Abstract
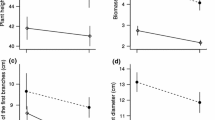
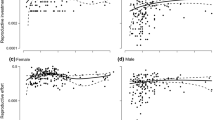
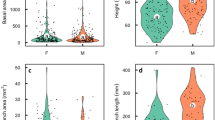
In flowering plants, many dioecious species display a certain degree of sexual dimorphism in non-reproductive traits, but this dimorphism tends to be much less striking than that found in animals. Sexual size dimorphism in plants may be limited because competition for light in crowded environments so strongly penalises small plants. The idea that competition for light constrains the evolution of strong sexual size dimorphism in plants (the size-constraint hypothesis) implies a strong dependency of the expression of sexual size dimorphism on the neighbouring density as a result of the capacity of plants to adjust their reproductive effort and investment in growth in response to their local environment. Here, we tested this hypothesis by experimentally altering the context of competition for light among male–female pairs of the light-demanding dioecious annual plant Mercurialis annua. We found that males were smaller than females across all treatments, but sexual size dimorphism was diminished for pairs grown at higher densities. This result is consistent with the size-constraint hypothesis. We discuss our results in terms of the tension between selection on size acting in opposite directions on males and females, which have different optima under sexual selection, and stabilizing selection for similar sizes in males and females, which have similar optima under viability selection for plasticity in size expression under different density conditions.



Similar content being viewed by others
References
Ågren J, Danell K, Elmqvist T, Ericson L, Hjalten J (1999) Sexual dimorphism and biotic interactions. In: Geber MA, Dawson TE, Delph LF (eds) Gender and sexual dimorphism in flowering plants. Springer, Berlin
Andersson M (1994) Sexual selection. Princeton University Press, Princeton, NJ
Ashman TL, Cole DH, Bradburn M (2004) Sex-differential resistance and tolerance to herbivory in a gynodioecious wild strawberry. Ecology 85:2550–2559. doi:10.1890/03-0495
Ballare CL (1999) Keeping up with the neighbours: phytochrome sensing and other signalling mechanisms. Trends Plant Sci 4:201. doi:10.1016/s1360-1385(99)01408-9
Ballare CL, Scopel AL, Sanchez RA (1990) Far-red radiation reflected from adjacent leaves: an early signal of competition in plant canopies. Science 247:329–332. doi:10.1126/science.247.4940.329
Barrett SCH, Hough J (2013) Sexual dimorphism in flowering plants. J Exp Bot 64:67–82. doi:10.1093/jxb/ers308
Bertiller MB, Sain CL, Bisigato AJ, Coronato FR, Aries JO, Graff P (2002) Spatial sex segregation in the dioecious grass Poa ligularis in northern Patagonia: the role of environmental patchiness. Biodivers Conserv 11:69–84. doi:10.1023/a:1014084024145
Bierzychudek P, Eckhart V (1988) Spatial segregation of the sexes of dioecious plants. Am Nat 132:34–43. doi:10.1086/284836
Bond WJ, Maze KE (1999) Survival costs and reproductive benefits of floral display in a sexually dimorphic dioecious shrub, Leucadendron xanthoconus. Evol Ecol 13:1–18. doi:10.1023/A:1006581412580
Bond WJ, Midgley JJ (1988) Allometry and sexual differences in leaf size. Am Nat 131:901–910. doi:10.1086/284830
Brunet J, Charlesworth D (1995) Floral sex allocation in sequentially blooming plants. Evolution 49:70–79. doi:10.2307/2410293
Conn JS, Blum U (1981) Differentiation between the sexes of Rumex hastatulus in net energy allocation, flowering and height. Bull Torrey Bot Club 108:446–455. doi:10.2307/2484445
