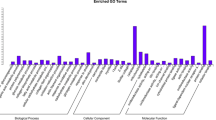
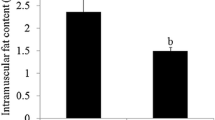
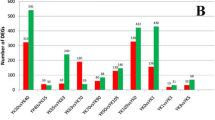
Abstract
Exposure to high ambient temperatures is detrimental to pig rearing and porcine meat quality. Deep molecular sequencing allows for genomic characterization of porcine skeletal muscles and helps understand how the genomic landscape may impact meat quality. To this end, we performed mRNA-seq to molecularly dissect the impact of heat stress on porcine skeletal muscles, longissimus dorsi. Sixteen castrated, male DLY pigs [which are crossbreeds between Duroc (D) boars and Landrace (L) × Yorkshire (Y) sows, 79.0 ± 1.5 kg BW] were evenly split into two groups that were subjected to either control (CON) (22 °C; 55 % humidity) or constant heat stress (H30; 30 °C; 55 % humidity) conditions for 21 days. Seventy-eight genes were found to be differentially expressed, of which 37 were up-regulated and 41 were down-regulated owing to constant heat stress. We predicted 5247 unknown genes and 6108 novel transcribed units attributed to alternative splicing (AS) events in the skeletal muscle. Furthermore, 30,761 and 31,360 AS events were observed in the CON and H30 RNA-seq libraries, respectively. The differentially expressed genes in the porcine skeletal muscles were involved in glycolysis, lactate metabolism, lipid metabolism, cellular defense, and stress responses. Additionally, the expression levels of these genes were associated with variations in meat quality between the CON and H30 groups, indicating that heat stress modulated genes crucial to skeletal muscle development and metabolism. Our transcriptomic analysis provides valuable information for understanding the molecular mechanisms governing porcine skeletal muscle development. Such insights may lead to innovative strategies to improve meat quality of pigs under heat stress.






Similar content being viewed by others
References
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11:R106
Bai Q, McGillivray C, da Costa N, Dornan S, Evans G, Stear MJ, Chang KC (2003) Development of a porcine skeletal muscle cDNA microarray: analysis of differential transcript expression in phenotypically distinct muscles. BMC Genom 4:8
Bazin R, Ferre P (2001) Assays of lipogenic enzymes. Methods Mol Biol 155:121–127
Bennett MJ, Russell LK, Tokunaga C, Narayan SB, Tan L, Seegmiller A, Boriack RL, Strauss AW (2006) Reye-like syndrome resulting from novel missense mutations in mitochondrial medium- and short-chain l-3-hydroxy-acyl-coa dehydrogenase. Mol Genet Metab 89:74–79
Cumming KT, Paulsen G, Wernbom M, Ugelstad I, Raastad T (2014) Acute response and subcellular movement of HSP27, αB-crystallin and HSP70 in human skeletal muscle after blood-flow-restricted low-load resistance exercise. Acta Physiol 211:634–646
Davoli R, Fontanesi L, Zambonelli P, Bigi D, Gellin J, Yerle M, Milc J, Braglia S, Cenci V, Cagnazzo M, Russo V (2002) Isolation of porcine expressed sequence tags for the construction of a first genomic transcript map of the skeletal muscle in pig. Anim Genet 33:3–18
Fujita J (1999) Cold shock response in mammalian cells. J Mol Microb Biotechnol 1:243–255
Gregory N (2010) How climatic changes could affect meat quality. Food Res Int 43:1866–1873
Hao Y, Feng YJ, Yang PG, Feng JH, Lin H, Gu XH (2014) Nutritional and physiological responses of finishing pigs exposed to a permanent heat exposure during 3 weeks. Arch Anim Nutri 68:296–308
Huang S, Zhang J, Li R, Zhang W, He Z, Lam TW, Peng Z, Yiu SM (2011) SOAPsplice: genome-wide ab initio detection of splice junctions from RNA-seq data. Front Genet 2:46
Huff Lonergan E, Zhang GW, Lonergan SM (2010) Biochemistry of postmortem muscle—lessons on mechanisms of meat tenderization. Meat Sci 86:184–195
Kamanga-Sollo E, Pampusch MS, White ME, Hathaway MR, Dayton WR (2011) Effects of heat stress on proliferation, protein turnover, and abundance of heat shock protein messenger ribonucleic acid in cultured porcine muscle satellite cells. J Anim Sci 89:3473–3480
Kim JS, Ingale SL, Lee SH, Choi YH, Kim EH, Lee DC, Kim YH, Chae BJ (2014) Impact of dietary fat sources and feeding level on adipose tissue fatty acids composition and lipid metabolism related gene expression in finisher pigs. Anim Feed Sci Tech 196:60–67
