Abstract
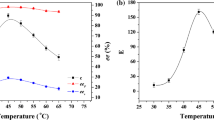
This study reports the lipase-catalyzed resolution of heteroaromatic secondary alcohols by succinic anhydride under different activation conditions by convenient procedure with succinic anhydride. The effects of succinic anhydride and the nature of the heteroatom are discussed in standard conditions in the kinetic resolution with lipases. The results recorded under microwave activation and ultrasonication is compared. (R)-4-chromanol was obtained in optically pure form (ee > 99 %) with a high selectivity E > 200 by Pseudomonas cepacia lipase (PCL) in diethyl ether, using microwave radiation and under ultrasonication. The reaction time is reduced compared to the conventional heating with a better control of the selectivity of the lipase PCL. A significant effect of the nature of the heteroatoms on the reactivity and selectivity of the lipase with succinic anhydride has been disclosed, regardless the conditions of activation. This method proved to be clean, fast, interesting alternative, and facilitates the use of a cyclic anhydride, by microwave or ultrasound especially with secondary alcohols. The process is a valuable prerequisite for the preparative scale production of enantiomerically heteroaromatic alcohols in sustainable chemistry.
Graphical abstract




Similar content being viewed by others
References
Faber K (1997) Biotransformations in organic chemistry, 6th edn. Springer, New York
Kazlauskas RJ, Bornscheuer UT (1998) Biotransformations with lipases. Biotechnology Set, 2nd edn. Wiley, Weinheim, pp 37–191
Lin H, Liu JY, Wang HB, Ahmed AAQ, Wu ZL (2011) Biocatalysis as an alternative for the production of chiral epoxides: a comparative review. J Mol Catal B Enzym 72:77–89
Meyer HP, Eichhorn E, Hanlon S, Lütz S, Schürmann M, Wohlgemuth R, Coppolecchia R (2013) The use of enzymes in organic synthesis and the life sciences: perspectives from the Swiss Industrial Biocatalysis Consortium (SIBC). Catal Sci Technol 3:29–40
Wong CH, Whitesides GM (1994) Enzymes in synthetic organic chemistry. Tetrahedron organic chemistry series, vol 12. Pergamon Press, London, p 195
Chen CS, Sih CJ (1989) General aspects and optimization of enantioselective biocatalysis in organic solvents: the use of lipases [new synthetic methods (76)]. Angew Chem Int Edit 28:695–707
Zheng GW, Xu JH (2011) New opportunities for biocatalysis: driving the synthesis of chiral chemicals. Curr Opin Biotech 22:784–792
Anastas PT, Warner JC (1998) Green chemistry: theory and practice. Oxford University Press, New York, p 135
Huisman GW, Collier SJ (2013) On the development of new biocatalytic processes for practical pharmaceutical synthesis. Curr Opin Chem Biol 17:284–292
McGrath BM, Walsh G (2006) Directory of therapeutic enzymes. CRC Press, London
Stepanenko V, De Jesús M, Correa W, Bermúdez L, Vázquez C, Guzmán I, Ortiz-Marciales M (2009) Chiral spiroaminoborate ester as a highly enantioselective and efficient catalyst for the borane reduction of furyl, thiophene, chroman, and thiochroman-containing ketones. Tetrahedron Asymmetry 20:2659–2665
Abdel-Megeed MF, Badr BE, Azaam MM, El-Hiti GA (2012) Synthesis, antimicrobial and anticancer activities of a novel series of diphenyl 1-(pyridin-3-yl) ethylphosphonates. Bioorgan Med Chem 20:2252–2258
Davis BG, Boyer V (2001) Biocatalysis and enzymes in organic synthesis. Nat Prod Rep 18:618–640
Ghanem A, Aboul-Enein HY (2004) Lipase-mediated chiral resolution of racemates in organic solvents. Tetrahedron Asymmetry 15:3331–3351
Jones TK, Mohan JJ, Xavier LC, Blacklock TJ, Mathre DJ, Sohar P, Grabowski EJJ (1991) An asymmetric synthesis of MK-0417. Observations on oxazaborolidine-catalyzed reductions. J Org Chem 56:763–769
Loupy A (2004) Synthèse organique sous micro-ondes en absence de solvant: un procédé efficace pour la chimie verte. Actual Chimique 272:18–26
Dintzner MR, Lyons TW, Akroush MH, Wucka P, Rzepka AT (2006) Microwave-assisted synthesis of a natural insecticide on basic montmorillonite K10 Clay. Green chemistry in the undergraduate organic laboratory. J Chem Educ 83:270
