Abstract
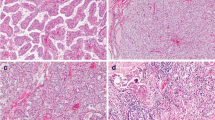
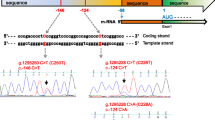
The prognostic value of BRAF V600E and TERT promoter mutation in papillary thyroid carcinoma (PTC) is controversial. We examined alterations in BRAF V600E and TERT promoter by PCR-direct sequencing in PTC of 144 Japanese patients. Alternative lengthening of telomeres was examined as another mechanism of telomere maintenance by immunohistochemical staining for ATRX and DAXX. Of the clinicopathological characteristics, regional lymph node metastasis, extra-thyroid extension, multifocality/intrathyroidal spread, and advanced stage (III/V) were associated with shorter disease-free survival rate (DFSR). TERT promoter mutation was found in eight patients (6 %), and this was significantly associated with total thyroidectomy, multifocality/intrathyroidal spread, lymph node metastasis and advanced stage. The BRAF V600E mutation was found in 53 patients (38.2 %) but was not associated with any clinicopathological factors. TERT mutations were not correlated with BRAF V600E mutation status. TERT mutation-positive tumors (TERT+) showed lower DFSR than BRAF V600E-mutation-positive tumors (BRAF V600E +), and TERT+/BRAF V600E+ tumors showed lower DFSR than BRAF V600E+ tumors. No cases showed loss of ATRX/DAXX expression by immunohistochemistry. TERT promoter mutations showed a lower prevalence in our series and appeared to be associated with aggressive behavior. In PTCs, telomerase activation by TERT promoter mutation might be more important than alternative lengthening of telomeres.



Similar content being viewed by others
References
Nixon IJ, Ganly I, Patel SG, Palmer FL, Whitcher MM, Ghossein R, Michael Tuttle R, Shaha AR, Shah JP (2012) Changing trends in well differentiated thyroid carcinoma over eight decades. Int J Surg 10:618–623
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, Mazzaferri EL, McIver B, Pacini F, Schlumberger M, Sherman SI, Steward DL, Tuttle RM (2009) Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association (ATA) guidelines taskforce on thyroid nodules and differentiated thyroid cancer. Thyroid 19.11:1167–1214
Ito Y, Kudo T, Kihara M, Takamura Y, Kobayashi K, Miya A, Miyauchi A (2012) Improvement of lymph node recurrence rate, but not distant recurrence and carcinoma death rates, in patients with papillary thyroid carcinoma after disease-free survival for 5 years. Endocr J 59:895–901
Durante C, Montesano T, Torlontano M, Attard M, Monzani F, Tumino S, Costante G, Meringolo D, Bruno R, Trulli F, Massa M, Maniglia A, D'Apollo R, Giacomelli L, Ronga G, Filetti S, PTC Study Group (2013) Papillary thyroid cancer: time course of recurrences during postsurgery surveillance. J Clin Endocrinol Metab 98:636–642
Lang BH, Tang AH, Wong KP, Shek TW, Wan KY, Lo CY (2012) Significance of size of lymph node metastasis on postsurgical stimulated thyroglobulin levels after prophylactic unilateral central neck dissection in papillary thyroid carcinoma. Ann Surg Oncol 19:3472–3478
Ahn D, Park JS, Sohn JH, Kim JH, Park SK, Seo AN, JY P (2012) BRAF V600E mutation does not serve as a prognostic factor in Korean patients with papillary thyroid carcinoma. Auris Nasus Larynx 39(2):198–203
Lupi C, Giannini R, Ugolini C, Proietti A, Berti P, Minuto M, Materazzi G, Elisei R, Santoro M, Miccoli P, Basolo F (2007) Association of BRAF V600E mutation with poor clinicopathological outcomes in 500 consecutive cases of papillary thyroid carcinoma. J Clin Endocrinol Metab 92.11:4085–4090
