Abstract
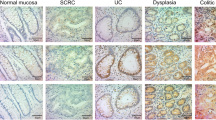
Inflammatory bowel diseases (IBDs; both ulcerative colitis [UC] and Crohn’s colitis [CC]) are well-established predisposing pathological conditions for colorectal cancer (CRC) development. In IBDs, both the endoscopy and the histology assessment of CRC precursors (i.e., dysplasia, also defined as intraepithelial neoplasia) are associated with low interobserver consistency, and no reliable dysplasia-specific biomarker is available. The programmed cell death 4 (PDCD4) tumor suppressor gene is involved in sporadic colorectal oncogenesis, but scanty information is available on its involvement in IBD-associated colorectal oncogenesis. One hundred twenty tissue samples representative of active and inactive IBD and of flat dysplasia were obtained from 30 cases of UC and 30 of CC who undergone colectomy. Twenty additional biopsy samples obtained from patients with irritable bowel syndrome acted as normal controls. PDCD4 expression was assessed by immunohistochemistry; the expression of miR-21 (a major PDCD4 regulator) was investigated by quantitative real-time PCR and in situ hybridization in different series of a hundred samples. Tissue specimens from both controls and inactive IBD consistently featured strong PDCD4 nuclear immunostain; conversely, lower PDCD4 nuclear expression was featured by both active IBD and IBD-associated dysplastic lesions. Significant PDCD4 down-regulation distinguished IBD-associated dysplasia (p < 0.001) versus active IBD. In both active IBD and dysplasia, PDCD4 down-regulation was significantly associated with miR-21 up-regulation. PDCD4 nuclear down-regulation (which parallels miR-21 up-regulation) is involved in the molecular pathway of IBD-associated carcinogenesis. PDCD4 nuclear expression may be usefully applied as ancillary maker in the histological assessment of IBD-associated dysplastic lesions.



Similar content being viewed by others
Abbreviations
- IHC:
-
Immunohistochemistry
- IBD:
-
Inflammatory bowel disease
- PDCD4:
-
Programmed cell death 4
- UC:
-
Ulcerative colitis
- CC:
-
Crohn’s colitis
References
Fiocchi C (1998) Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology 115:182–205
Crohn BB (1925) The sigmoidoscopic picture of chronic ulcerative colitis (non-specific). Am J Med Sci 170:220–228
Munkholm P (2003) The incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther 18(S2):1–5
Feagins LA, Souza RF, Spechler SJ (2009) Carcinogenesis in IBD: potential targets for the prevention of colorectal cancer. Nat Rev Gastroenterol Hepatol 6(5):297–305
Goel GA, Kandiel A, Achkar JP et al (2011) Molecular pathways underlying IBD-associated colorectal neoplasia: therapeutic implications. Am J Gastroenterol 106(4):719–730
Hamilton SR, Bosman FT, Boffetta P et al (2010) Carcinoma of the colon and rectum. In: Bosman FT, Carneiro F, Hruban R, Theise ND (eds) WHO classification of tumours of the digestive system, 4th edn. International Agency for Research on Cancer, Lyon, France, pp 134–182
Neumann H, Vieth M, Langner C et al (2011) Cancer risk in IBD: how to diagnose and how to manage DALM and ALM. World J Gastroenterol 17(27):3184–3191
Eaden J, Abrams K, McKay H et al (2001) Inter-observer variation between general and specialist gastrointestinal pathologists when grading dysplasia in ulcerative colitis. J Pathol 194(2):152–157
Eaden J (2004) Colorectal carcinoma and inflammatory bowel disease. Aliment Pharmacol Ther 20(S4):24–30
Bohm M, Sawicka K, Siebrasse JP et al (2003) The transformation suppressor protein Pdcd4 shuttles between nucleus and cytoplasm and binds RNA. Oncogene 22:4905–4910
