechnisierter Diagnostik und neuen Behandlungsmethoden haben Patienten mit einer PE noch immer eine hohe Mortalität innerhalb der ersten 3 Monate nach dem Ereignis. Die vorliegende Studie hat es sich zum Ziel gemacht, den zusätzlichen prognostischen Wert eines kompletten Blutbilds (kBB), des CRPs, von Markern der Nierenfunktion und von einem vereinfachten PE-Schweregrad Index (sPESI) in Bezug auf die Mortalität innerhalb der ersten 100 Tagen nach einer PE zu erheben.
Material und Methoden
Die Studie erfasste retrospektiv 208 konsekutive Patienten (79 (38 %) Männer, 129 (62 %) Frauen), die wegen der Diagnose einer akuten PE hospitalisiert worden waren. Die demographischen Charakteristika und Laborparameter wurden aus den elektronisch gespeicherten Spitalsdaten und individuellen Krankengeschichten erhoben. Der primäre Endpunkt der Studie war Tod jeder Ursache innerhalb der ersten 100 Tage.
Ergebnisse
Die Gesamtmortalität betrug 14,42 % innerhalb der ersten 100 Tage. Das mittlere Alter der Patienten lag bei 57,87 ± 18,17 (range: 16–93) Jahren. Die multivariate Regressionsanalyse ergab, dass die Verteilung der Erythrozytenbreite (RDW) und der erhobenen sPESI statistisch signifikante Prädiktoren der Mortalität nach PE waren. In dieser Analyse war die RDW mit einem 4,08 fachen (95 % CI: 1,229–13,335, P = 0,021) Anstieg der Mortalität nach PE verbunden.
Schlussfolgerungen
Die Ergebnisse dieser Studie zeigen, dass die RDW und sPESI im Bezug auf die Beurteilung der Prognose nach PE nützlich sein können. Erhöhte RDW könnten den Arzt auf eine möglicherweise schlechtere Prognose hinweisen.


Similar content being viewed by others
References
Giuntini C, Di Ricco G, Marini C, Melillo E, Palla A. Pulmonary embolism: epidemiology. Chest. 1995;107:3–9.
Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet. 1999;353:1386–9.
Torbicki A, Perrier A, Konstantinides S, Agnelli G, Galie N, Pruszczyk P, et al. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). Eur Heart J. 2008;29:2276–315.
Kearon C, Kahn SR, Agnelli G, Goldhaber SZ, Raskob G, Comerota J. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians evidence-based clinical practice guidelines (8th edition). Chest. 2008;133:454–545.
Zee RY, Glynn RJ, Cheng S, Steiner L, Rose L, Ridker PM. An evaluation of candidate genes of inflammation and thrombosis in relation to the risk of venous thromboembolism: the women’s genome health study. Circ Cardiovasc Genet. 2009;2:57–62.
Fox EA, Kahn SR. The relationship between inflammation and venous thrombosis. A systematic review of clinical studies. Thromb Haemost. 2005;94:362–5.
Rhodes CJ, Wharton J, Howard LS, Gibbs JS, Wilkins MR. Red cell distribution width outperforms other potential circulating biomarkers in predicting survival in idiopathic pulmonary arterial hypertension. Heart. 2011;97:1054–60.
Margolis KL, Manson JE, Greenland P, Rodabough RJ, Bray PF, Safford M, et al. Leukocyte count as a predictor of cardiovascular events and mortality in postmenopausal women: the women’s health initiative observational study. Arch Intern Med. 2005;165:500–8.
Horne BD, Anderson JL, John JM, Weaver A, Bair TL, Jensen KR, et al; Intermountain Heart Collaborative Study Group. Which white blood cell subtypes predict increased cardiovascular risk? J Am Coll Cardiol. 2005;45:1638–43.
Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348:138–50.
Onsrud M, Thorsby E. Influence of in vivo hydrocortisone on some human blood lymphocyte subpopulations. Effect on natural killer cell activity. Scand J Immunol. 1981;13:573–9.
Cho KH, Jeong MH, Ahmed K, Hachinohe D, Choi HS, Chang SY, et al. Value of early risk stratification using hemoglobin level and neutrophil-to-lymphocyte ratio in patients with ST elevation myocardial infarction undergoing primary percutaneous coronary intervention. Am J Cardiol. 2011;107:849–56.
Uthamalingam S, Patvardhan EA, Subramanian S, Ahmed W, Martin W, Daley M, et al. Utility of the neutrophil to lymphocyte ratio in predicting long-term outcomes in acute decompensated heart failure. Am J Cardiol. 2011;107:433–8.
Aujesky D, Obrosky DS, Stone RA, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172:1041–6.
Jiménez D, Aujesky D, Moores L, Gómez V, Lobo JL, Uresandi F, et al; RIETE Investigators. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med. 2010;170:1383–9.
World Health Organization. Nutritional anemia: report of a WHO Scientific Group. Geneva: World Health Organization; 1968.
Lapez-Jimanez L, Montero M, Gonzailez-Fajardo JA, Arcelus JI, Suairez C, Lobo JL, et al; RIETE Investigators. Venous thromboembolism in very elderly patients: findings from a prospective registry (RIETE). Haematologica. 2006;91(8):1046–51.
Zahorec R. Ratio of neutrophil to lymphocyte counts—rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy. 2001;102:5–14.