Wu Z, Borretto E, Medlock J, Bonrath W, Cravotto G (2014) Effects of ultrasound and microwaves on selective reduction: catalyst preparation and reactions. Chem Cat Chem 6:2762–2783
Lin G, Lin WY (1998) Microwave-promoted lipase-catalyzed reactions. Tetrahedron Lett 39:4333–4336
Yadav GD, Pawar SV (2012) Synergism between microwave irradiation and enzyme catalysis in transesterification of ethyl-3-phenylpropanoate with n-butanol. Bioresour Technol 109:1–6
Nogueira BM, Carretoni C, Cruz R, Freitas S, Melo PA Jr, Costa-Félix R, Nele M (2010) Microwave activation of enzymatic catalysts for biodiesel production. J Mol Catal B Enzym 67:117–121
Caddick S, Fitzmaurice R (2009) Microwave enhanced synthesis. Tetrahedron 65:3325–3355
Lin G, Liu HC (1995) Ultrasound-promoted lipase-catalyzed reactions. Tetrahedron Lett 36:6067–6068
Xiao Y, Wu Q, Cai Y, Lin X (2005) Ultrasound-accelerated enzymatic synthesis of sugar esters in nonaqueous solvents. Carbohyd Res 340:2097–2103
Bachu P, Gibson JS, Sperry J, Brimble MA (2007) The influence of microwave irradiation on lipase-catalyzed kinetic resolution of racemic secondary alcohols. Tetrahedron Asymmetry 18:1618–1624
Yu D, Ma D, Wang Z, Wang Y, Pan Y, Fang X (2012) Microwave-assisted enzymatic resolution of (R, S)-2-octanol in ionic liquid. Process Biochem 47:479–484
Brenelli ECS, Fernandes JLN (2003) Stereoselective acylations of 1, 2-azidoalcohols with vinyl acetate, catalyzed by lipase Amano PS. Tetrahedron Asymmetry 14:1255–1259
Dhake KP, Deshmukh KM, Wagh YS, Singhal RS, Bhanage BM (2012) Investigation of steapsin lipase for kinetic resolution of secondary alcohols and synthesis of valuable acetates in non-aqueous reaction medium. J Mol Catal B Enzym 77:15–23
Ribeiro SS, Raminelli C, Porto AL (2013) Enzymatic resolution by CALB of organofluorine compounds under conventional condition and microwave irradiation. J Fluorine Chem 154:53–59
Lerin LA, Loss RA, Remonatto D, Zenevicz MC, Balen M, Netto VO, de Oliveira DA (2014) A review on lipase-catalyzed reactions in ultrasound-assisted systems. Bioproc Biosyst Eng 37:2381–2394
Wang YF, Lalonde JJ, Momongan M, Bergbreiter DE, Wong CH (1988) Lipase-catalyzed irreversible transesterifications using enol esters as acylating reagents: preparative enantio-and regioselective syntheses of alcohols, glycerol derivatives, sugars and organometallics. J Am Chem Soc 110:7200–7205
Degueil-Castaing M, De Jeso B, Drouillard S, Maillard B (1987) Enzymatic reactions in organic synthesis: 2-ester interchange of vinyl esters. Tetrahedron Lett 28:953–954
Hanefeld U (2003) Reagents for irreversible enzymatic acylations. Org Biomol Chem 1:2405–2415
Terao Y, Tsuji K, Murata M, Achiwa K, Nishio T, Watanabe N, Seto K (1989) Facile process for enzymatic resolution of racemic alcohols. Chem Pharm Bull 37:1653–1655
Fiaud JC, Gil R, Legros JY, Aribi-Zouioueche L, König WA (1992) Kinetic resolution of 3-tbutyl and 3-phenyl cyclobutylidenethanols through lipase-catalyzed acylation with succinic anhydride. Tetrahedron Lett 33:6967–6970
Gutman AL, Brenner D, Boltanski A (1993) Convenient practical resolution of racemic alkyl-aryl alcohols via enzymatic acylation with succinic anhydride in organic solvents. Tetrahedron Asymmetry 4:839–844
Gonzalo G, Brieva R, Sánchez VM, Bayod M, Gotor V (2003) Anhydrides as acylating agents in the enzymatic resolution of an intermediate of (−)-paroxetine. J Org Chem 68:3333–3336
Sorgedrager MJ, Malpique R, van Rantwijk F, Sheldon RA (2004) Lipase catalyzed resolution of nitro aldol adducts. Tetrahedron Asymmetry 15:1295–1299
Wang Y, Wang R, Li Q, Zhang Z, Feng Y (2009) Kinetic resolution of rac-alkyl alcohols via lipase-catalyzed enantioselective acylation using succinic anhydride as acylating agent. J Mol Catal B Enzym 56:142–145
Teixeira R, Lourenço NM (2014) Enzymatic kinetic resolution of sec-alcohols using an ionic liquid anhydride as acylating agent. Tetrahedron Asymmetry 25:944–948
Bouzemi N, Debbeche H, Aribi-Zouioueche L, Fiaud JC (2004) On the use of succinic anhydride as acylating agent for practical resolution of aryl–alkyl alcohols through lipase-catalyzed acylation. Tetrahedron Lett 45:627–630
