Abstract
Main conclusion
Roots of plants with high artemisinin-producing leaves increased leaf production of artemisinin in low-producing plants and vice versa indicating roots are involved in controlling artemisinin biosynthesis in shoots.
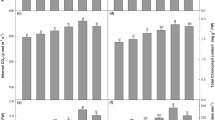
The anti-malarial sesquiterpene, artemisinin, is produced and stored in glandular trichomes (GLTs) of Artemisia annua. Evidence suggested roots, which produce no significant artemisinin nor precursor compounds, regulate production of artemisinin biosynthesis in the leaves. Using grafting, we studied the relationship between rootstock and scion by measuring GLTs and five artemisinic metabolites (artemisinin, deoxyartemisinin, dihydroartemisinic acid, artemisinic acid, arteannuin B) in scions of ungrafted, self-grafted, and cross-grafted plants among three cultivars: S and 15 both having GLTs with artemisinin at 1.49 and 0.57 %, respectively, and G producing neither GLTs nor detectable artemisinin. All artemisinin-producing self-grafts, e.g., S/S (scion/rootstock) and 15/15, produced more artemisinin than ungrafted plants, likely from grafting stress. S/S grafts also produced more GLTs. The 15/S grafts produced more artemisinin than S/15, suggesting rootstocks from high producing S plants stimulated artemisinin production in 15 scions. S/15 grafts yielded less artemisinin than S/S, but more than either 15/15 or ungrafted n15 and nS; S/15 grafts also had a lower density of GLTs than S/S, suggesting rootstock inhibition of the scion. The S rootstock induced trace artemisinin production in G scions, but did not induce GLT formation in G/S grafts. Different grafts exhibited different trichome morphologies and effects on artemisinic pathway flux. This study provides new information regarding the role of roots in GLT development and artemisinin production in this important medicinal plant.







Similar content being viewed by others
Abbreviations
- AA:
-
Artemisinic acid
- AAA:
-
Artemisinic aldehyde
- AACT:
-
Acetoacetyl-CoA thiolase
- AAOH:
-
Artemisinic alcohol
- AB:
-
Arteannuin B
- ADS:
-
Amorpha-4, 11-diene synthase
- ALDH1:
-
Aldehyde dehydrogenase 1
- AN:
-
Artemisinin
- CYP:
-
CYP71AV1, cytochrome P450-dependent hydroxylase
- DBR2:
-
Double bond reductase 2
- DHAA:
-
Dihydroartemisinic acid
- DHAAA:
-
Dihydroartemisinic aldehyde
- DHOH:
-
Dihydroartemisinic alcohol
- dAN:
-
Deoxyartemisinin
- FLV:
-
Flavonoid
- FPP:
-
Farnesyl pyrophosphate
- G:
-
Glandless artemisinin null mutant of A. annua
- GLT:
-
Glandular trichome
- RED1:
-
Dihydroartemisinic aldehyde reductase 1
- ROS:
-
Reactive oxygen species
- ShAM:
-
Shoot apical meristem
- S:
-
SAM cultivar of A. annua
- 15:
-
#15 cultivar of A. annua
References
Arsenault PR, Vail D, Wobbe KK, Erickson K, Weathers PJ (2010) Reproductive development modulates gene expression and metabolite levels with possible feedback inhibition of artemisinin in Artemisia annua L. Plant Physiol 154:958–968
Arvouet-Grand A, Vennat B, Pourrat A, Legret P (1994) Standardization of a propolis extract and identification of the main constituents. J Pharm Belgi 49(6):462–468
Banyai W, Mii M, Supaibulwatana K (2011) Enhancement of artemisinin content and biomass in Artemisia annua by exogenous GA3 treatment. Plant Growth Regul 63(1):45–54
Bouwmeester HJ, Wallaart TE, Janssen MH, van Loo B, Jansen BJ, Posthumus MA, Schmidt CO, De Kraker J-W, König WA, Franssen MC (1999) Amorpha-4, 11-diene synthase catalyses the first probable step in artemisinin biosynthesis. Phytochemistry 52(5):843–854
