Abstract
Purpose
A prolonged time to treatment initiation (TTI) correlates with an adverse prognosis in different cancer types including resectable pancreatic cancer (PC). Only limited evidence on the correlation between TTI and prognosis in advanced PC exists.
Methods
Consecutive PC patients (n = 368) who were diagnosed or treated at our high-volume comprehensive cancer center were included in a prospectively maintained database. We retrospectively analyzed time from first imaging showing advanced PC to initiation of palliative first-line chemotherapy. Lead time bias and waiting time paradox were addressed by landmark analysis and correlation of tumor burden with TTI.
Results
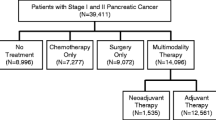
Two hundred and ninety-seven patients met the pre-specified in- and exclusion criteria of our study. Median TTI was 29 days (range: 1–124 days). Most common reasons for prolonged TTI (> 21 days) were referral from an external treatment center (39%) and a second biopsy (31%). A TTI above the median-, 75th or 90th percentile (43 or 60 days, respectively) had no impact on overall survival. Furthermore, no correlation between levels of carbohydrate antigen 19-9 (CA 19-9) at time of treatment initiation and TTI was observed.
Conclusion
While a timely work-up of advanced PC patients remains important, delays in treatment initiation due to repeated biopsies, inclusion in a clinical study or transfer to a specialized cancer center appear to be justified in light of the absence of a strong adverse effect of prolonged TTI on prognosis in advanced PC patients.



Similar content being viewed by others
Data availability
Not applicable.
References
Abdel-Rahman O, Xu Y, Tang PA, Lee-Ying RM, Cheung WY (2018) A real-world, population-based study of patterns of referral, treatment, and outcomes for advanced pancreatic cancer. Cancer Med 7:6385–6392. https://doi.org/10.1002/cam4.1841
Conroy T et al (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364:1817–1825. https://doi.org/10.1056/NEJMoa1011923
Crawford SC, Davis JA, Siddiqui NA, de Caestecker L, Gillis CR, Hole D, Penney G (2002) The waiting time paradox: population based retrospective study of treatment delay and survival of women with endometrial cancer in Scotland. BMJ 325:196
Faluyi OO, Connor JL, Chatterjee M, Ikin C, Wong H, Palmer DH (2017) Advanced pancreatic adenocarcinoma outcomes with transition from devolved to centralised care in a regional Cancer Centre. Br J Cancer 116:424–431. https://doi.org/10.1038/bjc.2016.406
Ferlay J et al (2013) Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer 49:1374–1403. https://doi.org/10.1016/j.ejca.2012.12.027
Glant JA et al (2011) Does the interval from imaging to operation affect the rate of unanticipated metastasis encountered during operation for pancreatic adenocarcinoma? Surgery 150:607–616. https://doi.org/10.1016/j.surg.2011.07.048
Gobbi PG, Bergonzi M, Comelli M, Villano L, Pozzoli D, Vanoli A, Dionigi P (2013) The prognostic role of time to diagnosis and presenting symptoms in patients with pancreatic cancer. Cancer Epidemiol 37:186–190. https://doi.org/10.1016/j.canep.2012.12.002
Haas M et al (2013) Prognostic value of CA 19-9, CEA, CRP, LDH and bilirubin levels in locally advanced and metastatic pancreatic cancer: results from a multicenter, pooled analysis of patients receiving palliative chemotherapy. J Cancer Res Clin Oncol 139:681–689. https://doi.org/10.1007/s00432-012-1371-3
Javed MA et al (2019) Impact of intensified chemotherapy in metastatic pancreatic ductal adenocarcinoma (PDAC) in clinical routine in Europe. Pancreatology 19:97–104. https://doi.org/10.1016/j.pan.2018.10.003(Official journal of the International Association of Pancreatology)
Jooste V, Dejardin O, Bouvier V, Arveux P, Maynadie M, Launoy G, Bouvier AM (2016) Pancreatic cancer: wait times from presentation to treatment and survival in a population-based study. Int J Cancer 139:1073–1080. https://doi.org/10.1002/ijc.30166
Khorana AA et al (2019) Time to initial cancer treatment in the United States and association with survival over time: an observational study. PloS One 14:e0213209. https://doi.org/10.1371/journal.pone.0213209
Lee SH et al (2019) Association of time interval between cancer diagnosis and initiation of palliative chemotherapy with overall survival in patients with unresectable pancreatic cancer. Cancer Med. https://doi.org/10.1002/cam4.2254
Murphy CT, Galloway TJ, Handorf EA, Wang L, Mehra R, Flieder DB, Ridge JA (2015) Increasing time to treatment initiation for head and neck cancer: an analysis of the National Cancer Database. Cancer 121:1204–1213. https://doi.org/10.1002/cncr.29191
