Abstract
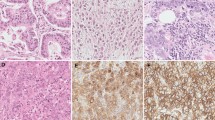
Pancreatic cancer remains one of the deadliest malignancies in the world. Inflammatory response and tumor environment are thought to play a major role in its pathogenesis. Knowledge on TLR expression and impact on patient survival in pancreatic cancer is limited. The study’s aim was to clarify the role of different TLRs in pancreatic cancer. TLR2, TLR4, and TLR9 expression was investigated in 65 surgically resected pancreatic ductal adenocarcinoma specimens by immunohistochemistry. The association between TLR expression, clinical parameters, and local inflammatory response to the tumor was assessed using chi-square test. Relation between patient survival and TLR expression was calculated with multivariable Cox regression, adjusted for age, sex, and tumor stage. We found TLR2, TLR4, and TLR9 to be expressed in pancreatic cancer. There was no association between TLR expression and tumor stage, tumor size, lymph node metastasis, or tumor necrosis. Contrary to our initial hypothesis, high cytoplasmic TLR9 expression was associated with longer patient survival, and multivariate analysis identified low TLR9 expression as an independent risk factor for cancer-specific death (HR 3.090, 95% CI 1.673–5.706). The results suggest that high TLR9 expression in pancreatic ductal adenocarcinoma indicates improved prognosis. The prognostic effect of TLR9 might be associated with bacterial exposure, but this needs further evidence.



Similar content being viewed by others
References
Li D, Xie K, Wolff R, Abbruzzese JL (2004) Pancreatic cancer. Lancet 363:1049–1057
Falasca M, Kim M, Casari I (2016) Pancreatic cancer: current research and future directions. Biochim. Biophys. Acta
Silvestris N, Longo V, Cellini F, Reni M, Bittoni A, Cataldo I, Partelli S, Falconi M, Scarpa A, Brunetti O, Lorusso V, Santini D, Morganti A, Valentini V, Cascinu S (2016) Neoadjuvant multimodal treatment of pancreatic ductal adenocarcinoma. Crit Rev OncolHematol 98:309–324
Takeda K and Akira S (2007) Toll-like receptors. Curr. Protoc. Immunol; Chapter 14:Unit 14.12
Cook DN, Pisetsky DS, Schwartz DA (2004) Toll-like receptors in the pathogenesis of human disease. Nat Immunol 5:975–979
Botos I, Segal DM, Davies DR (2011) The structural biology of toll-like receptors. Structure 19:447–459
Sabroe I, Read RC, Whyte MK, Dockrell DH, Vogel SN, Dower SK (2003) Toll-like receptors in health and disease: complex questions remain. J Immunol 171:1630–1635
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140:805–820
Pandey S, Singh S, Anang V, Bhatt AN, Natarajan K, Dwarakanath BS (2015) Pattern recognition receptors in cancer progression and metastasis. Cancer Growth Metastasis 8:25–34
Castano-Rodriguez N, Kaakoush NO, Mitchell HM (2014) Pattern-recognition receptors and gastric cancer. Front Immunol 5:336
Kauppila JH, Selander KS (2014) Toll-like receptors in esophageal cancer. Front Immunol 5:200
Cammarota R, Bertolini V, Pennesi G, Bucci EO, Gottardi O, Garlanda C, Laghi L, Barberis MC, Sessa F, Noonan DM, Albini A (2010) The tumor microenvironment of colorectal cancer: stromal TLR-4 expression as a potential prognostic marker. J Transl Med 8:112 5876-8-112
Kauppila JH, Mattila AE, Karttunen TJ, Salo T (2013) Toll-like receptor 5 (TLR5) expression is a novel predictive marker for recurrence and survival in squamous cell carcinoma of the tongue. Br J Cancer 108:638–643
Gonzalez-Reyes S, Fernandez JM, Gonzalez LO, Aguirre A, Suarez A, Gonzalez JM, Escaff S, Vizoso FJ (2011) Study of TLR3, TLR4, and TLR9 in prostate carcinomas and their association with biochemical recurrence. Cancer Immunol Immunother 60:217–226
Vaz J, Akbarshahi H, Andersson R (2013) Controversial role of toll-like receptors in acute pancreatitis. World J Gastroenterol 19:616–630
Huynh AS, Chung WJ, Cho HI, Moberg VE, Celis E, Morse DL, Vagner J (2012) Novel toll-like receptor 2 ligands for targeted pancreatic cancer imaging and immunotherapy. J Med Chem 55:9751–9762
Fan P, Zhang JJ, Wang B, Wu HQ, Zhou SX, Wang CY, Zhang JH, Tian Y, Wu HS (2012) Hypoxia-inducible factor-1 up-regulates the expression of toll-like receptor 4 in pancreatic cancer cells under hypoxic conditions. Pancreatology 12:170–178