Cornwallis CK, Uller T (2010) Towards an evolutionary ecology of sexual traits. Trends Ecol Evol 25:145–152. doi:10.1016/j.tree.2009.09.008
Darwin C (1871) The descent of man, and selection in relation to sex. Murray, London
Dawson TE, Geber MA (1999) Dimorphism in physiology and morphology. In: Geber MA, Dawson TE, Delph LF (eds) Gender and sexual dimorphism in flowering plants. Springer, Berlin
Delph LF (1999) Sexual dimorphism in live history. In: Geber MA, Dawson TE, Delph LF (eds) Gender and sexual dimorphism in flowering plants. Springer, Berlin
Delph LF, Bell DL (2008) A test of the differential-plasticity hypothesis for variation in the degree of sexual dimorphism in Silene latifolia. Evol Ecol Res 10:61–75
Delph LF, Galloway LF, Stanton ML (1996) Sexual dimorphism in flower size. Am Nat 148:299–320. doi:10.1086/285926
Delph LF, Knapczyk FN, Taylor DR (2002) Among-population variation and correlations in sexually dimorphic traits of Silene latifolia. J Evol Biol 15:1011–1020. doi:10.1046/j.1420-9101.2002.00467.x
Delph LF, Frey FM, Steven JC, Gehring JL (2004) Investigating the independent evolution of the size of floral organs via G-matrix estimation and artificial selection. Evol Dev 6:438–448. doi:10.1111/j.1525-142X.2004.04052.x
Dudley LS (2006) Ecological correlates of secondary sexual dimorphism in Salix glauca (Salicaceae). Am J Bot 93:1775–1783. doi:10.3732/ajb.93.12.1775
Durand B (1963) Le complexe Mercurialis annua L. s.i.: une étude biosystématique. Ann Sci Nat Bot Paris 12:579–736
Eppley SM (2006) Females make tough neighbors: sex-specific competitive effects in seedlings of a dioecious grass. Oecologia 146:549–554. doi:10.1007/s00442-005-0026-3
Eppley SM, Pannell JR (2007) Density-dependent self-fertilization and male versus hermaphrodite siring success in an androdioecious plant. Evolution 61:2349–2359. doi:10.1111/j.1558-5646.2007.00195.x
Eppley SM, Stanton ML, Grosberg RK (1998) Intrapopulation sex ratio variation in the salt grass Distichlis spicata. Am Nat 152:659–670. doi:10.1086/286197
Fairbairn DJ (1997) Allometry for sexual size dimorphism: pattern and process in the coevolution of body size in males and females. Annu Rev Ecol Syst 28:659–687. doi:10.1146/annurev.ecolsys.28.1.659
Field DL, Pickup M, Barrett SCH (2013) Comparative analyses of sex ratio variation in dioecious flowering plants. Evolution 67:661–672. doi:10.1111/evo.12001
Forrest JRK (2014) Plant size, sexual selection, and the evolution of protandry in dioecious plants. Am Nat 184:338–351. doi:10.1086/677295
Gleiser G, Verdu M, Segarra-Moragues JG, Gonzalez-Martinez SC, Pannell JR (2008) Disassortative mating, sexual specialization, and the evolution of gender dimorphism in heterodichogamous Acer opalus. Evolution 62:1676–1688. doi:10.1111/j.1558-5646.2008.00394.x
Gross KL, Soule JD (1981) Differences in biomass allocation to reproductive and vegetative structures of male and female plants of a dioecious, perennial herb, Silene alba (Miller) Krause. Am J Bot 68:801–807. doi:10.2307/2443186
Harper JL (1977) Population biology of plants. Academic Press, London
Harris MS, Pannell JR (2008) Roots, shoots and reproduction: sexual dimorphism in size and costs of reproductive allocation in an annual herb. Proc R Soc B Biol Sci 275:2595–2602. doi:10.1098/rspb.2008.0585
Harris MS, Pannell JR (2010) Canopy seed storage is associated with sexual dimorphism in the woody dioecious genus Leucadendron. J Ecol 98:509–515. doi:10.1111/j.1365-2745.2009.01623.x
Hesse E, Pannell JR (2011) Sexual dimorphism in a dioecious population of the wind-pollinated herb Mercurialis annua: the interactive effects of resource availability and competition. Ann Bot 107:1039–1045. doi:10.1093/aob/mcr046
