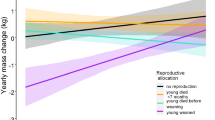
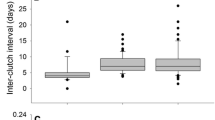
Abstract
Optimisation of reproductive investment is crucial for Darwinian fitness, and detailed long-term studies are especially suited to unravel reproductive allocation strategies. Allocation strategies depend on the timing of resource acquisition, the timing of resource allocation, and trade-offs between different life-history traits. A distinction can be made between capital breeders that fuel reproduction with stored resources and income breeders that use recently acquired resources. In capital breeders, but not in income breeders, energy allocation may be decoupled from energy acquisition. Here, we tested the influence of extrinsic (weather conditions) and intrinsic (female characteristics) factors during energy storage, vitellogenesis and early gestation on reproductive investment, including litter mass, litter size, offspring mass and the litter size and offspring mass trade-off. We used data from a long-term study of the viviparous lizard, Lacerta (Zootoca) vivipara. In terms of extrinsic factors, rainfall during vitellogenesis was positively correlated with litter size and mass, but temperature did not affect reproductive investment. With respect to intrinsic factors, litter size and mass were positively correlated with current body size and postpartum body condition of the previous year, but negatively with parturition date of the previous year. Offspring mass was negatively correlated with litter size, and the strength of this trade-off decreased with the degree of individual variation in resource acquisition, which confirms theoretical predictions. The combined effects of past intrinsic factors and current weather conditions suggest that common lizards combine both recently acquired and stored resources to fuel reproduction. The effect of past energy store points out a trade-off between current and future reproduction.




Similar content being viewed by others
References
Adolph SC, Porter WP (1993) Temperature, activity, and lizard life histories. Am Nat 142:273–295. doi:10.1086/285538
Alonso-Alvarez C, Bertrand S, Devevey G, Prost J, Faivre B, Sorci G (2004) Increased susceptibility to oxidative stress as a proximate cost of reproduction. Ecol Lett 7:363–368. doi:10.1111/j.1461-0248.2004.00594.x
Avery RA (1974) Storage lipids in the lizard Lacerta vivipara: a quantitative study. J Zool 173:419–425. doi:10.1111/j.1469-7998.1974.tb04124.x
Avery RA (1975) Clutch size and reproductive effort in the lizard Lacerta vivipara Jacquin. Oecologia 19:165–170. doi:10.1007/BF00369099
Bauwens D (1981) Survivorship during hibernation in the European common lizard, Lacerta vivipara. Copeia 1981:741–744. doi:10.2307/1444592
Bauwens D, Verheyen RF (1985) The timing of reproduction in the lizard Lacerta vivipara: differences between individual females. J Herpetol 19:353–364. doi:10.2307/1564263
Berglund A, Rosenqvist G (1986) Reproductive costs in the prawn Palaemon adspersus: effects on growth and predator vulnerability. Oikos 46:349–354. doi:10.2307/3565833
Bleu J, Le Galliard J-F, Meylan S, Massot M, Fitze PS (2011) Mating does not influence reproductive investment, in a viviparous lizard. J Exp Zool 315A:458–464. doi:10.1002/jez.693
Bleu J, Massot M, Haussy C, Meylan S (2012) Experimental litter size reduction reveals costs of gestation and delayed effects on offspring in a viviparous lizard. Proc R Soc B 279:489–498. doi:10.1098/rspb.2011.0966
Bonnet X, Naulleau G, Shine R, Lourdais O (2001) Short-term versus long-term effects of food intake on reproductive output in a viviparous snake, Vipera aspis. Oikos 92:297–308. doi:10.1034/j.1600-0706.2001.920212.x
Brown CA (2003) Offspring size-number trade-offs in scorpions: an empirical test of the van Noordwijk and de Jong model. Evolution 57:2184–2190. doi:10.1111/j.0014-3820.2003.tb00397.x
