Abstract
Genetic variability and population structure of Sapindus trifoliatus L. (Sapindaceae), collected from Gujarat, Karnataka and Uttar Pradesh states were estimated using three DNA fingerprinting methods viz., random amplified polymorphic DNA (RAPD), directed amplification of minisatellite DNA (DAMD) and inter-simple sequence repeats (ISSR). The cumulative data analysis carried out for all three markers showed 69.42 % polymorphism. The intra-population genetic diversity analysis revealed the highest values of Nei’s genetic diversity (0.16), Shannon information index (0.24) and polymorphic loci (43.99 %) among Bhavnagar (BH) population, whereas lowest values were found in Junagarh (JU) population. The maximum inter-population average genetic distance (0.20) was between Allahabad (AL) and JU populations. Analysis of molecular variance (AMOVA) showed highest percentage of variation among individuals of populations (56 %) followed by 25 % among populations and 19 % among regions. Principal coordinate analysis and UPGMA dendrogram revealed that genetic diversity was in congruence with the geographical diversity. The data strongly suggest that low genetic flow, geographic isolation and to some extent genetic drift are the major factors responsible for high genetic differentiation. Preservation of genetic diversity of S. trifoliatus is important, both to promote adaptability of the populations to changing environment as well as to preserve a large gene pool for future genetic improvement. The present study using RAPD, DAMD and ISSR profiles of S. trifoliatus provide the means of rapid characterization of accessions within the populations, and thus enable the selection of appropriate accessions for further utilization in conservation and prospection programs of this important plant genetic resource.





Similar content being viewed by others
Abbreviations
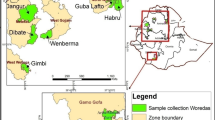
- AL:
-
Allahabad
- BH:
-
Bhavnagar
- HS:
-
Hassan
- JU:
-
Junagarh
- SH:
-
Shimoga
- SR:
-
Sirsi
- UP:
-
Uttar Pradesh
References
Anderson TH, Nilsson-Tillgren T (1997) A fungal minisatellite. Nature 386:771
Anonymous (1972) The wealth of India-raw materials, vol IX. CSIR, New Delhi
Arulmozhi DK, Sridhar N, Bodhankar SL, Veeranjaneyulu A, Arora SK (2004) In vitro pharmacological investigations of Sapindus trifoliatus in various migraine targets. J Ethnopharmacol 95:239–245
Asthana P, Jaiswal VS, Jaiswal U (2011) Micropropagation of Sapindus trifoliatus L. and assessment of genetic fidelity of micropropagated plants using RAPD analysis. Acta Physiol Plant 33:1821–1829
Avise JC (1994) Molecular markers, natural history and evolution. Chapman and Hall, New York
Basha SD, Sujatha M (2007) Inter and intra-population variability of Jatropha curcas (L.) characterized by RAPD and ISSR markers and development of population-specific SCAR markers. Euphytica 156:375–386
Basha SD, Francis G, Makkar HPS, Becker K, Sujatha M (2009) A comparative study of biochemical traits and molecular markers for assessment of genetic relationships between Jatropha curcas L. germplasm from different countries. Plant Sci 176:812–823
Botstein D, White RL, Skolnick M, Davis RW (1980) Construction of a genetic linkage map in man using restriction fragment length polymorphism. Am J Hum Genet 32:314–331
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Heath DD, Iwama GK, Devlin RH (1993) PCR primed with the VNTR core sequences yields species specific patterns and hypervariable probes. Nucleic Acids Res 21:5782–5785
Hogbin PM, Peakall R (1999) Evaluation of the contribution of genetic research to the management of the endangered plant Zieria prostrate. Conserv Biol 13:514–522
Jeffreys AJ, Wilson V, Thein SL (1985) Individual-specific fingerprints of human DNA. Nature 316:76–79
Kirtikar KR, Basu BD (2001) Indian medicinal plants, vol III, 2nd edn. Oriental Enterprises, Dehradun
Lewontin RC (1972) The apportionment of human diversity. Evol Biol 6:381–398
Lorenz M, Partensky F, Borner T, Hess WR (1995) Sequencing of RAPD fragments amplified from the genome of the prokaryote Prochlorococcus marinus (prochlorophyta). Biochem Mol Biol Int 36:705–713
Mahar KS, Rana TS, Ranade SA (2011a) Molecular analyses of genetic variability in soap nut (Sapindus mukorossi Gaertn.). Ind Crop Prod 34:1111–1118
Mahar KS, Rana TS, Ranade SA, Meena B (2011b) Genetic variability and population structure in Sapindus emarginatus Vahl from India. Gene 485:32–39
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Narzary D, Mahar KS, Rana TS, Ranade SA (2009) Analysis of genetic diversity among wild pomegranates in Western Himalayas, using PCR methods. Sci Hortic 121:237–242
Narzary D, Rana TS, Ranade SA (2010) Genetic diversity in inter-simple sequence repeat profiles across natural populations of Indian pomegranate Punica granatum L. Plant Biol 12:806–813
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci USA 70:3321–3323
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269–5273
Page RDM (2001) TreeView (Win32) ver. 1.6.5. Available from http://taxonomy.zoology.gla.ac.uk/rod/treeview.html. Accessed 15 Dec 2011