Yang HS, Cho MH, Zakowicz H et al (2004) A novel function of the MA-3 domains in transformation and translation suppressor Pdcd4 is essential for its binding to eukaryotic translation initiation factor 4A. Mol Cell Biol 24:3894–3906
Afonja O, Juste D, Das S et al (2004) Induction of PDCD4 tumor suppressor gene expression by RAR agonists, antiestrogen and HER-2/neu antagonist in breast cancer cells. Evidence for a role in apoptosis. Oncogene 23:8135–8145
Bitomsky N, Bohm M, Klempnauer KH (2004) Transformation suppressor protein Pdcd4 interferes with JNK-mediated phosphorylation of c-Jun and recruitment of the coactivator p300 by c-Jun. Oncogene 23:7484–7493
Zakowicz H, Yang HS, Stark C et al (2005) Mutational analysis of the DEAD-box RNA helicase eIF4AII characterizes its interaction with transformation suppressor Pdcd4 and eIF4GI. RNA 11:261–274
Jansen AP, Camalier CE, Colburn NH (2005) Epidermal expression of the translation inhibitor programmed cell death 4 suppresses tumorigenesis. Cancer Res 65:6034–6041
Dorrello NV, Peschiaroli A, Guardavaccaro D et al (2006) S6K1-and betaTRCP-mediated degradation of PDCD4 promotes protein translation and cell growth. Science 314:467–471
Bitomsky N, Wethkamp N, Marikkannu R et al (2008) SiRNA-mediated knockdown of Pdcd4 expression causes upregulation of p21(Waf1/Cip1) expression. Oncogene 27:4820–4829
Carayol N, Katsoulidis E, Sassano A et al (2008) Suppression of programmed cell death 4 (PDCD4) protein expression by BCRABL-regulated engagement of the mTOR/p70 S6 kinase pathway. J Biol Chem 283:8601–8610
Mudduluru G, Medved F, Grobholz R et al (2007) Loss of programmed cell death 4 expression marks adenoma–carcinoma transition, correlates inversely with phosphorylated protein kinase B, and is an independent prognostic factor in resected colorectal cancer. Cancer 110:1697–1707
Allgayer H (2010) Pdcd4, a colon cancer prognostic that is regulated by a microRNA. Crit Rev Oncol Hematol 73:185–191
Fassan M, Pizzi M, Battaglia G et al (2010) Programmed cell death 4 (PDCD4) expression during multistep Barrett’s carcinogenesis. J Clin Pathol 63:692–696
Chen Y, Knosel T, Kristiansen G et al (2003) Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. J Pathol 200:640–646
Zhang H, Ozaki I, Mizuta T et al (2006) Involvement of programmed cell death 4 in transforming growth factor-beta1-induced apoptosis in human hepatocellular carcinoma. Oncogene 25:6101–6112
Wang Q, Sun Z, Yang HS (2008) Downregulation of tumor suppressor Pdcd4 promotes invasion and activates both betacatenin/Tcf and AP-1-dependent transcription in colon carcinoma cells. Oncogene 27:1527–1535
Baffa R, Fassan M, Volinia S et al (2009) MicroRNA expression profiling of human metastatic cancers identifies cancer gene targets. J Pathol 219:214–221
Yamamichi N, Shimomura R, Inada K et al (2009) Locked nucleic acid in situ hybridization analysis of miR-21 expression during colorectal cancer development. Clin Cancer Res 15:4009–4016
Fassan M, Cagol M, Pennelli G et al (2010) Programmed cell death 4 (PDCD4) protein in esophageal cancer. Oncol Rep 24:135–139
Goke R, Barth P, Schmidt A et al (2004) Programmed cell death protein 4 suppresses CDK1/cdc2 via induction of p21 (Waf1/Cip1). Am J Physiol Cell Physiol 287:C1541–C1546
Dalal SR, Kwon JH (2010) The role of microRNA in inflammatory bowel disease. Gastroenterol Hepatol (NY) 6:714–722
Wu F, Zhang S, Dassopoulos T, Harris ML et al (2010) Identification of microRNAs associated with ileal and colonic Crohn’s disease. Inflamm Bowel Dis 16:1729–1738