Kebebew E, Weng J, Bauer J, Ranvier G, Clark OH, Duh QY, Shibru D, Bastian B, Griffin A (2007) The prevalence and prognostic value of BRAF mutation in thyroid cancer. Ann Surg 246:466–470
Lin KL, Wang OC, Zhang XH, Dai XX, XQ H, JM Q (2010) The BRAF mutation is predictive of aggressive clinicopathological characteristics in papillary thyroid microcarcinoma. Ann Surg Oncol 17:3294–3300
Howell GM, Nikiforova MN, Carty SE, Armstrong MJ, Hodak SP, Stang MT, McCoy KL, Nikiforov YE, Yip L (2013) BRAF V600E mutation independently predicts central compartment lymph node metastasis in patients with papillary thyroid cancer. Ann Surg Oncol 20.1:47–52
Elisei R, Viola D, Torregrossa L, Giannini R, Romei C, Ugolini C, Molinaro E, Agate L, Biagini A, Lupi C, Valerio L, Materazzi G, Miccoli P, Piaggi P, Pinchera A, Vitti P, Basolo F (2012) The BRAF V600E mutation is an independent, poor prognostic factor for the outcome of patients with low-risk intrathyroid papillary thyroid carcinoma: single-institution results from a large cohort study. J Clin Endocrinol Metab 97:4390–4398
Xing M, Alzahrani AS, Carson KA, Viola D, Elisei R, Bendlova B, Yip L, Mian C, Vianello F, Tuttle RM, Robenshtok E, Fagin JA, Puxeddu E, Fugazzola L, Czarniecka A, Jarzab B, O'Neill CJ, Sywak MS, Lam AK, Riesco-Eizaguirre G, Santisteban P, Nakayama H, Tufano RP, Pai SI, Zeiger MA, Westra WH, Clark DP, Clifton-Bligh R, Sidransky D, Ladenson PW, Sykorova V (2013) Association between BRAF V600E mutation and mortality in patients with papillary thyroid cancer. JAMA 309.14:1493–1501
Xing M, Westra WH, Tufano RP, Cohen Y, Rosenbaum E, Rhoden KJ, Carson KA, Vasko V, Larin A, Tallini G, Tolaney S, Holt EH, Hui P, Umbricht CB, Basaria S, Ewertz M, Tufaro AP, Califano JA, Ringel MD, Zeiger MA, Sidransky D, Ladenson PWBRAF (2005) Mutation predicts a poorer clinical prognosis for papillary thyroid cancer. J Clin Endocrinol Metab 90:6373–6379
Elisei R, Ugolini C, Viola D, Lupi C, Biagini A, Giannini R, Romei C, Miccoli P, Pinchera A (2008) Basolo F. BRAFV600E mutation and outcome of patients with papillary thyroid carcinoma: a 15-year median follow-up study. J Clin Endocrinol Metab 93:3943–3949
Fugazzola L, Mannavola D, Cirello V, Vannucchi G, Muzza M, Vicentini L, Beck-Peccoz PBRAF (2004) Mutations in an Italian cohort of thyroid cancers. Clin Endocrinol 61:239–243
Fugazzola L, Puxeddu E, Avenia N, Romei C, Cirello V, Cavaliere A, Faviana P, Mannavola D, Moretti S, Rossi S, Sculli M, Bottici V, Beck-Peccoz P, Pacini F, Pinchera A, Santeusanio F, Elisei R (2006) Correlation between BRAFV600E mutation and clinico–pathologic parameters in papillary thyroid carcinoma: data from a multicentric Italian study and review of the literature. Endocr Relat Cancer. 13.2:455–464
Brzeziańska E, Pastuszak-Lewandoska D, Wojciechowska K, Migdalska-Sek M, Cyniak-Magierska A, Nawrot E, Lewiński A (2007) Investigation of V600E BRAF mutation in papillary thyroid carcinoma in the polish population. Neuro Endocrinol Lett 28:351–359
Henke LE, Pfeifer JD, Ma C, Perkins SM, DeWees T, El-Mofty S, Moley JF, Nussenbaum B, Haughey BH, Baranski TJ, Schwarz JK, Grigsby PWBRAF (2015) Mutation is not predictive of long-term outcome in papillary thyroid carcinoma. Cancer Med 4.6:791–799