Murphy CT et al (2016) Survival impact of increasing time to treatment initiation for patients with head and neck cancer in the United States. J Clin Oncol 34:169–178. https://doi.org/10.1200/JCO.2015.61.5906
Neal RD et al (2015) Is increased time to diagnosis and treatment in symptomatic cancer associated with poorer outcomes? Systematic review. Br J Cancer 112(Suppl 1):S92–S107. https://doi.org/10.1038/bjc.2015.48
Pitter JG, Lukacs G, Csanadi M, Voko Z (2018) Clinical impact of treatment delay in pancreatic cancer patients revisited. Int J Cancer 142:2621–2622. https://doi.org/10.1002/ijc.31263
Quante AS et al (2016) Projections of cancer incidence and cancer-related deaths in Germany by 2020 and 2030. Cancer Med 5:2649–2656. https://doi.org/10.1002/cam4.767
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM (2014) Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 74:2913–2921. https://doi.org/10.1158/0008-5472.can-14-0155
Raptis DA, Fessas C, Belasyse-Smith P, Kurzawinski TR (2010) Clinical presentation and waiting time targets do not affect prognosis in patients with pancreatic cancer. Surgeon 8:239–246. https://doi.org/10.1016/j.surge.2010.03.001(Journal of the Royal Colleges of Surgeons of Edinburgh and Ireland)
Sakahara H et al (1986) Serum CA 19-9 concentrations and computed tomography findings in patients with pancreatic carcinoma. Cancer 57:1324–1326
Sanjeevi S et al (2015) Impact of delay between imaging and treatment in patients with potentially curable pancreatic cancer. Br J Surg. https://doi.org/10.1002/bjs.10046
Sauerbrei W, Taube SE, McShane LM, Cavenagh MM, Altman DG (2018) Reporting recommendations for tumor marker prognostic studies (REMARK): an abridged explanation and elaboration. J Natl Cancer Inst 110:803–811. https://doi.org/10.1093/jnci/djy088
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65:5–29. https://doi.org/10.3322/caac.21254
Suzuki R et al (2019) An observation study of the prognostic effect of waiting times in the management of pancreatic ductal adenocarcinoma. Oncol Lett 17:587–593. https://doi.org/10.3892/ol.2018.9626
Von Hoff DD et al (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369:1691–1703. https://doi.org/10.1056/NEJMoa1304369
Yu J, Blackford AL, Dal Molin M, Wolfgang CL, Goggins M (2015) Time to progression of pancreatic ductal adenocarcinoma from low-to-high tumour stages. Gut 64:1783–1789. https://doi.org/10.1136/gutjnl-2014-308653
Yun YH et al (2012) The influence of hospital volume and surgical treatment delay on long-term survival after cancer surgery. Ann Oncol 23:2731–2737. https://doi.org/10.1093/annonc/mds101
Funding
Stephan Kruger is supported by the “Else Kröner-Forschungskolleg: Cancer Immunotherapy”.
Author information
Authors and Affiliations
Contributions
Writing of the manuscript and conception of the study: SK and SB. Assistance in writing the manuscript: SebK. Data analysis: SK, KS and AC. Analysis of tumor material (to confirm diagnosis of PC): SO, TK. Inclusion of patients: SK, MH, JS, JM, JGDH, WGK, JR, MI, LG, CBW, MvB, JW, VH, SB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study was approved by the local ethics committee of Ludwig-Maximilians-University of Munich (approval number 134-15). The study was performed in accordance with the Declaration of Helsinki.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2019_3061_MOESM1_ESM.eps
Suppl. Figure 1 Progression-free survival (PFS) according to time to treatment initiation (TTI). Data on PFS was available for 268 patients. Patients were divided into groups according to different cutoffs for TTI (median TTI, 75th and 90th percentile). After 29, 43 and 60 days one, three and four patients in the whole cohort had died, respectively, and were excluded by landmark analysis. Progression-free survival was calculated from the date of first imaging showing advanced pancreatic cancer. The log-rank test was used to test for a survival difference between groups. (EPS 112 kb)
Rights and permissions
About this article
Cite this article
Kruger, S., Schirle, K., Haas, M. et al. Prolonged time to treatment initiation in advanced pancreatic cancer patients has no major effect on treatment outcome: a retrospective cohort study controlled for lead time bias and waiting time paradox. J Cancer Res Clin Oncol 146, 391–399 (2020). https://doi.org/10.1007/s00432-019-03061-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-03061-4