Zhang JJ, Wu HS, Wang L, Tian Y, Zhang JH, Wu HL (2010) Expression and significance of TLR4 and HIF-1alpha in pancreatic ductal adenocarcinoma. World J Gastroenterol 16:2881–2888
Ochi A, Nguyen AH, Bedrosian AS, Mushlin HM, Zarbakhsh S, Barilla R, Zambirinis CP, Fallon NC, Rehman A, Pylayeva-Gupta Y, Badar S, Hajdu CH, Frey AB, Bar-Sagi D, Miller G (2012) MyD88 inhibition amplifies dendritic cell capacity to promote pancreatic carcinogenesis via Th2 cells. J Exp Med 209:1671–1687
Zambirinis CP, Levie E, Nguy S, Avanzi A, Barilla R, Xu Y, Seifert L, Daley D, Greco SH, Deutsch M, Jonnadula S, Torres-Hernandez A, Tippens D, Pushalkar S, Eisenthal A, Saxena D, Ahn J, Hajdu C, Engle DD, Tuveson D, Miller G (2015) TLR9 ligation in pancreatic stellate cells promotes tumorigenesis. J Exp Med 212:2077–2094
Wu HQ, Wang B, Zhu SK, Tian Y, Zhang JH, Wu HS (2011) Effects of CPG ODN on biological behavior of PANC-1 and expression of TLR9 in pancreatic cancer. World J Gastroenterol 17:996–1003
Huhta H, Helminen O, Kauppila JH, Salo T, Porvari K, Saarnio J, Lehenkari PP, Karttunen TJ (2016) The expression of toll-like receptors in normal human and murine gastrointestinal organs and the effect of microbiome and cancer. J Histochem Cytochem 64:470–482
Michaud DS, Izard J (2014) Microbiota, oral microbiome, and pancreatic cancer. Cancer J 20:203–206
Wang C, Li J (2015) Pathogenic microorganisms and pancreatic cancer. Gastrointest Tumors 2:41–47
Huhta H, Helminen O, Kauppila JH, Takala H, Metsikko K, Lehenkari P, Saarnio J, Karttunen T (2015) Toll-like receptor 9 expression in the natural history of Barrett mucosa. Virchows Arch 467:9–18
Helminen O, Huhta H, Takala H, Lehenkari PP, Saarnio J, Kauppila JH, Karttunen TJ (2014) Increased toll-like receptor 5 expression indicates esophageal columnar dysplasia. Virchows Arch 464:11–18
Laurila JJ, Ala-Kokko TI, Laurila PA, Saarnio J, Koivukangas V, Syrjala H, Karttunen TJ (2005) Histopathology of acute acalculous cholecystitis in critically ill patients. Histopathology 47:485–492
Morse DL, Balagurunathan Y, Hostetter G, Trissal M, Tafreshi NK, Burke N, Lloyd M, Enkemann S, Coppola D, Hruby VJ, Gillies RJ, Han H (2010) Identification of novel pancreatic adenocarcinoma cell-surface targets by gene expression profiling and tissue microarray. Biochem Pharmacol 80:748–754
Vaisanen MR, Jukkola-Vuorinen A, Vuopala KS, Selander KS, Vaarala MH (2013) Expression of toll-like receptor-9 is associated with poor progression-free survival in prostate cancer. Oncol Lett 5:1659–1663
Kauppila JH, Korvala J, Siirila K, Manni M, Makinen LK, Hagstrom J, Atula T, Haglund C, Selander KS, Saarnio J, Karttunen TJ, Lehenkari PP, Salo T (2015) Toll-like receptor 9 mediates invasion and predicts prognosis in squamous cell carcinoma of the mobile tongue. J Oral Pathol Med 44:571–577
Kauppila JH, Takala H, Selander KS, Lehenkari PP, Saarnio J, Karttunen TJ (2011) Increased toll-like receptor 9 expression indicates adverse prognosis in oesophageal adenocarcinoma. Histopathology 59:643–649
Korvala J, Harjula T, Siirila K, Almangush A, Aro K, Makitie AA, Grenman R, Karttunen TJ, Leivo I, Kauppila JH, Salo T (2014) Toll-like receptor 9 expression in mucoepidermoid salivary gland carcinoma may associate with good prognosis. J. Oral Pathol. Med. 43:530–537
Ronkainen H, Hirvikoski P, Kauppila S, Vuopala KS, Paavonen TK, Selander KS, Vaarala MH (2011) Absent toll-like receptor-9 expression predicts poor prognosis in renal cell carcinoma. J Exp Clin Cancer Res 30:84 9966-30-84
Tuomela J, Sandholm J, Karihtala P, Ilvesaro J, Vuopala KS, Kauppila JH, Kauppila S, Chen D, Pressey C, Harkonen P, Harris KW, Graves D, Auvinen PK, Soini Y, Jukkola-Vuorinen A, Selander KS (2012) Low TLR9 expression defines an aggressive subtype of triple-negative breast cancer. Breast Cancer Res Treat 135:481–493
Zhang Y, Wang Q, Ma A, Li Y, Li R, Wang Y (2014) Functional expression of TLR9 in esophageal cancer. Oncol Rep 31:2298–2304