Takami H, Ito Y, Okamoto T, Yoshida A (2011) Therapeutic strategy for differentiated thyroid carcinoma in Japan based on a newly established guideline managed by Japanese Society of Thyroid Surgeons and Japanese Association of Endocrine Surgeons. World J Surg 35.1:111–121
Günes C, Rudolph KL (2013) The role of telomeres in stem cells and cancer. Cell 152.3:390–393
Horn S, Figl A, Rachakonda PS, Fischer C, Sucker A, Gast A, Kadel S, Moll I, Nagore E, Hemminki K, Schadendorf D, Kumar RTERT (2013) Promoter mutations in familial and sporadic melanoma. Science 339.6122:959–961
Huang FW, Hodis E, MJ X, Kryukov GV, Chin L, Garraway LA (2013) Highly recurrent TERT promoter mutations in human melanoma. Science 339.6122:957–959
Landa I, Ganly I, Chan TA, Mitsutake N, Matsuse M, Ibrahimpasic T, Ghossein RA, Fagin JA (2013) Frequent somatic TERT promoter mutations in thyroid cancer: higher prevalence in advanced forms of the disease. J Clin Endocrinol Metab 98.9:E1562–E1566
Vinagre J, Almeida A, Pópulo H, Batista R, Lyra J, Pinto V, Coelho R, Celestino R, Prazeres H, Lima L, Melo M, da Rocha AG, Preto A, Castro P, Castro L, Pardal F, Lopes JM, Santos LL, Reis RM, Cameselle-Teijeiro J, Sobrinho-Simões M, Lima J, Máximo V, Soares P (2013) Frequency of TERT promoter mutations in human cancers. Nat Commun 4:2185
Liu T, Wang N, Cao J, Sofiadis A, Dinets A, Zedenius J, Larsson C, Xu D (2014) The age-and shorter telomere-dependent TERT promoter mutation in follicular thyroid cell-derived carcinomas. Oncogene 33.42:4978–4984
Bryan TM, Englezou A, Gupta J, Bacchetti S, Reddel RR (1995) Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J 14(17):4240–4248
Perrem K, Bryan TM, Englezou A, Hackl T, Moy EL, Reddel RR (1999) Repression of an alternative mechanism for lengthening of telomeres in somatic cell hybrids. Oncogene 1822:3383–3390
Lovejoy CA, Li W, Reisenweber S, Thongthip S, Bruno J, de Lange T, De S, Petrini JH, Sung PA, Jasin M, Rosenbluh J, Zwang Y, Weir BA, Hatton C, Ivanova E, Macconaill L, Hanna M, Hahn WC, Lue NF, Reddel RR, Jiao Y, Kinzler K, Vogelstein B, Papadopoulos N, Meeker AK, Starr Cancer ALT (2012) Consortium. Loss of ATRX, genome instability, and an altered DNA damage response are hallmarks of the alternative lengthening of telomeres pathway. PLoS Genet 8:e1002772
Cheung NK, Zhang J, Lu C, Parker M, Bahrami A, Tickoo SK, Heguy A, Pappo AS, Federico S, Dalton J, Cheung IY, Ding L, Fulton R, Wang J, Chen X, Becksfort J, Wu J, Billups CA, Ellison D, Mardis ER, Wilson RK, Downing JR, Dyer MA (2012) St Jude Children’s Research Hospital–Washington University Pediatric Cancer Genome Project. Association of Age at Diagnosis and Genetic Mutations in Patients with Neuroblastoma. JAMA 307:1062–1071
Chen X, Bahrami A, Pappo A, Easton J, Dalton J, Hedlund E, Ellison D, Shurtleff S, Wu G, Wei L, Parker M, Rusch M, Nagahawatte P, Wu J, Mao S, Boggs K, Mulder H, Yergeau D, Lu C, Ding L, Edmonson M, Qu C, Wang J, Li Y, Navid F, Daw NC, Mardis ER, Wilson RK, Downing JR, Zhang J, Dyer MA (2014) St. Jude Children’s Research Hospital–Washington University Pediatric Cancer Genome Project. Recurrent somatic structural variations contribute to tumorigenesis in pediatric osteosarcoma. Cell Rep 7:104–112
Drané P, Ouararhni K, Depaux A, Shuaib M, Hamiche A (2010) The death-associated protein DAXX is a novel histone chaperone involved in the replicationindependent deposition of H3.3. Genes Dev 24:1253–1265