Cadby CD, While GM, Hobday AJ, Uller T, Wapstra E (2010) Multi-scale approach to understanding climate effects on offspring size at birth and date of birth in a reptile. Integr Zool 5:164–175. doi:10.1111/j.1749-4877.2010.00201.x
Chamaillé-Jammes S, Massot M, Aragón P, Clobert J (2006) Global warming and positive fitness response in mountain populations of common lizards Lacerta vivipara. Global Change Biol 12:392–402. doi:10.1111/j.1365-2486.2005.01088.x
Charnov EL, Downhower JF, Brown LP (1995) Optimal offspring sizes in small litters. Evol Ecol 9:57–63. doi:10.1007/BF01237697
Christians JK (2000) Trade-offs between egg size and number in waterfowl: an interspecific test of the van Noordwijk and de Jong model. Funct Ecol 14:497–501. doi:10.1046/j.1365-2435.2000.00444.x
Clobert J, Massot M, Lecomte J, Sorci G, De Fraipont M, Barbault R (1994) Determinants of dispersal behavior: the common lizard as a case study. In: Vitt LJ, Pianka ER (eds) Lizard ecology: historical and experimental perspectives. Princeton University Press, Princeton, pp 183–206
Clutton-Brock TH, Guinness FE, Albon SD (1983) The costs of reproduction to red deer hinds. J Anim Ecol 52:367–383. doi:10.2307/4560
Cox RM, Calsbeek R (2010) Severe costs of reproduction persist in Anolis lizards despite the evolution of a single-egg clutch. Evolution 64:1321–1330. doi:10.1111/j.1558-5646.2009.00906.x
Cox RM, Parker EU, Cheney DM, Liebl AL, Martin LB, Calsbeek R (2010) Experimental evidence for physiological costs underlying the trade-off between reproduction and survival. Funct Ecol 24:1262–1269. doi:10.1111/j.1365-2435.2010.01756.x
Doughty P, Shine R (1997) Detecting life history trade-offs: measuring energy stores in “capital” breeders reveals costs of reproduction. Oecologia 110:508–513. doi:10.1007/s004420050187
Doughty P, Shine R (1998) Reproductive energy allocation and long-term energy stores in a viviparous lizard (Eulamprus tympanum). Ecology 79:1073–1083. doi:10.1890/0012-9658(1998)079[1073:REAALT]2.0.CO;2
Festa-Bianchet M (1998) Condition-dependent reproductive success in bighorn ewes. Ecol Lett 1:91–94. doi:10.1046/j.1461-0248.1998.00023.x
French SS, DeNardo DF, Moore MC (2007) Trade-offs between the reproductive and immune systems: facultative responses to resources or obligate responses to reproduction? Am Nat 170:79–89. doi:10.1086/518569
Glazier DS (1999) Trade-offs between reproductive and somatic (storage) investments in animals: a comparative test of the Van Noordwijk and De Jong model. Evol Ecol 13:539–555. doi:10.1023/A:1006793600600
González-Suárez M, Mugabo M, Decencière B, Perret S, Claessen D, Le Galliard J-F (2011) Disentangling the effects of predator body size and prey density on prey consumption in a lizard. Funct Ecol 25:158–165. doi:10.1111/j.1365-2435.2010.01776.x
Gregory PT (2006) Influence of income and capital on reproduction in a viviparous snake: direct and indirect effects. J Zool 270:414–419. doi:10.1111/j.1469-7998.2006.00149.x
Gustafsson L, Nordling D, Andersson MS, Sheldon BC, Qvarnström A (1994) Infectious diseases, reproductive effort and the cost of reproduction in birds. Philos Trans R Soc Lond B 346:323–331. doi:10.1098/rstb.1994.0149
Hadley GL, Rotella JJ, Garrott RA (2007) Evaluation of reproductive costs for Weddell seals in Erebus Bay, Antarctica. J Anim Ecol 76:448–458. doi:10.1111/j.1365-2656.2007.01219.x
Hanssen SA, Hasselquist D, Folstad I, Erikstad KE (2005) Cost of reproduction in a long-lived bird: incubation effort reduces immune function and future reproduction. Proc R Soc B 272:1039–1046. doi:10.1098/rspb.2005.3057
Hare KM, Cree A (2010) Exploring the consequences of climate-induced changes in cloud cover on offspring of a cool-temperate viviparous lizard. Biol J Linn Soc 101:844–851. doi:10.1111/j.1095-8312.2010.01536.x
