Abstract
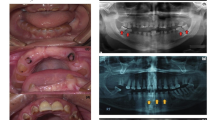
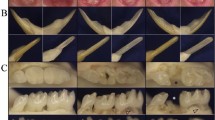
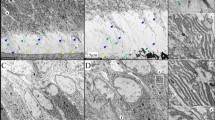
The current system for the classification of hereditary defects of tooth dentin is based upon clinical and radiographic findings and consists of two types of dentin dysplasia (DD) and three types of dentinogenesis imperfecta (DGI). However, whether DGI type III should be considered a distinct phenotype or a variation of DGI type II is debatable. In the 30 years since the classification system was first proposed, significant advances have been made regarding the genetic etiologies of inherited dentin defects. DGI type II is recognized as an autosomal dominant disorder with almost complete penetrance and a low frequency of de novo mutations. We have identified a mutation (c.52G→T, p.V18F) at the first nucleotide of exon 3 of the DSPP (dentin sialophosphoprotein) gene in a Korean family (de novo) and a Caucasian family. This mutation has previously been reported as causing DGI type II in a Chinese family. These findings suggest that this mutation site represents a mutational “hot spot” in the DSPP gene. The clinical and radiographic features of these two families include the classic phenotypes associated with both DGI type II and type III. Finding that a single mutation causes both phenotypic patterns strongly supports the conclusion that DGI type II and DGI type III are not separate diseases but rather the phenotypic variation of a single disease. We propose a modification of the current classification system such that the designation “hereditary opalescent dentin” or “DGI type II” should be used to describe both the DGI type II and type III phenotypes.


Similar content being viewed by others
References
Bixler D, Conneally PM, Christen AG (1969) Dentinogenesis imperfecta: genetic variations in a six-generation family. J Dent Res 48:1196–1199
Clark F, Thanaraj TA (2002) Categorization and characterization of transcript-confirmed constitutively and alternatively spliced introns and exons from human. Hum Mol Genet 11:451–464
Esposito S, Vergo TJ Jr (1978) Removable overdentures in the oral rehabilitation of patients with dentinogenesis imperfecta. J Pedod 2:304–315
Heimler A, Sciubba J, Lieber E, Kamen S (1985) An unusual presentation of opalescent dentin and Brandywine isolate hereditary opalescent dentin in an Ashkenazic Jewish family. Oral Surg Oral Med Oral Pathol 59:608–615
Kim J-W, Nam S-H, Jang K-T, Lee S-H, Kim C-C, Hahn S-H, Hu JC-C, Simmer JP (2004) A novel splice acceptor mutation in the DSPP gene causing dentinogenesis imperfecta type II. Hum Genet 115:248–254
Malmgren B, Lundberg M, Lindskog S (1988) Dentinogenesis imperfecta in a six-generation family. A clinical, radiographic and histologic comparison of two branches through three generations. Swed Dent J 12:73–84
Malmgren B, Lindskog S, Elgadi A, Norgren S (2004) Clinical, histopathologic, and genetic investigation in two large families with dentinogenesis imperfecta type II. Hum Genet 114:491–498
Rajpar MH, Koch MJ, Davies RM, Mellody KT, Kielty CM, Dixon MJ (2002) Mutation of the signal peptide region of the bicistronic gene DSPP affects translocation to the endoplasmic reticulum and results in defective dentine biomineralization. Hum Mol Genet 11:2559–2565
Reiskin A (1981) Dentinogenesis imperfecta. Quintessence Int 12:617–622
Roberts E, Schour I (1939) Hereditary opalescent dentine (dentinogenesis imperfecta). Am J Orthod Oral Surg 25:267–276
Sapir S, Shapira J (2001) Dentinogenesis imperfecta: an early treatment strategy. Pediatr Dent 23:232–237
Shields ED, Bixler D, el-Kafrawy AM (1973) A proposed classification for heritable human dentine defects with a description of a new entity. Arch Oral Biol 18:543–553
Skillen WG (1937) Histologic and clinical study of hereditary opalescent dentin. J Am Dent Assoc 24:1426–1433
Sreenath T, Thyagarajan T, Hall B, Longenecker G, D’Souza R, Hong S, Wright JT, MacDougall M, Sauk J, Kulkarni AB (2003) Dentin sialophosphoprotein knockout mouse teeth display widened predentin zone and develop defective dentin mineralization similar to human dentinogenesis imperfecta type III. J Biol Chem 278:24874–24880
Witkop CJ Jr (1957) Hereditary defects in enamel and dentin. Acta Genet Stat Med 7:236–239
Witkop C Jr (1965) Genetic diseases of the oral cavity. In: Tiecke RW (ed) Oral pathology. McGraw Hill, New York, pp 786–843
Witkop CJ Jr (1971) Manifestations of genetic diseases in the human pulp. Oral Surg Oral Med Oral Pathol 32:278–316
Witkop CJ Jr (1989) Amelogenesis imperfecta, dentinogenesis imperfecta and dentin dysplasia revisited: problems in classification. J Oral Path 17:547–553
Xiao S, Yu C, Chou X, Yuan W, Wang Y, Bu L, Fu G, Qian M, Yang J, Shi Y, Hu L, Han B, Wang Z, Huang W, Liu J, Chen Z, Zhao G, Kong X (2001) Dentinogenesis imperfecta 1 with or without progressive hearing loss is associated with distinct mutations in DSPP (erratum appears in Nat Genet 2001, 27(3):345). Nat Genet 27:201–204
Zhang X, Zhao J, Li C, Gao S, Qiu C, Liu P, Wu G, Qiang B, Lo WH, Shen Y (2001) DSPP mutation in dentinogenesis imperfecta Shields type II. Nat Genet 27:151–152
Acknowledgements
We thank all the family members for their cooperation. This investigation was supported in part by the Foundation of the American Academy of Pediatric Dentistry, and USPHS Research Grants DE15846 and DE11301 from the National Institute of Dental and Craniofacial Research, National Institutes of Health, Bethesda, MD 29892.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, JW., Hu, J.CC., Lee, JI. et al. Mutational hot spot in the DSPP gene causing dentinogenesis imperfecta type II. Hum Genet 116, 186–191 (2005). https://doi.org/10.1007/s00439-004-1223-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-004-1223-6