Debbeche H, Toffano M, Fiaud JC, Aribi-Zouioueche L (2010) Multi-substrate screening for lipase-catalyzed resolution of arylalkylethanols with succinic anhydride as acylating agent. J Mol Catal B Enzym 66:319–324
Kucher OV, Kolodyazhnaya AO, Smolii OB, Boiko AI, Kubyshkin VS, Mykhailiuk PK, Tolmachev AA (2014) Enzymatic resolution of chroman-4-ol and its core analogues with Burkholderia cepacia lipase. Tetrahedron Asymmetry 25:563–567
Lozano P, Bernal JM, Navarro A (2012) A clean enzymatic process for producing flavour esters by direct esterification in switchable ionic liquid/solid phases. Green Chem 14:3026–3033
Martins AB, Graebin NG, Lorenzoni AS, Fernandez-Lafuente R, Ayub MA, Rodrigues RC (2011) Rapid and high yields of synthesis of butyl acetate catalyzed by Novozyme 435: reaction optimization by response surface methodology. Process Biochem 46:2311–2316
Chen CS, Fujimoto Y, Girdaukas G, Sih CJ (1982) Quantitative analyses of biochemical kinetic resolutions of enantiomers. J Am Chem Soc 104:7294–7299
Kagan HB, Fiaud JC (1988) In kinetic resolution topics in stereochemistry, vol 18. Wiley, New York, pp 249–330
Torres S, Castro GR (2004) Non-aqueous biocatalysis in homogeneous solvent systems. Food Technol Biotechnol 42:271–277
Doukyu N, Ogino H (2010) Organic solvent-tolerant enzymes. Biochem Eng J 48:270–282
Rufino AR, Biaggio FC, Santos JC, De Castro HF (2010) Screening of lipases for the synthesis of xylitol monoesters by chemoenzymatic esterification and the potential of microwave and ultrasound irradiations to enhance the reaction rate. Int J Biol Macromol 47:5–9
Yadav GD, Sajgure AD, Dhoot SB (2008) Insight into microwave irradiation and enzyme catalysis in enantioselective resolution of RS-(±)-methyl mandelate. J Chem Technol Biot 83:1145–1153
Yu D, Wang Z, Chen P, Jin L, Cheng Y, Zhou J, Cao S (2007) Microwave-assisted resolution of (R, S)-2-octanol by enzymatic transesterification. J Mol Catal B Enzym 48:51–57
Itoh T, Han S, Matsushita Y, Hayase S (2004) Enhanced enantioselectivity and remarkable acceleration on the lipase-catalyzed transesterification using novel ionic liquids. Green Chem 6:437–439
Fransson ABL, Borén L, Pàmies O, Bäckvall JE (2005) Kinetic resolution and chemoenzymatic dynamic kinetic resolution of functionalized γ-hydroxy amides. J Org Chem 70:2582–2587
Kwiatkowska B, Bennett J, Akunna J, Walker GM, Bremner DH (2011) Stimulation of bioprocesses by ultrasound. Biotechnol Adv 29:768–780
Chen HC, Chen JH, Chang C, Shieh CJ (2011) Optimization of ultrasound-accelerated synthesis of enzymatic caffeic acid phenethyl ester by response surface methodology. Ultrason Sonochem 18:455–459
Hagenson LC, Doraiswamy LK (1998) Comparison of the effects of ultrasound and mechanical agitation on a reacting solid-liquid system. Chem Eng Sci 53:131–148
Solanki K, Gupta MN (2008) Optimising biocatalyst design for obtaining high transesterification activity by α-chymotrypsin in non-aqueous media. Chem Cent J 2:2
Zhao D, Yue H, Chen G, Jiang L, Zhang H, Wang Z, Liu G (2014) Enzymatic resolution of ibuprofen in an organic solvent under ultrasound irradiation. Biotechnol Appl Biochem 61(6):655–659
An BY, Xie XN, Xun EN, Wang R, Li CY, Sun RX, Wang L, Wang Z (2011) Ultrasound-promoted lipase-catalyzed enantioselective transesterification of (R, S)-glycidol. Chem Res Chin Univ 27:845–849
Acknowledgments
Algerian Ministry of Higher Education and Scientific Research (FNR 2000 and PNR), Fund for Scientific Research—FNRS are gratefully acknowledged for financial support of this work. Catholic University of Louvain is acknowledged for a grant “cooperation development” to Nedjma MELAIS.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Melais, N., Boukachabia, M., Aribi-Zouioueche, L. et al. Easy preparation of enantiomerically enriched heteroaromatic alcohols through lipase-catalyzed acylation with succinic anhydride under unconventional activation. Bioprocess Biosyst Eng 38, 1579–1588 (2015). https://doi.org/10.1007/s00449-015-1400-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-015-1400-0