Okubo M, Tahara T, Shibita T et al (2011) Association study of common genetic variants in pre-microRNAs in patients with ulcerative colitis. J Clin Immunol 31:69–73
Goldman H (1994) Interpretation of large intestinal mucosal biopsy specimens. Hum Pathol 25(11):1150–1159
Cornaggia M, Leutner M, Mescoli C et al (2011) Chronic idiopathic inflammatory bowel diseases: the histology report. Dig Liver Dis 43(4):293–303
Rugge M, Correa P, Dixon MF (2000) Padova classification gastric dysplasia: the Padova international classification. Am J Surg Pathol 24(2):167–176, Review
Rugge M, Fassan M, Clemente R et al (2008) Bronchopulmonary carcinoid: phenotype and long-term outcome in a single institution series of Italian patients. Clin Cancer Res 14:149–154
Yoshinaga H, Matsuhashi S, Fujiyama C et al (1999) Novel human PDCD4 (H731) gene expressed in proliferative cells is expressed in the small duct epithelial cells of the breast as revealed by an anti-H731 antibody. Pathol Int 49:1067–1077
Fassan M, Volinia S, Palatini J et al (2011) MicroRNA expression profiling in human Barrett’s carcinogenesis. Int J Cancer 129(7):1661–1670
Fassan M, Pizzi M, Giacomelli L et al (2011) PDCD4 nuclear loss inversely correlates with miR-21 levels in colon carcinogenesis. Virchows Arch 458(4):413–419
Chang KH, Miller N, Kheirelseid EA et al (2011) MicroRNA-21 and PDCD4 expression in colorectal cancer. Eur J Surg Oncol 37(7):597–603
Yu Y, Kanwar SS, Patel BB et al (2012) MicroRNA-21 induces stemness by downregulating transforming growth factor beta receptor 2 (TGFβR2) in colon cancer cells. Carcinogenesis 33(1):68–76
Itzkowitz SH (2006) Molecular biology of dysplasia and cancer in inflammatory bowel disease. Gastroenterol Clin North Am 35(3):553–571
Gerrits MM, Chen M, Theeuwes M et al (2011) Biomarker-based prediction of inflammatory bowel disease-related colorectal cancer: a case–control study. Cell Oncol (Dordr) 34(2):107–117
Hiyoshi Y, Kamohara H, Karashima R et al (2009) Micro-RNA-21 regulates the proliferation and invasion in esophageal squamous cell carcinoma. Clin Cancer Res 15(6):1915–1922
Gerrits MM, Chen M, Theeuwes M et al (2011) Biomarker-based prediction of inflammatory bowel disease-related colorectal cancer: a case–control study. Cell Oncol 34:107–117
Rutter M, Saunders B, Wilkinson K et al (2004) Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology 126:451–459
Brest P, Lapaquette P, Souidi M et al (2011) A synonymous variant in IRGM alters a binding site for miR-196 and causes deregulation of IRGM-dependent xenophagy in Crohn's disease. Nat Genet 43(3):242–245
Acknowledgments
We wish to acknowledge the continuous support of the “G. Berlucchi” and the “G.B. Morgagni” Foundations. This work was partially supported by an AIRC Regional grant (2008). We also thank Dr. Cristiano Lanza, Dr. Vincenza Guzzardo, and Dr. Vanni Lazzarin for their excellent technical support. All authors of this research paper participated directly in the planning and execution of the study and in the analysis of the results.
Conflict of interest
The authors have no competing interests to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Kathrin Ludwig and Matteo Fassan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ludwig, K., Fassan, M., Mescoli, C. et al. PDCD4/miR-21 dysregulation in inflammatory bowel disease-associated carcinogenesis. Virchows Arch 462, 57–63 (2013). https://doi.org/10.1007/s00428-012-1345-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-012-1345-5