Karnad A, Poskitt TR. The automated complete blood cell count. Use of the red blood cell volume distribution width and mean platelet volume in evaluating anemia and thrombocytopenia. Arch Intern Med. 1985;145:1270–2.
Thompson WG, Meola T, Lipkin M Jr, Freedman ML. Red cell distribution width, mean corpuscular volume, and transferrin saturation in the diagnosis of iron deficiency. Arch Intern Med. 1988;148:2128–30.
Felker GM, Allen LA, Pocock SJ, Shaw LK, McMurray JJ, Pfeffer MA, et al. Red cell distribution width as a novel prognostic marker in heart failure: data from the CHARM Program and the Duke Databank. J Am Coll Cardiol. 2007;50:40–7.
Förhécz Z, Gombos T, Borgulya G, Pozsonyi Z, Prohászka Z, Jánoskuti L. Red cell distribution width in heart failure: prediction of clinical events and relationship with markers of ineffective erythropoiesis, inflammation, renal function, and nutritional state. Am Heart J. 2009;158:659–66.
Ozsu S, Abul Y, Gunaydin S, Orem A, Ozlu T. Prognostic value of red cell distribution width in patients with pulmonary embolism. Clin Appl Thromb Hemost. 2012. Epub ahead of print.
Zorlu A, Bektasoglu G, Guven FM, Dogan OT, Gucuk E, Ege MR, Altay H, Cinar Z, Tandogan I, Yilmaz MB. Usefulness of admission red cell distribution width as a predictor of early mortality in patients with acute pulmonary embolism. Am J Cardiol. 2012;109:128–34.
Donze J, Labarere J, Mean M, Jimenez D, Aujesky D. Prognostic importance of anaemia in patients with acute pulmonary embolism. Thromb Haemost. 2011;106:289–95.
Jagroop IA, Mikhailidis DP. Mean platelet volume is a useful parameter: a reproducible routine method using a modified Coulter thrombocytometer. Platelets. 2001;12:171.
Samara WM, Gurbel PA. The role of platelet receptors and adhesion molecules in coronary artery disease. Coron Artery Dis. 2003;14:65–79.
Kostrubiec M, Labyk A, Pedowska-Wloszek J, Hrynkiewicz-Szymanska A, Pacho S, Jankowski K, et al. Mean platelet volume predicts early death in acute pulmonary embolism. Heart. 2010;96:460–5.
Braekkan SK, Mathiesen EB, Njølstad I, Wilsgaard T, Størmer J, Hansen JB. Mean platelet volume is a risk factor for venous thromboembolism: the Tromsø study. J Thromb Haemost. 2010;8:157–62.
Righini M, Roy PM, Meyer G, Verschuren F, Aujesky D, Le Gal G. The Simplified Pulmonary Embolism Severity Index (PESI): validation of a clinical prognostic model for pulmonary embolism. J Thromb Haemost. 2011;9:2115–7.
Laporte S, Mismetti P, De’cousus H, Uresandi F, Otero R, Lobo JL, et al; RIETE Investigators. Clinical predictors for fatal pulmonary embolism in 15,520 patients with venous thromboembolism: findings from the Registro Informatizado de la Enfermedad TromboEmbolica venosa (RIETE) registry. Circulation. 2008;117:1711–6.
Nijkeuter M, Sohne M, Tick LW, Kamphuisen PW, Kramer MH, Laterveer L, et al; Christopher Study Investigators. The natural course of hemodynamically stable pulmonary embolism: clinical outcome and risk factors in a large prospective cohort study. Chest. 2007;131:517–23.
Shantikumar S, Grant PJ, Catto AJ, Bamford JM, Carter AM. Elevated C-reactive protein and long-term mortality after ischaemic stroke: relationship with markers of endothelial cell and platelet activation. Stroke. 2009;40:977–9.
Boekholdt SM, Hack CE, Sandhu MS, Luben R, Bingham SA, Wareham NJ, et al. C-reactive protein levels and coronary artery disease incidence and mortality in apparently healthy men and women: the EPIC-Norfolk prospective population study 1993–2003. Atherosclerosis. 2006;187:415–22.
Marchena Yglesias PJ, Nieto Rodríguez JA, Serrano Martínez S, Belinchón Moya O, Cortés Carmona A, Díaz de Tuesta A, et al. Acute-phase reactants and markers of inflammation in venous thromboembolic disease: correlation with clinical and evolution parameters. An Med Interna. 2006;23:105–10.
Hogg K, Hinchliffe E, Haslam S, Sethi B, Carrier M, Lecky F. Predicting short term mortality after investigation for venous thromboembolism. Thromb Res. 2013;131:141–6.
Abul Y, Karakurt S, Ozben B, Toprak A, Celikel T. C-reactive protein in acute pulmonary embolism. J Investig Med. 2011;59:8–14.
Barritt DW, Jordan SC. Anticoagulant drugs in the treatment of pulmonary embolism: a controlled trial. Lancet. 1960;1:1309–12.
Konstantinides S. Thrombolysis in submassive pulmonary embolism? Yes. J Thromb Haemost. 2003;1:1127–9.
Dalen JE. Thrombolysis in submassive pulmonary embolism? No. J Thromb Haemost. 2003;1:1130–2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sen, H., Abakay, O., Tanrikulu, A. et al. Is a complete blood cell count useful in determining the prognosis of pulmonary embolism?. Wien Klin Wochenschr 126, 347–354 (2014). https://doi.org/10.1007/s00508-014-0537-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-014-0537-1