Iglesias MC, Bell G (1989) The small scale spatial distribution of male and female plants. Oecologia 80:229–235
Jing SW, Coley PD (1990) Dioecy and herbivory: the effect of growth rate on plant defense in Acer negundo. Oikos 58:369–377. doi:10.2307/3545228
Klinkhamer PGL, deJong TJ, Metz H (1997) Sex and size in cosexual plants. Trends Ecol Evol 12:260–265. doi:10.1016/s0169-5347(97)01078-1
Korpelainen H (1991) Sex ratio variation and spatial segregation of the sexes in populatios of Rumex acetosa and R. acetosella (Polygonaceae). Plant Syst Evol 174:183–195. doi:10.1007/bf00940339
Lande R (1980) Sexual dimorphism, sexual selection, and adaptation in polygenic characters. Evolution 34:292–305. doi:10.2307/2407393
Lankinen A, Madjidian JA (2011) Enhancing pollen competition by delaying stigma receptivity: pollen deposition schedules affect siring ability, paternal diversity, and seed production in Collinsia heterophylla (Plantaginaceae). Am J Bot 98:1191–1200. doi:10.3732/ajb.1000510
Lloyd DG, Webb CJ (1977) Secondary sex characters in plants. Bot Rev 43:177–216. doi:10.1007/bf02860717
Lokker C, Susko D, Lovettdoust L, Lovettdoust J (1994) Population genetic structure of Vallisneria americana, a dioecious clonal macrophyte. Am J Bot 81:1004–1012. doi:10.2307/2445294
Lonsdale WM (1990) The self-thinning rule: dead or alive. Ecology 71:1373–1388. doi:10.2307/1938275
Lovett- Doust JL, Obrien G, Doust LL (1987) Effect of density on secondary sex characteristics and sex ratio in Silene alba (Caryophyllaceae). Am J Bot 74:40–46. doi:10.2307/2444329
Lovich JE, Gibbons JW (1992) A review of techniques for quantifying sexual size dimorphism. Growth Dev Aging 56:269–281
Meagher TR (1992) The quantitative genetics of sexual dimorphism in Silene latifolia (Caryophyllaceae). 1. Genetic variation. Evolution 46:445–457. doi:10.2307/2409863
Moore JC, Pannell JR (2011) Sexual selection in plants. Curr Biol 21:R176–R182. doi:10.1016/j.cub.2010.12.035
Obeso JR (2002) The costs of reproduction in plants. New Phytol 155:321–348. doi:10.1046/j.1469-8137.2002.00477.x
Pannell J (1997) Variation in sex ratios and sex allocation in androdioecious Mercurialis annua. J Ecol 85:57–69. doi:10.2307/2960627
Pannell JR, Ojeda F (2000) Patterns of flowering and sex-ratio variation in the Mediterranean shrub Phillyrea angustifolia (Oleaceae): implications for the maintenance of males with hermaphrodites. Ecol Lett 3:495–502. doi:10.1046/j.1461-0248.2000.00173.x
Pannell JR, Obbard DJ, Buggs RJA (2004) Polyploidy and the sexual system: what can we learn from Mercurialis annua? Biol J Linn Soc 82:547–560. doi:10.1111/j.1095-8312.2004.00340.x
Pickup M, Barrett SCH (2012) Reversal of height dimorphism promotes pollen and seed dispersal in a wind-pollinated dioecious plant. Biol Lett 8:245–248. doi:10.1098/rsbl.2011.0950
R Core Team (2013) R: a language and environment for statistical computing, 3.0.2. R Foundation for Statistical Computing, Vienna
Ruberti I, Sessa G, Ciolfi A, Possenti M, Carabelli M, Morelli G (2012) Plant adaptation to dynamically changing environment: the shade avoidance response. Biotechnol Adv 30:1047–1058. doi:10.1016/j.biotechadv.2011.08.014
Sanchez Vilas J, Pannell JR (2011) Sexual dimorphism in resource acquisition and deployment: both size and timing matter. Ann Bot 107:119–126. doi:10.1093/aob/mcq209
Sanchez-Vilas J, Turner A, Pannell JR (2011) Sexual dimorphism in intra- and interspecific competitive ability of the dioecious herb Mercurialis annua. Plant Biol 13:218–222. doi:10.1111/j.1438-8677.2010.00408.x