Kim TH, Kim NS, Lim D, Lee KT, Oh JH, Park HS, Jang GW, Kim HY, Jeon M, Choi BH, Lee HY, Chung HY, Kim H (2006) Generation and analysis of large-scale expressed sequence tags (ESTs) from a full-length enriched cDNA library of porcine backfat tissue. BMC Genom 7:36
Komiyama M, Zhou ZH, Maruyama K, Shimada Y (1992) Spatial relationship of nebulin relative to other myofibrillar proteins during myogenesis in embryonic chick skeletal muscle cells in vitro. J Muscle Res Cell M 13:48–54
Labeit S, Kolmerer B (1995) Titins: giant proteins in charge of muscle ultrastructure and elasticity. Science 270:293–296
Langmead B, Trapnell C, Pop M, Salzberg SL (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:R25
Li J, Buchner J (2013) Structure, function and regulation of the hsp90 machinery. Biomed J 36:106–117
Li Y, Xu Z, Li H, Xiong Y, Zuo B (2010) Differential transcriptional analysis between red and white skeletal muscle of chinese meishan pigs. Int J Biol Sci 6:350–360
Lindquist S (1986) The heat-shock response. Annu Rev Biochem 55:1151–1191
Liu CY, Grant AL, Kim KH, Mills SE (1994) Porcine somatotropin decreases acetyl-coa carboxylase gene expression in porcine adipose tissue. Domest Anim Endocrin 11:125–132
Liu Y, Steinacker JM (2001) Changes in skeletal muscle heat shock proteins: pathological significance. Front Biosci 6:D12–D25
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Lobjois V, Liaubet L, SanCristobal M, Glenisson J, Feve K, Rallieres J, Le Roy P, Milan D, Cherel P, Hatey F (2008) A muscle transcriptome analysis identifies positional candidate genes for a complex trait in pig. Anim Genet 39:147–162
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the kegg orthology (ko) as a controlled vocabulary. Bioinformatics 21:3787–3793
Melody JL, Lonergan SM, Rowe LJ, Huiatt TW, Mayes MS, Huff-Lonergan E (2004) Early postmortem biochemical factors influence tenderness and water-holding capacity of three porcine muscles. J Anim Sci 82:1195–1205
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5:621–628
Mustafi SB, Chakraborty PK, Dey RS, Raha S (2009) Heat stress upregulates chaperone heat shock protein 70 and antioxidant manganese superoxide dismutase through reactive oxygen species (ros), p38mapk, and akt. Cell Stress Chaperon 14:579–589
Neufer PD, Benjamin IJ (1996) Differential expression of B-crystallin and Hsp27 in skeletal muscle during continuous contractile activity. Relationship to myogenic regulatory factors. J Biol Chem 271:24089–24095
Nussbaum EL, Locke M (2007) Heat shock protein expression in rat skeletal muscle after repeated applications of pulsed and continuous ultrasound. Arch Phys Med Rehab 88:785–790
Pan PJ, Hsu CF, Tsai JJ, Chiu JH (2012) The role of oxidative stress response revealed in preconditioning heat stimulation in skeletal muscle of rats. J Surg Res 176:108–113
Patience J, Umboh J, Chaplin R, Nyachoti C (2005) Nutritional and physiological responses of growing pigs exposed to a diurnal pattern of heat stress. Livest Prod Sci 96:205–214
Ponsuksili S, Jonas E, Murani E, Phatsara C, Srikanchai T, Walz C, Schwerin M, Schellander K, Wimmers K (2008) Trait correlated expression combined with expression QTL analysis reveals biological pathways and candidate genes affecting water holding capacity of muscle. BMC Genom 9:367
Qiao Y, Huang ZG, Li QF, Liu ZS, Hao CL, Shi GQ, Dai R, Xie Z (2007) Developmental changes of the FAS and HSL mRNA expression and their effects on the content of intramuscular fat in Kazak and Xinjiang sheep. J Genet Genom 34:909–917
Ropka-Molik K, Zukowski K, Eckert R, Gurgul A, Piórkowska K, Oczkowicz M (2014) Comprehensive analysis of the whole transcriptomes from two different pig breeds using RNA-Seq method. Anim Genet 45:674–684
Sebastian S, Faralli H, Yao ZZ, Rakopoulos P, Palii C, Cao Y, Singh K, Liu QC, Chu A, Aziz A, Brand M, Tapscott SJ, Dilworth FJ (2013) Tissue-specific splicing of a ubiquitously expressed transcription factor is essential for muscle differentiation. Gene Dev 27:1247–1259
Sparrow JC, Nowak KJ, Durling HJ, Beggs AH, Wallgren-Pettersson C, Romero N, Nonaka I, Laing NG (2003) Muscle disease caused by mutations in the skeletal muscle alpha-actin gene (acta1). Neuromuscular Disord 13:519–531