Brown GD (2010) The biosynthesis of artemisinin (Qinghaosu) and the phytochemistry of Artemisia annua L. (Qinghao). Molecules 15(11):7603–7698
Brown GD, Sy L-K (2004) In vivo transformations of dihydroartemisinic acid in Artemisia annua plants. Tetrahedron 60(5):1139–1159
Brown GD, Sy L-K (2007) In vivo transformations of artemisinic acid in Artemisia annua plants. Tetrahedron 63(38):9548–9566
Corbesier L, Vincent C, Jang S, Fornara F, Fan Q, Searle I, Giakountis A, Farrona S, Gissot L, Turnbull C (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316(5827):1030–1033
Covello PS (2008) Making artemisinin. Phytochemistry 69(17):2881–2885
Duke MV, Paul RN, Elsohly HN, Sturtz G, Duke SO (1994) Localization of artemisinin and artemisitene in foliar tissues of glanded and glandless biotypes of Artemisia annua L. Int J Plant Sci 155(3):365–372
Efferth T (2009) Artemisinin: a versatile weapon from traditional Chinese medicine. In: Ramawat KG (ed) Herbal drugs: Ethnomedicine to modern medicine. Springer, Berlin Heidelberg, pp 173–194
Elford BC, Roberts MF, Phillipson JD, Wilson RJ (1987) Potentiation of the antimalarial activity of qinghaosu by methoxylated flavones. Trans Roy Soc Trop Med H 81(3):434–436
Ferreira JF, Janick J (1995) Floral morphology of Artemisia annua with special reference to trichomes. Int J Plant Sci 807–815
Ferreira JF, Janick J (1996) Roots as an enhancing factor for the production of artemisinin in shoot cultures of Artemisia annua. Plant Cell Tiss Org 44(3):211–217
Ferreira JFS, Luthria DL, Sasaki T, Heyerick A (2010) Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 15(5):3135–3170
Firestone GL, Sundar SN (2009) Anticancer activities of artemisinin and its bioactive derivatives. Expert Rev Mol Medicine 11:e32
Ganesh D, Fuehrer HP, Starzengrüber P, Swoboda P, Khan WA, Reismann JA, Mueller MS, Chiba P, Noedl H (2012) Antiplasmodial activity of flavonol quercetin and its analogues in Plasmodium falciparum: evidence from clinical isolates in Bangladesh and standardized parasite clones. Parasitol Res 110(6):2289–2295
Graham IA, Besser K, Blumer S, Branigan CA, Czechowski T, Elias L, Guterman I, Harvey D, Isaac PG, Khan AM, Larson TR, Li Y, Pawson T, Penfield T, Rae AM, Rathbone DA, Reid S, Ross J, Smallwood MF, Segura V, Townsend T, Vyas D, Winzer T, Bowles D (2010) The genetic map of artemisia annua L. identifies loci affecting yield of the antimalarial drug artemisinin. Science 327(5963):328–331
Jessing KK, Cedergreen N, Mayer P, Libous-Bailey L, Strobel BW, Rimando A, Duke SO (2013) Loss of artemisinin produced by Artemisia annua L. to the soil environment. Ind Crop Prod 43:132–140
Joo JH, Bae YS, Lee JS (2001) Role of auxin-induced reactive oxygen species in root gravitropism. Plant Physiol 126(3):1055–1060
Kapoor R, Chaudhary V, Bhatnagar A (2007) Effects of arbuscular mycorrhiza and phosphorus application on artemisinin concentration in Artemisia annua L. Mycorrhiza 17(7):581–587
Kehr J (2012) Long-distance signaling by small RNAs. In: Kragler F, Hülskamp M (eds) Short and long distance signaling. Springer, New York, pp 131–149
Klayman DL (1985) Qinghaosu (artemisinin): an antimalarial drug from China. Science 228(4703):1049–1055
Lai HC, Singh NP, Sasaki T (2013) Development of artemisinin compounds for cancer treatment. Invest New Drug 31(1):230–246