Pant PC (2000) Sapindaceae. In: Singh NP, Bohra JN, Hajra PK, Singh DK (eds) Flora of India, vol V. Botanical Survey of India, Calcutta
Pavlicek A, Hrda S, Flegr J (1999) FreeTree—freeware program for construction of phylogenetic trees on the basis of distance data and bootstrapping/jackknife analysis of the tree robustness. Application in the RAPD analysis of the genus Frenkelia. Folia Biol (Praha) 45:97–99 Available from http://www.natur.cuni.cz/~flegr/programs/freetree.htm. Accessed 15 Dec 2011
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295 Available from http://www.anu.edu.au/BoZo/GenAlEx/. Accessed 15 Dec 2011
Powell W, Morgante M, Andre C, Hanafey M, Vogel J, Tingey S, Rafalski A (1996) The comparison of RFLP, RAPD, AFLP and SSR (microsatellite) markers for germplasm analysis. Mol Breed 2:225–238
Provost A, Wilkinson MJ (1999) A new system of comparing PCR primers applied to ISSR fingerprinting of potato cultivars. Theor Appl Genet 98:107–112
Rana TS, Verma S, Srivastava A, Srivastava J, Narzary D, Ranade SA (2007) Molecular distinction amongst some cultivated and wild species of Bauhinia L. Leguminosae: Caesalpinioideae in India using PCR based methods. Plant Cell Biotech Mol Biol 8:179–186
Ranade SA, Srivastava AP, Rana TS, Srivastava J, Tuli R (2008) Easy assessment of diversity in Jatropha curcas L. plants using two single primer amplification reaction (SPAR) methods. Biomass Bioenerg 32:533–540
Ranade SA, Rana TS, Narzary D (2009) SPAR profile and genetic diversity amongst pomegranate (Punica granatum L.) genotypes. Physiol Mol Biol Plants 15:61–70
Rohlf FJ (1998) NTSYS-pc: numerical taxonomy and multivariate analysis system. Ver. 2.02e. Exeter Software, Setauket, New York
Sharma SK, Rawat D, Kumar S, Kumar A, Kumaria S, Rao SR (2010) Single primer amplification reaction (SPAR) reveals intra-specific natural variation in Prosopis cineraria (L.) Druce. Trees 24:855–864
Sharma SK, Kumar S, Rawat D, Kumaria S, Kumar A, Rao SR (2011) Genetic diversity and gene flow estimation in Prosopis cineraria (L.) Druce: a key stone tree species of Indian Thar Desert. Biochem Syst Ecol 39:9–13
Slatkin M (1993) Isolation by distance in equilibrium and non-equilibrium populations. Evolution 47:264–279
Srivastava AP, Chandra R, Ranade SA (2005) Applicability of PCR based molecular markers for parentage analysis of three commercial mango hybrids. Ind J Plant Breed Genet 64:275–280
Srivastava AP, Chandra R, Saxena S, Rajan S, Ranade SA, Prasad V (2007) A PCR-based assessment of genetic diversity, and parentage analysis among commercial mango cultivars and hybrids. J Hortic Sci Biotech 82:951–959
Tourmente S, Deragon JM, Lafleurial J, Tutois S, Pelissier T, Cuvillier C, Espagnol MC, Picard G (1994) Characterization of minisatellites in Arabidopsis thaliana with sequence similarity to the human minisatellite core sequence. Nucleic Acids Res 22(160):3317–3321
Welsh J, McClelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acid Res 18:7213–7218
Williams JGK, Kubelik AR, Livak KJ, Rafaleski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acid Res 18:6531–6535
Winberg BC, Shori Z, Dallas JF, McIntyre CL, Gustafson JP (1993) Characterization of minisatellite sequences from Oryza sativa. Genome 36:978–983
Yeh FC, Yang RC, Boyle T (1997) POPGENE—ver. 1.32 (32 bit): a microsoft windows-based freeware for population genetic analysis. Available from http://www.ualberta.ca/~fcyeh/. Accessed 15 Dec 2011
Zheng DJ, Xie LS, Zhu JH, Zhang ZL (2012) Low genetic diversity and local adaptive divergence of Dracaena cambodiana (Liliaceae) populations associated with historical population bottlenecks and natural selection: an endangered long-lived tree endemic to Hainan Island, China. Plant Biol doi:10.1111/j.1438-8677.2011.00559.x
Zhou Z, Bebeli PJ, Somers DJ, Gustafson JP (1997) Direct amplification of minisatellite-region DNA with VNTR core sequences in the genus Oryza. Theor Appl Genet 95:942–949
Zong M, Liu H-L, Qiu Y-X, Yang S-Z, Zhao M-S, Fu C-X (2008) Genetic diversity in geographic differentiation in the threatened species Dysosma pleiantha in China as revealed by ISSR analysis. Biochem Genet 46:180–196
Acknowledgments
We are thankful to the Director, CSIR-National Botanical Research Institute, Lucknow, India for facilities and encouragement. We are also thankful to Dr. R. R. Rao (Emeritus Scientist) for his help and unrelenting support during the collection of plant material from various locales of Western Ghats. This study was financially supported by the Department of Science and Technology, Government of India, New Delhi (DST No: SP/SOPS-81/05).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by F. Canovas.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahar, K.S., Rana, T.S., Ranade, S.A. et al. Estimation of genetic variability and population structure in Sapindus trifoliatus L., using DNA fingerprinting methods. Trees 27, 85–96 (2013). https://doi.org/10.1007/s00468-012-0770-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-012-0770-z