Sangoi L, Gracietti MA, Rampazzo C, Bianchetti P (2002) Response of Brazilian maize hybrids from different eras to changes in plant density. Field Crops Res 79:39–51. doi:10.1016/s0378-4290(02)00124-7
Schlichting CD (1986) The evolution of phenotypic plasticity in plants. Annu Rev Ecol Syst 17:667–693. doi:10.1146/annurev.es.17.110186.003315
Schmitt J, Wulff RD (1993) Light spectral quality, phytochrome and plant competition. Trends Ecol Evol 8:47–51. doi:10.1016/0169-5347(93)90157-k
Schwinning S, Weiner J (1998) Mechanisms determining the degree of size asymmetry in competition among plants. Oecologia 113:447–455. doi:10.1007/s004420050397
Shea MM, Dixon PM, Sharitz RR (1993) Size differences, sex ratio, and spatial distribution of male and female water tupelo, Nyssa aquatica (Nyssaceae). Am J Bot 80:26–30. doi:10.2307/2445116
Shine R (1989) Ecological causes for the evolution of sexual dimorphism: a review of the evidence. Q Rev Biol 64:419–461. doi:10.1086/416458
Shuster SM, Wade MJ (2003) Mating systems and strategies. In: Monographs in behavior and ecology. Princeton University Press, NJ
Skogsmyr I, Lankinen A (2002) Sexual selection: an evolutionary force in plants. Biol Rev 77:537–562. doi:10.1017/s1464793102005973
Slatkin M (1984) Ecological causes of sexual dimorphism. Evolution 38:622–630. doi:10.2307/2408711
Sleeman JD, Dudley SA, Pannell JR, Barrett SCH (2002) Responses of carbon acquisition traits to irradiance and light quality in Mercurialis annua (Euphorbiaceae): evidence for weak integration of plastic responses. Am J Bot 89:1388–1400. doi:10.3732/ajb.89.9.1388
Stark LR, McLetchie DN, Mishler BD (2005) Sex expression, plant size, and spatial segregation of the sexes across a stress gradient in the desert moss Syntrichia caninervis. Bryologist 108:183–193. doi:10.1639/0007-2745(2005)108[0183:sepsas]2.0.co;2
Stillwell RC, Blanckenhorn WU, Teder T, Davidowitz G, Fox CW (2010) Sex differences in phenotypic plasticity affect variation in sexual size dimorphism in insects: from physiology to evolution. Annu Rev Entomol 55:227–245
Sultan SE (2000) Phenotypic plasticity for plant development, function and life history. Trends Plant Sci 5:537–542. doi:10.1016/s1360-1385(00)01797-0
Tutin TG et al (1968) Flora Europea. Cambridge University Press, Cambridge
Vanderwerf HMG, Wijlhuizen M, Deschutter JAA (1995) Plant density and self thinning affect yield and quality of fiber hemp (Cannabis sativa L.). Field Crops Res 40:153–164. doi:10.1016/0378-4290(94)00103-j
Wallace CS, Rundel PW (1979) Sexual dimorphism and resource allocation in male and female shrubs of Simmondsia chinensis. Oecologia 44:34–39. doi:10.1007/bf00346394
Weller DE (1987) A reevaluation of the −3/2 power rule of plant self-thinning. Ecol Monogr 57:23–43. doi:10.2307/1942637
Acknowledgments
We thank T. Martignier, M. Voillemot, I. El M’Ghari and K. Gullotta for field assistance and data collection, R. Piault for statistical advice, and the Swiss National Science Foundation for funding (grant no. 31003A_141052, to J. R. P.). We thank Anne Worley and the anonymous reviewers for helpful comments on the manuscript.
Author contribution
A. M. L. and J. R. P. conceived and designed the experiment; A. M. L. performed the experiment and analysed the data. A. M. L. and J. R. P. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Anne Worley.
Rights and permissions
About this article
Cite this article
Labouche, AM., Pannell, J.R. A test of the size-constraint hypothesis for a limit to sexual dimorphism in plants. Oecologia 181, 873–884 (2016). https://doi.org/10.1007/s00442-016-3616-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-016-3616-3