Houston AI, Stephens PA, Boyd IL, Harding KC, McNamara JM (2007) Capital or income breeding? A theoretical model of female reproductive strategies. Behav Ecol 18:241–250. doi:10.1093/beheco/arl080
Huey RB (1982) Temperature, physiology, and the ecology of reptiles. In: Gans C, Pough FH (eds) Biology of the reptilia. Academic Press, New-York, pp 25–91
Jordan MA, Snell HL (2002) Life history trade-offs and phenotypic plasticity in the reproduction of Galapagos lava lizards (Microlophus delanonis). Oecologia 130:44–52. doi:10.1007/s004420100776
Koivula M, Koskela E, Mappes T, Oksanen TA (2003) Cost of reproduction in the wild: manipulation of reproductive effort in the bank vole. Ecology 84:398–405. doi:10.1890/0012-9658(2003)084[0398:CORITW]2.0.CO;2
Landwer AJ (1994) Manipulation of egg production reveals costs of reproduction in the tree lizard (Urosaurus ornatus). Oecologia 100:243–249. doi:10.1007/BF00316951
Le Galliard J-F, Cote J, Fitze PS (2008) Lifetime and intergenerational fitness consequences of harmful male interactions for female lizards. Ecology 89:56–64. doi:10.1890/06-2076.1
Le Galliard J-F, Marquis O, Massot M (2010) Cohort variation, climate effects and population dynamics in a short-lived lizard. J Anim Ecol 79:1296–1307. doi:10.1111/j.1365-2656.2010.01732.x
Le Galliard J-F, Massot M, Baron J-P, Clobert J (2012) Ecological effects of climate change on European reptiles. In: Brodie JF, Post E, Doak D (eds) Conserving wildlife populations in a changing climate. University of Chicago Press, Chicago
Lourdais O, Bonnet X, Shine R, Denardo D, Naulleau G, Guillon M (2002) Capital-breeding and reproductive effort in a variable environment: a longitudinal study of a viviparous snake. J Anim Ecol 71:470–479. doi:10.1046/j.1365-2656.2002.00612.x
Lourdais O, Bonnet X, Shine R, Taylor EN (2003) When does a reproducing female viper (Vipera aspis) “decide” on her litter size? J Zool 259:123–129. doi:10.1017/s0952836902003059
Lourdais O, Shine R, Bonnet X, Guillon M, Naulleau G (2004) Climate affects embryonic development in a viviparous snake, Vipera aspis. Oikos 104:551–560. doi:10.1111/j.0030-1299.2004.12961.x
Marquis O, Massot M, Le Galliard J-F (2008) Intergenerational effects of climate generate cohort variation in lizard reproductive performance. Ecology 89:2575–2583. doi:10.1890/07-1211.1
Massot M, Clobert J (1995) Influence of maternal food availability on offspring dispersal. Behav Ecol Sociobiol 37:413–418. doi:10.1007/BF00170589
Massot M, Clobert J, Montes-Poloni L, Haussy C, Cubo J, Meylan S (2011) An integrative study of ageing in a wild population of common lizards. Funct Ecol 25:848–858. doi:10.1111/j.1365-2435.2011.01837.x
Mugabo M, Marquis O, Perret S, Le Galliard J-F (2011) Direct and socially mediated effects of food availability late in life on life-history variation in a short-lived lizard. Oecologia 166:949–960. doi:10.1007/s00442-011-1933-0
Panigel M (1956) Contribution à l’étude de l’ovoviviparité chez les reptiles: gestation et parturition chez le lézard vivipare Zootoca vivipara. Ann Sci Nat Zool 18:569–668
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-PLUS. Springer, New-York
Qualls CP, Shine R (1995) Maternal body-volume as a constraint on reproductive output in lizards: evidence from the evolution of viviparity. Oecologia 103:73–78. doi:10.1007/BF00328427
Reading CJ (2004) The influence of body condition and prey availability on female breeding success in the smooth snake (Coronella austriaca Laurenti). J Zool 264:61–67. doi:10.1017/S0952836904005515
Richner H, Tripet F (1999) Ectoparasitism and the trade-off between current and future reproduction. Oikos 86:535–538. doi:10.2307/3546657
