Abstract
Purpose
The purpose of this study was to determine the relationship between hypomethylation of HOXA10 gene’s promoter and high expression in malignant ovarian tissues, and to confirm the level of hypomethylation in ovarian cell lines.
Experimental design
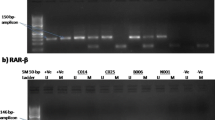
We performed the methylation status of 29 samples from ovarian carcinomas and 16 from normal tissues by methylation-specific polymerase chain reaction (MSP). Then, we evaluated the expression of mRNA and protein of HOXA10 in all samples to work out the relationship between the methylation status of HOXA10 and its expression in transcriptional and translational levels. We then confirmed our present study usingSKOV3 and HEY ovarian cancer cell lines treated with the demethylating agent 5-aza-2′-deoxycytidine (5-aza-dC) to detect whether the expression of HoxA10 in the two cell lines was altered.
Results
Increased expression of HOXA10 was detected in almost all ovarian carcinomas (p < 0.05). Promoter hypomethylation was found in (17 of 29) 58.62% ovarian cancers and (4 of 16) 25% normal ovaries (p < 0.05). The HOXA10 expression is higher when the status of HOXA10 gene promoter is hypomethylated than in methylated tissues (p < 0.05). After 5-aza-dC treatment, the expression level of HOXA10 mRNA transcript was increased in the two cell lines.
Conclusion
Our results indicate that promoter hypomethylation is an important mechanism for high expression of HOXA10 in human ovarian cancer and may be a potential prognostic factor in ovarian cancer.




Similar content being viewed by others
References
Balch C, Huang TH, Brown R, Nephew KP (2004) The epigenitics of ovarian cancer drug resistance and resensitization. Am J Obstet Gynecol 191:1552–1572
Cheng W, Liu J, Yoshida H, Rosen D, Naora H (2005) Lineage infidelity of epithelial ovarian cancers is controlled by HOX genes that specify regional identity in thec reproductive tract. Nat Med 11:531–537
Daffary GS, Troy PJ, Bagot CN, Young SL, Taylor HS (2002) Direct regulation of β3-integrin subunit gene expression by HOXA10 in endometrial cells. Mol Endocrinol 16(3):571–579
Das PM, Singal R (2004) DNA methylation and cancer. J Clin Oncol 22:4632–4642
Ehrlich M, Woods CB, Yu MC, Dubeau L, Yang F, Campan M, Weisenberger DJ, Long T, Youn B, Fiala ES, Laird PW (2006) Quantitative analysis of associations between DNA hypermethylations, hypomethylation, and DNMT RNA levels in ovarian tumors. Oncogene 25:2636–2645
Feinberg AP, Tycko B (2004) The history of cancer epigenetics. Nat Rev Cancer 4:143–153
Feinberg AP, Vogelstein B (1983) Hypomethylation distinguishes genes of some human cancers from their normal counterparts. Nature 301:89–92
Fraga MF, Agrelo R, Esteller M (2007) Cross-talk between aging and cancer: the epigenetic language. Ann NY Acad Sci 1100:60–74
Herman JG, Graff JR, Myohanen S, Nelkin BD, Baylin SB (1996) Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci USA 93:9821–9826
Issa JA (2000) CpG-island methylation in aging and cancer. Curr Top Microbiol Immunol 249:101–118
Jemal A, Siegel R, Ward E et al (2009) Cancer statistics, 2009. CA Cancer J Clin 59(4):225–249
Jones PA, Baylin SB (2002) The fundamental role of epigenetic events in cancer. Nat Rev Genet 3:415–428
Kelly M, Daftary G, Taylor HS (2006) An autoregulatory element maintains HOXA10 expression in endometrial epithelial cells. Am J Obstet Gynecol 194:1100–1109
Lechner JF, Fugaro JM, Wong Y, Pass HI, Harris CC, Belinsky SA (2001) Perspective: cell differentiation theory may advance early detection of and therapy for lung cancer. Radiat Res 155:235–238
Miller GJ, Miller HL, van Bokhoven A, Lambert JR, Werahera PN, Schirripa O, Lucia MS, Nordeen SK (2003) Aberrant HOXC expression accompanies the malignant phenotype in human prostate. Cancer Res 63:5879–5888
Momparler RL (2003) Cancer epigenetics. Oncogene 22:6479–6483
Ones PA, Takai D (2001) The role of DNA methylation in mammalian epigenetics. Science 293:1068–1070
Pakneshan P, Szyf M, Farias-Eisner R, Rabbani SA (2004) Reversal of the hypomethylation status of urokinase(uPA) promoter blocks breast cancer growth and metastasis. J Biol Chem 279:31735–31744
Pattamadilok J, Huapai N, Rattanatanyong P, Vasurattana A, Triratanachat S, Tresukosol D, Mutirangura A (2008) LINE-1 hypomethylation level as potential prognostic factor for epithelial ovarian cancer. Int J Gynecol Cancer 18:711–717
Shen JC, Richeout WM 3rd, Jones PA (1992) High frequency mutagenesis by a DNA methyltransferase. Cell 71:1073–1080
Shi H, Wang MX, Caldwell CW (2007) CpG islands: their potential as biomarker for cancer. Expert Rev Mol Diagn 7(5):519–531
Swanton C, Nicke B, Downward J (2004) RNA interference, DNA methylation, and gene silencing: a bright future for cancer therapy? Lancet Oncol 5:653–654
Szyf M, Pakneshan P, Rabbani SA (2004) DNA demethylation and cancer: therapeutic implications. Cancer Lett 211:133–143
Vider BZ, Zimber A, Hirsch D, Estlein D, Chastre E, Prevot S, Gespach C, Yaniv A, Gazit A (1997) Human colorectal carcinogenesis is associated with deregulation of homeobox gene expression. Biochem Biophys Res Commun 232(3):742–748
Vider BZ, Zimber A, Chastre E, Gespach C, Halperin M, Mashiah P, Yaniv A, Gazit A (2000) Deregulated expression of homeobox-cotaining genes, HOXB6, B8, C8, C9, and Cdx-1, in human colon cancer cell lines. Biochem Biophys Res Commun 272:513–518
Wang C, Zhang B, Shen H (2004) Expression of HOXA9 mRNA and its clinical significance in primary hepatocarcinoma. China J Mod Med 1:14
Acknowledgments
We would like to thank Dr. Xin Lu of Fudan University, Shanghai, China, for the human ovarian cancer cell lines of SKOV3 and HEY for help in cell experiments. This work was supported in part by the National Natural Science Foundation of China (30772332); Jiangsu Province Health Department Program Grant (H200617).
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cheng, W., Jiang, Y., Liu, C. et al. Identification of aberrant promoter hypomethylation of HOXA10 in ovarian cancer. J Cancer Res Clin Oncol 136, 1221–1227 (2010). https://doi.org/10.1007/s00432-010-0772-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-010-0772-4