Lehane AM, Saliba KJ (2008) Common dietary flavonoids inhibit the growth of the intraerythrocytic malaria parasite. BMC Res Notes 1(1):26
Liu KC-SC, Yang SL, Roberts M, Elford B, Phillipson J (1992) Antimalarial activity of Artemisia annua flavonoids from whole plants and cell cultures. Plant Cell Rep 11(12):637–640
Maes L, Van Nieuwerburgh FC, Zhang Y, Reed DW, Pollier J, Vande Casteele SR, Inzé D, Covello PS, Deforce DL, Goossens A (2011) Dissection of the phytohormonal regulation of trichome formation and biosynthesis of the antimalarial compound artemisinin in Artemisia annua plants. New Phytol 189(1):176–189
Mandal S, Upadhyay S, Wajid S, Ram M, Jain DC, Singh VP, Abdin MZ, Kapoor R (2014) Arbuscular mycorrhiza increase artemisinin accumulation in Artemisia annua by higher expression of key biosynthesis genes via enhanced jasmonic acid levels. Mycorrhiza. doi:10.1007/s00572-014-0614-3
Mannan A, Liu C, Arsenault PR, Towler MJ, Vail DR, Lorence A, Weathers PJ (2010) DMSO triggers the generation of ROS leading to an increase in artemisinin and dihydroartemisinic acid in Artemisia annua shoot cultures. Plant Cell Rep 29(2):143–152
Nguyen KT, Arsenault PR, Weathers PJ (2011) Trichomes + roots + ROS = artemisinin: regulating artemisinin biosynthesis in Artemisia annua L. Vitro Cell Dev Biol-Plant 47(3):329–338
Nguyen KT, Towler MJ, Weathers PJ (2013) The effect of roots and media constituents on trichomes and artemisinin production in Artemisia annua L. Plant Cell Rep 32(2):207–218
Olsson ME, Olofsson LM, Lindahl A-L, Lundgren A, Brodelius M, Brodelius PE (2009) Localization of enzymes of artemisinin biosynthesis to the apical cells of glandular secretory trichomes of Artemisia annua L. Phytochemistry 70(9):1123–1128
Omid A, Keilin T, Glass A, Leshkowitz D, Wolf S (2007) Characterization of phloem-sap transcription profile in melon plants. J Exp Bot 58(13):3645–3656
Rydén AM, Ruyter-Spira C, Quax WJ, Osada H, Muranaka T, Kayser O, Bouwmeester H (2010) The molecular cloning of dihydroartemisinic aldehyde reductase and its implication in artemisinin biosynthesis in Artemisia annua. Planta Med 76(15):1778
Schramek N, Wang H, Römisch-Margl W, Keil B, Radykewicz T, Winzenhörlein B, Beerhues L, Bacher A, Rohdich F, Gershenzon J (2010) Artemisinin biosynthesis in growing plants of Artemisia annua. A 13CO2 study. Phytochemistry 71(2):179–187
Shen Q, Yan T, Fu X, Tang K (2016) Transcriptional regulation of artemisinin biosynthesis in Artemisia annua L. Science Bulletin 61(1):18–25
Soetaert SS, Van Neste CM, Vandewoestyne ML, Head SR, Goossens A, Van Nieuwerburgh FC, Deforce DL (2013) Differential transcriptome analysis of glandular and filamentous trichomes in Artemisia annua. BMC Plant Biol 13(1):220
Spiegelman Z, Golan G, Wolf S (2013) Don’t kill the messenger: long-distance trafficking of mRNA molecules. Plant Sci 213:1–8
Suberu J, Song L, Slade S, Sullivan N, Barker G, Lapkin AA (2013) A rapid method for the determination of artemisinin and its biosynthetic precursors in Artemisia annua L. crude extracts. J Pharmaceut Biomed 84:269–277
Tellez MR, Canel C, Rimando AM, Duke SO (1999) Differential accumulation of isoprenoids in glanded and glandless Artemisia annua L. Phytochemistry 52(6):1035–1040
Teoh KH, Polichuk DR, Reed DW, Nowak G, Covello PS (2006) Artemisia annua L. (Asteraceae) trichome-specific cDNAs reveal CYP71AV1, a cytochrome P450 with a key role in the biosynthesis of the antimalarial sesquiterpene lactone artemisinin. FEBS Lett 580(5):1411–1416