Roff DA (2002) Life history evolution. Sinauer Assoc, Sunderland
Shine R (2005) Life-history evolution in reptiles. Annu Rev Ecol Evol Syst 36:23–46. doi:10.1146/annurev.ecolsys.36.102003.152631
Shine R, Downes SJ (1999) Can pregnant lizards adjust their offspring phenotypes to environmental conditions? Oecologia 119:1–8. doi:10.1007/s004420050754
Shine R, Harlow P (1993) Maternal thermoregulation influences offspring viability in a viviparous lizard. Oecologia 96:122–127. doi:10.1007/BF00318039
Stephens PA, Boyd IL, McNamara JM, Houston AI (2009) Capital breeding and income breeding: their meaning, measurement, and worth. Ecology 90:2057–2067. doi:10.1890/08-1369.1
Stewart JR, Ecay TW, Heulin B (2009) Calcium provision to oviparous and viviparous embryos of the reproductively bimodal lizard Lacerta (Zootoca) vivipara. J Exp Biol 212:2520–2524. doi:10.1242/jeb.030643
Swain R, Jones SM (2000) Maternal effects associated with gestation conditions in a viviparous lizard, Niveoscincus metallicus. Herpetol Monogr 14:432–440. doi:10.2307/1467056
Uller T, Olsson M (2005) Trade-offs between offspring size and number in the lizard Lacerta vivipara: a comparison between field and laboratory conditions. J Zool 265:295–299. doi:10.1017/s0952836904006326
Uller T, While GM, Wapstra E, Warner DA, Goodman BA, Schwarzkopf L, Langkilde T, Doughty P, Radder RS, Rohr DH, Bull CM, Shine R, Olsson M (2009) Evaluation of offspring size-number invariants in 12 species of lizard. J Evol Biol 22:143–151. doi:10.1111/j.1420-9101.2008.01629.x
Uller T, While GM, Cadby CD, Harts A, O’Connor K, Pen I, Wapstra E (2011) Altitudinal divergence in maternal thermoregulatory behaviour may be driven by differences in selection on offspring survival in a viviparous lizard. Evolution 65:2313–2324. doi:10.1111/j.1558-5646.2011.01303.x
van Noordwijk AJ, de Jong G (1986) Acquisition and allocation of resources: their influence on variation in life history tactics. Am Nat 128:137–142. doi:10.1086/284547
Wapstra E (2000) Maternal basking opportunity affects juvenile phenotype in a viviparous lizard. Funct Ecol 14:345–352. doi:10.1046/j.1365-2435.2000.00428.x
Warner DA, Bonnet X, Hobson KA, Shine R (2008) Lizards combine stored energy and recently acquired nutrients flexibly to fuel reproduction. J Anim Ecol 77:1242–1249. doi:10.1111/j.1365-2656.2008.01442.x
Webb JK, Shine R, Christian KA (2006) The adaptive significance of reptilian viviparity in the tropics: testing the maternal manipulation hypothesis. Evolution 60:115–122. doi:10.1554/05-460.1
Winkler DW, Wallin K (1987) Offspring size and number: a life history model linking effort per offspring and total effort. Am Nat 129:708–720. doi:10.1086/284667
Acknowledgments
We are grateful to the students who helped collect data and to the Parc National des Cévennes and the Office National des Forêts for providing good conditions during fieldwork. We thank Météo-France for providing the weather data used in this study. Financial support was received from the Agence Nationale de la Recherche (grant 07-JCJC-0120 to J.F.L.G and grant 07-BLAN-0217 to M.M. and J.C.), the Spanish Ministry of Education and Science (CGL2005-01187, CGL2008-01522, and Programa Ramón y Cajal to P. S. F.), the Swiss National Science Foundation (PPOOP3_128375) and the Ministère de l’Éducation Supérieure et de la Recherche (PhD grant to J.B.).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Raoul Van Damme.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bleu, J., Le Galliard, JF., Fitze, P.S. et al. Reproductive allocation strategies: a long-term study on proximate factors and temporal adjustments in a viviparous lizard. Oecologia 171, 141–151 (2013). https://doi.org/10.1007/s00442-012-2401-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-012-2401-1