Goldberg AD, Banaszynski LA, Noh KM, Lewis PW, Elsaesser SJ, Stadler S, Dewell S, Law M, Guo X, Li X, Wen D, Chapgier A, DeKelver RC, Miller JC, Lee YL, Boydston EA, Holmes MC, Gregory PD, Greally JM, Rafii S, Yang C, Scambler PJ, Garrick D, Gibbons RJ, Higgs DR, Cristea IM, Urnov FD, Zheng D, Allis CD (2010) Distinct factors control histone variant H3.3 localization at specific genomic regions. Cell 140:678–691
Wong LH, McGhie JD, Sim M, Anderson MA, Ahn S, Hannan RD, George AJ, Morgan KA, Mann JR, Choo KHATRX (2010) Interacts with H3.3 in maintaining telomere structural integrity in pluripotent embryonic stem cells. Genome Res 20:351–360
LiVolsi VA, Mazzaferri EL, Albores-Saavedra J (2004) Papillary carcinoma. In: RA DL, RV L, PU H, et al. (eds) WHO Classification of Tumours. Pathology and genetics of tumours of endocrine organs. IARC Press, Lyon, France, pp. 57–66
Sobin LH, Wittekind C (2002) UICC: TNM classfication of malignant tumors, 6th edn. Wiley-Liss, New York
Koelsche C, Sahm F, Capper D, Reuss D, Sturm D, Jones DT, Kool M, Northcott PA, Wiestler B, Böhmer K, Meyer J, Mawrin C, Hartmann C, Mittelbronn M, Platten M, Brokinkel B, Seiz M, Herold-Mende C, Unterberg A, Schittenhelm J, Weller M, Pfister S, Wick W, Korshunov A, von Deimling A (2013) Distribution of TERT promoter mutations in pediatric and adult tumors of the nervous system. Acta Neuropathol 126:907–915
Imamhasan A, Mitomi H, Saito T, Arakawa A, Yao T (2012) Clear cell variant of squamous cell carcinoma originating in the esophagus: report of a case with immunohistochemical and oncogenetic analyses. Pathol Int 62:137–143
Liu X, Wu G, Shan Y, Hartmann C, von Deimling A, Xing M (2013) Highly prevalent TERT promoter mutations in bladder cancer and glioblastoma. Cell Cycle 12:1637–1638
Killela PJ, Reitman ZJ, Jiao Y, Bettegowda C, Agrawal N, LA D Jr, Friedman AH, Friedman H, Gallia GL, Giovanella BC, Grollman AP, He TC, He Y, Hruban RH, Jallo GI, Mandahl N, Meeker AK, Mertens F, Netto GJ, Rasheed BA, Riggins GJ, Rosenquist TA, Schiffman M, IeM S, Theodorescu D, MS T, VE V, TL W, Wentzensen N, LD W, Zhang M, RE ML, DD B, KW K, Vogelstein B, Papadopoulos N, Yan H (2013) TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc Natl Acad Sci U S A 110.15:6021–6026
Xing M, Liu R, Liu X, Murugan AK, Zhu G, Zeiger MA, Pai S, Bishop J (2014) BRAF V600E and TERT promoter mutations cooperatively identify the most aggressive papillary thyroid cancer with highest recurrence. J Clin Oncol 32.25:2718–2726
Liu X, Bishop J, Shan Y, Pai S, Liu D, Murugan AK, Sun H, El-Naggar AK, Xing M (2013) Highly prevalent TERT promoter mutations in aggressive thyroid cancers. Endocr Relat Cancer 20.4:603–610
Vinagre J, Pinto V, Celestino R, Reis M, Pópulo H, Boaventura P, Melo M, Catarino T, Lima J, Lopes JM, Máximo V, Sobrinho-Simões M, Soares P (2014) Telomerase promoter mutations in cancer: an emerging molecular biomarker? Virchows Arch 465.2:119–133
Liu X, Qu S, Liu R, Sheng C, Shi X, Zhu G, Murugan AK, Guan H, Yu H, Wang Y, Sun H, Shan Z, Teng W, Xing MTERT (2014) Promoter mutations and their association with BRAF V600E mutation and aggressive clinicopathological characteristics of thyroid cancer. J Clin Endocrinol Metab 99.6:1130–1136