Stallings JD, Ippolito DL, Rakesh V, Baer CE, Dennis WE, Helwig BG, Jackson DA, Leon LR, Lewis JA, Reifman J (2014) Patterns of gene expression associated with recovery and injury in heat-stressed rats. BMC Genom 15:1058–1076
Stossel TP, Condeelis J, Cooley L, Hartwig JH, Noegel A, Schleicher M, Shapiro SS (2001) Filamins as integrators of cell mechanics and signalling. Nat Rev Mol Cell Bio 2:138–145
Sugden MC, Holness M (1994) Interactive regulation of the pyruvate dehydrogenase complex and the carnitine palmitoyltransferase system. FASEB J 8:54–61
Tang S, Yang L, Tang X, Liu M (2014) The role of oxidized atm in the regulation of oxidative stress-induced energy metabolism reprogramming of cafs. Cancer Lett 353:133–144
Trapnell C, Pachter L, Salzberg SL (2009) Tophat: discovering splice junctions with rna-seq. Bioinformatics 25:1105–1111
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by rna-seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515
Trappe TA, Carrithers JA, White F, Lambert CP, Evans WJ, Dennis RA (2002) Titin and nebulin content in human skeletal muscle following eccentric resistance exercise. Muscle Nerve 25:289–292
Watt MJ, Hoy AJ (2012) Lipid metabolism in skeletal muscle: generation of adaptive and maladaptive intracellular signals for cellular function. Am J Physiol Endocrinol. Metab 302:E1315–E1328
Wei B, Jin JP (2011) Troponin T isoforms and posttranscriptional modifications: evolution, regulation and function. Arch Biochem Biophys 505:144–154
Xu Y, Qian H, Feng X, Xiong Y, Lei M, Ren Z, Zuo B, Xu D, Ma Y, Yuan H (2012) Differential proteome and transcriptome analysis of porcine skeletal muscle during development. J Proteomics 75:2093–2108
Yang PG, Hao Y, Feng JH, Lin H, Feng YJ, Wu X, Yang X, Gu XH (2014) The expression of carnosine and its effect on the antioxidant capacity of longissimus dorsi muscle in finishing pigs exposed to constant heat stress. Asian Austral J Anim Sci 27:1763–1772
Yao J, Coussens PM, Saama P, Suchyta S, Ernst CW (2002) Generation of expressed sequence tags from a normalized porcine skeletal muscle cDNA library. Anim Biotechnol 13:211–222
Yoshihara T, Naito H, Kakigi R, Ichinoseki-Sekine N, Ogura Y, Sugiura T, Katamoto S (2013) Heat stress activates the akt/mtor signalling pathway in rat skeletal muscle. Acta Physiol 207:416–426
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for rna-seq: accounting for selection bias. Genome Biol 11:R14
Zhu JY, Shi X, Lu HZ, Xia B, Li YF, Li X, Zhang QL, Yang GS (2015) RNA-seq transcriptome analysis of extensor digitorum longus and soleus muscles in large white pigs. Mol Genet Genom 291:687–701
Acknowledgments
The authors thank Novogene for excellent technical help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors declare any conflict of interest.
Ethical approval
Animal procedures were conducted in accordance with the Guidelines for Experimental Animals as established by the Ministry of Science and Technology (Beijing, China). All animal protocols were approved by the Institutional Animal Care and Use Committee of the Institute of Animal Sciences, Chinese Academy of Agricultural Sciences (CAAS) (Beijing, China).
Funding
This study was supported by grants from the Major State Basic Research Development Program of China (National 973 Project: 2012CB124706), National Natural Science of China (31501961), the Agricultural Science and Technology Innovation Program (ASTIP-IAS07), and the Key Project in the National Science and Technology Pillar Program (2012BAD39B0202). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2016_1242_MOESM2_ESM.pdf
Table S2: Gene Ontology (GO) assignments according to biological process, molecular function, and cellular component (PDF 132 kb)
438_2016_1242_MOESM4_ESM.pdf
SuppleTable S4: Primers for real-time RT-PCR of 11 randomly selected, differentially expressed genesmentary material 4 (PDF 72 kb)
Rights and permissions
About this article
Cite this article
Hao, Y., Feng, Y., Yang, P. et al. Transcriptome analysis reveals that constant heat stress modifies the metabolism and structure of the porcine longissimus dorsi skeletal muscle. Mol Genet Genomics 291, 2101–2115 (2016). https://doi.org/10.1007/s00438-016-1242-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-016-1242-8