Teoh KH, Polichuk DR, Reed DW, Covello PS (2009) Molecular cloning of an aldehyde dehydrogenase implicated in artemisinin biosynthesis in Artemisia annua. Botany 87(6):635–642
Ting HM, Wang B, Rydén AM, Woittiez L, Herpen T, Verstappen FW, Ruyter-Spira C, Beekwilder J, Bouwmeester HJ, Krol A (2013) The metabolite chemotype of Nicotiana benthamiana transiently expressing artemisinin biosynthetic pathway genes is a function of CYP71AV1 type and relative gene dosage. New Phytol 199(2):352–366
Towler MJ, Weathers PJ (2007) Evidence of artemisinin production from IPP stemming from both the mevalonate and the nonmevalonate pathways. Plant Cell Rep 26(12):2129–2136
Towler MJ, Weathers PJ (2015) Variations in key artemisinic and other metabolites throughout plant development in Artemisia annua L. for potential therapeutic use. Ind Crop Prod 67:185–191
Wallaart TE, Bouwmeester HJ, Hille J, Poppinga L, Maijers NC (2001) Amorpha-4, 11-diene synthase: cloning and functional expression of a key enzyme in the biosynthetic pathway of the novel antimalarial drug artemisinin. Planta 212(3):460–465
Wang W, Wang Y, Zhang Q, Qi Y, Guo D (2009) Global characterization of Artemisia annua glandular trichome transcriptome using 454 pyrosequencing. BMC Genom 10(1):1
Wang Y, Chen S, Yu O (2011) Metabolic engineering of flavonoids in plants and microorganisms. Appl Microbiol Biotechnol 91(4):949–956
Weathers PJ, Towler MJ (2012) The flavonoids casticin and artemetin are poorly extracted and are unstable in an Artemisia annua tea infusion. Planta Med 78(10):1024–1026
Weathers P, Reed K, Hassanali A, Lutgen P, Engeu PO (2014) Whole plant approaches to therapeutic use of Artemisia annua L. (Asteraceae). In: Aftab T, Ferreira JFS, Khan MMA, Naeem M (eds) Artemisia annua-Pharmacology and biotechnology. Springer, Berlin Heidelberg, pp 51–74
WHO (2015) World malaria report 2015. Geneva: WHO, 2015. http://www.who.int/malaria/publications/world-malaria-report-2015/report/en/
Woerdenbag HJ, Lüers JF, van Uden W, Pras N, Malingré TM, Alfermann AW (1993) Production of the new antimalarial drug artemisinin in shoot cultures of Artemisia annua L. Plant Cell Tiss Org 32(2):247–257
Yang K, Monafared RS, Wang H, Lundgren A, Brodelius PE (2015) The activity of the artemisinic aldehyde Δ11 (13) reductase promoter is important for artemisinin yield in different chemotypes of Artemisia annua L. Plant Mol Biol 88(4–5):325–340
Zhang Y, Teoh KH, Reed DW, Maes L, Goossens A, Olson DJ, Ross AR, Covello PS (2008) The molecular cloning of artemisinic aldehyde Δ11 (13) reductase and its role in glandular trichome-dependent biosynthesis of artemisinin in Artemisia annua. J Biol Chem 283(31):21501–21508
Zhang Y, Nowak G, Reed DW, Covello PS (2011) The production of artemisinin precursors in tobacco. Plant Biotechnol J 9(4):445–454
Acknowledgments
The authors are grateful for the partial financial support of WPI and the assistance of Dr. Abdul Mannan (COMSATS, Pakistan), and Andy Butler, Hailey Cambra, Dr. Liwen Fei, and Professors Kristen Wobbe and Luis Vidali of WPI for suggestions and technical help on this project.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, S., Towler, M.J. & Weathers, P.J. Root regulation of artemisinin production in Artemisia annua: trichome and metabolite evidence. Planta 244, 999–1010 (2016). https://doi.org/10.1007/s00425-016-2560-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-016-2560-0