Melo M, da Rocha AG, Vinagre J, Batista R, Peixoto J, Tavares C, Celestino R, Almeida A, Salgado C, Eloy C, Castro P, Prazeres H, Lima J, Amaro T, Lobo C, Martins MJ, Moura M, Cavaco B, Leite V, Cameselle-Teijeiro JM, Carrilho F, Carvalheiro M, Máximo V, Sobrinho-Simões M, Soares PTERT (2014) Promoter mutations are a major indicator of poor outcome in differentiated thyroid carcinomas. J Clin Endocrinol Metab 99:E754–E765
Heaphy CM, de Wilde RF, Jiao Y, Klein AP, Edil BH, Shi C, Bettegowda C, Rodriguez FJ, Eberhart CG, Hebbar S, Offerhaus GJ, McLendon R, Rasheed BA, He Y, Yan H, Bigner DD, Oba-Shinjo SM, Marie SK, Riggins GJ, Kinzler KW, Vogelstein B, Hruban RH, Maitra A, Papadopoulos N, Meeker AK (2011) Altered telomeres in tumors with ATRX and DAXX mutations. Science 333:425
Jiao Y, Shi C, Edil BH, de Wilde RF, Klimstra DS, Maitra A, Schulick RD, Tang LH, Wolfgang CL, Choti MA, Velculescu VE, LA D Jr, Vogelstein B, Kinzler KW, Hruban RH, Papadopoulos NDAXX (2011) ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science 331:1199–1203
Liau JY, Tsai JH, Jeng YM, Lee JC, Hsu HH, Yang CY (2015) Leiomyosarcoma with alternative lengthening of telomeres is associated with aggressive histologic features, loss of ATRX expression, and poor clinical outcome. Am J Surg Pathol 39:236–244
Heaphy CM, Subhawong AP, Hong SM, Goggins MG, Montgomery EA, Gabrielson E, GJ N, JI E, TL L, WH W, IeM S, CA I-D, Maitra A, QK L, CG E, JM T, Rakheja D, RJ K, TC W, RB R, Argani P, AM DM, Terracciano L, Torbenson M, AK M (2011) Prevalence of the alternative lengthening of telomeres telomere maintenance mechanism in human cancer subtypes. Am J Pathol 179:1608–1615
Fukushima T, Suzuki S, Mashiko M, Ohtake T, Endo Y, Takebayashi Y, Sekikawa K, Hagiwara K, Takenoshita SBRAF (2003) Mutations in papillary carcinomas of the thyroid. Oncogene 22:6455–6457
Namba H, Nakashima M, Hayashi T, Hayashida N, Maeda S, Rogounovitch TI, Ohtsuru A, Saenko VA, Kanematsu T, Yamashita S (2003) Clinical implication of hot spot BRAF mutation, V599E, in papillary thyroid cancers. J Clin Endocrinol Metab 88:4393–4397
Ito Y, Yoshida H, Maruo R, Morita S, Takano T, Hirokawa M, Yabuta T, Fukushima M, Inoue H, Tomoda C, Kihara M, Uruno T, Higashiyama T, Takamura Y, Miya A, Kobayashi K, Matsuzuka F, Miyauchi A (2009) BRAF mutation in papillary thyroid carcinoma in a Japanese population: its lack of correlation with high-risk clinicopathological features and disease-free survival of patients. Endocr J 56(1):89–97
Macerola E, Loggini B, Giannini R, Garavello G, Giordano M, Proietti A, Niccoli C, Basolo F, Fontanini G (2015) Coexistence of TERT promoter and BRAF mutations in cutaneous melanoma is associated with more clinicopathological features of aggressiveness. Virchows Arch 467:177–184
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Ethical approval
This article does not contain any studies involving human participants and/or animals performed by any of the authors. This study was approved by the research ethics committee of our institution (2014034).
Rights and permissions
About this article
Cite this article
Nasirden, A., Saito, T., Fukumura, Y. et al. In Japanese patients with papillary thyroid carcinoma, TERT promoter mutation is associated with poor prognosis, in contrast to BRAF V600E mutation. Virchows Arch 469, 687–696 (2016). https://doi.org/10.1007/s00428-016-2027-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-016-2027-5