Kauppila JH, Karttunen TJ, Saarnio J, Nyberg P, Salo T, Graves DE, Lehenkari PP, Selander KS (2013) Short DNA sequences and bacterial DNA induce esophageal, gastric, and colorectal cancer cell invasion. APMIS 121:511–522
Jacobs C, Duewell P, Heckelsmiller K, Wei J, Bauernfeind F, Ellermeier J, Kisser U, Bauer CA, Dauer M, Eigler A, Maraskovsky E, Endres S, Schnurr M (2011) An ISCOM vaccine combined with a TLR9 agonist breaks immune evasion mediated by regulatory T cells in an orthotopic model of pancreatic carcinoma. Int J Cancer 128:897–907
Silva A, Mount A, Krstevska K, Pejoski D, Hardy MP, Owczarek C, Scotney P, Maraskovsky E, Baz MA (2015) The combination of ISCOMATRIX adjuvant and TLR agonists induces regression of established solid tumors in vivo. J Immunol 194:2199–2207
Pratesi G, Petrangolini G, Tortoreto M, Addis A, Belluco S, Rossini A, Selleri S, Rumio C, Menard S, Balsari A (2005) Therapeutic synergism of gemcitabine and CpG-oligodeoxynucleotides in an orthotopic human pancreatic carcinoma xenograft. Cancer Res 65:6388–6393
Fan X, Alekseyenko AV, Wu J, Peters BA, Jacobs EJ, Gapstur SM, Purdue MP, Abnet CC, Stolzenberg-Solomon R, Miller G, Ravel J, Hayes RB, Ahn J (2016) Human oral microbiome and prospective risk for pancreatic cancer: a population-based nested case-control study. Gut
Michaud DS, Izard J, Wilhelm-Benartzi CS, You DH, Grote VA, Tjonneland A, Dahm CC, Overvad K, Jenab M, Fedirko V, Boutron-Ruault MC, Clavel-Chapelon F, Racine A, Kaaks R, Boeing H, Foerster J, Trichopoulou A, Lagiou P, Trichopoulos D, Sacerdote C, Sieri S, Palli D, Tumino R, Panico S, Siersema PD, Peeters PH, Lund E, Barricarte A, Huerta JM, Molina-Montes E, Dorronsoro M, Quiros JR, Duell EJ, Ye W, Sund M, Lindkvist B, Johansen D, Khaw KT, Wareham N, Travis RC, Vineis P, Bueno-de-Mesquita HB, Riboli E (2013) Plasma antibodies to oral bacteria and risk of pancreatic cancer in a large European prospective cohort study. Gut 62:1764–1770
Zambirinis CP, Pushalkar S, Saxena D, Miller G (2014) Pancreatic cancer, inflammation, and microbiome. Cancer J 20:195–202
Yamamura K, Baba Y, Nakagawa S, Mima K, Miyake K, Nakamura K, Sawayama H, Kinoshita K, Ishimoto T, Iwatsuki M, Sakamoto Y, Yamashita Y, Yoshida N, Watanabe M, Baba H (2016) Human microbiome Fusobacterium nucleatum in esophageal cancer tissue is associated with prognosis. Clin. Cancer Res
Mima K, Nishihara R, Qian ZR, Cao Y, Sukawa Y, Nowak JA, Yang J, Dou R, Masugi Y, Song M, Kostic AD, Giannakis M, Bullman S, Milner DA, Baba H, Giovannucci EL, Garraway LA, Freeman GJ, Dranoff G, Garrett WS, Huttenhower C, Meyerson M, Meyerhardt JA, Chan AT, Fuchs CS, Ogino S (2015) Fusobacterium nucleatum in colorectal carcinoma tissue and patient prognosis. Gut
Mitsuhashi K, Nosho K, Sukawa Y, Matsunaga Y, Ito M, Kurihara H, Kanno S, Igarashi H, Naito T, Adachi Y, Tachibana M, Tanuma T, Maguchi H, Shinohara T, Hasegawa T, Imamura M, Kimura Y, Hirata K, Maruyama R, Suzuki H, Imai K, Yamamoto H, Shinomura Y (2015) Association of Fusobacterium species in pancreatic cancer tissues with molecular features and prognosis. Oncotarget 6:7209–7220
Otte JM, Cario E, Podolsky DK (2004) Mechanisms of cross hyporesponsiveness to toll-like receptor bacterial ligands in intestinal epithelial cells. Gastroenterology 126:1054–1070
Vaz J, Andersson R (2014) Intervention on toll-like receptors in pancreatic cancer. World J Gastroenterol 20:5808–5817
Acknowledgements
We thank Erja Tomperi and Riitta Vuento for expertise in preparation of the immunohistochemical stainings.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by grants from the Finnish Cultural Foundation (J. L), Finnish Medical Foundation (J. L), Sigrid Jusélius Foundation (J.H.K.), Mary and Georg C. Ehrnroot Foundation (J.H.K.), Thelma Mäkikyrö Foundation (J.H.K.), Orion Research Foundation (J.H.K.), and Medical Research Center Oulu Focus Group Funding (T.J.K.).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Leppänen, J., Helminen, O., Huhta, H. et al. High toll-like receptor (TLR) 9 expression is associated with better prognosis in surgically treated pancreatic cancer patients. Virchows Arch 470, 401–410 (2017). https://doi.org/10.1007/s00428-017-2087-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-017-2087-1