Abstract
Main conclusion
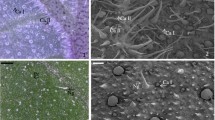
The leaves of Withania somnifera contained four morphologically distinct trichome types: glandular capitate, non-glandular dendritic (branched), non-glandular bicellular and non-glandular multicellular trichomes. Major phytochemical compounds present within glandular and non-glandular trichomes were alkaloids and phenolic compounds.
The aim of this study was to characterize the micromorphology of the foliar trichomes of Withania somnifera as well as to elucidate the location and composition of the secretory products. Trichome density and length was also determined in three developmental stages of the leaves. Light microscopy and scanning electron microscopy showed the presence of four morphologically distinct trichome types: glandular capitate, non-glandular dendritic, non-glandular bicellular and non-glandular multicellular. The dendritic trichomes exhibited cuticular warts which are involved in the “Lotus-Effect”. Glandular capitate and non-glandular dendritic trichomes were aggregated on the mid-vein of young and mature leaves, possibly to protect underlying vasculature. Histochemical staining also revealed the presence of two major classes of phytochemical compounds that are of medicinal importance, i.e. alkaloids and phenolic compounds. These compounds are used to treat a wide variety of ailments and also act as chemical deterrents in plants. The results of this study explain possible roles of four morphologically distinct trichome types based on their morphology, foliar distribution and content.













Similar content being viewed by others
References
Adedeji O, Ajuwon OY, Babawale OO (2007) Foliar epidermal studies, organographic distribution and taxonomic importance of trichomes in the family Solanaceae. Int J Bot 3:276–282. doi:10.3923/ijb.2007.276.282
Arnott HJ, Webb MA (2000) Twinned raphides of calcium oxalate in grapes (Vitis): implications for crystal stability and function. Int J Plant Sci 161:133–142 (pmid: 10648203)
Ascensão L, Pais MSS (1987) Glandular trichomes of Artemisia campestris (ssp. Maritima): ontogeny and histochemistry of the secretory product. Bot Gazet 148:221–227. doi:10.1086/337650
Atal CK, Schwarting AE (1961) Ashwagandha: an ancient Indian drug. Econ Bot 15:256–263. doi:10.1007/BF02862166
Barthlott W, Neinhuis C (1997) Purity of the sacred Lotus, or escape from contamination in biological surfaces. Planta 202:1–8. doi:10.1007/s004250050096
Bhatt A, Naidoo Y, Nicolas A (2010) The foliar trichomes of Hypoestes aristata (Vahl) Sol. Ex Roem. & Schult var aristata (Acanthaceae) a widespread medicinal plant species in tropical sub-Saharan Africa: which comments on its possible phylogenetic significance. Biol Res 43:403–409 (pmid: 21526266)
Boix YF, Victório CP, Defaveri ACA, Arruda RDCDO, Sato A, Lage CLS (2011) Glandular trichomes of Rosmarinus officinalis L.: anatomical and phytochemical analysis of leaf volatiles. Plant Biosyst 145:848–856 (agid: 344995)
Cain AJ (1947) The use of Nile blue in the examination of lipids. Q J Microsc Sci 88:383–392
Chatterjee S, Srivastava S, Khalid A, Singh N, Sangwan RS, Sidhu OP, Ray R, Khetrapal CL, Tuli R (2010) Comprehensive metabolic fingerprinting of Withania somnifera leaf and root extracts. Phytochemistry 71:1085–1094. doi:10.1016/j.phytochem.2010.04.001
Chaurasiya ND, Gupta VK, Sangwan RS (2007) Leaf ontogenic phase-related dynamics of withaferin A and withanone biogenesis in Ashwagandha (Withania somnifera Dunal)—an important medicinal herb. J Plant Biol 50:508–513. doi:10.1007/BF03030691
Cosgrove DJ (2000) Loosening of plant cell walls by expansins. Nature 407:321–326. doi:10.1038/35030000
Dai X, Wang G, Yang DS, Tang Y, Broun P, Marks MD, Summer LW, Dixon RA, Zhao PX (2010) Trichome: a comparative omics database for plant trichomes. Plant Physiol 152:44–54. doi:10.1104/pp.109.145813
Debnath M, Malik CP, Bisen PS (2006) Micropropagation: a tool for the production of high quality plant-based medicines. Cur Pharmaceut Biotech 7:33–49 (doi: 0.2174/138920106775789638)
Duke SO (1994) Glandular trichomes—a focal point of chemical and structural interactions. Int J Plant Sci 155:617–620
Fahn A (1988) Secretory tissues in vascular plants. New Phytol 108:229–257. doi:10.1111/j.1469-8137.1988.tb04159.x
Fahn A (2000) Structure and function of secretory cells. Adv Bot Res 31:37–75. doi:10.1016/S0065-2296(00)31006-0
Fordyce JA, Agrawal AA (2001) The role of plant trichomes and caterpillar group size on growth and defence of the pipevine swallowtail Battus philenor. J Anim Ecol 70:997–1005. doi:10.1046/j.0021-8790.2001.00568.x
Franceschi VR, Horner HT Jr (1980) Calcium oxalate crystals in plants. Bot Rev 46:361–429
Franceschi VR, Nakata PA (2005) Calcium oxalate in plants: formation and function. Ann Rev Plant Biol 56:41–71. doi:10.1146/annurev.arplant.56.032604.144106
Furr M, Mahlberg PG (1981) Histochemical analyses of laticifers and glandular trichomes in Cannabis sativa. J Nat Prod 44:153–159. doi:10.1021/np50014a002
Gairola S, Naidoo Y, Bhatt A, Nicholas A (2008) An investigation of the foliar trichomes of Tetradenia riparia (Hochst.) Codd [Lamiaceae]: an important medicinal plant of Southern Africa. Flora 204:325–330. doi:10.1016/j.flora.2008.04.002
Glas JJ, Schimmel BCJ, Alba JM, Escobar-Bravo R, Scuurink RC, Kant MR (2012) Plant glandular trichomes as targets for breeding or engineering of resistance to herbivores. Int J Mol Sci. doi:10.3390/ijms131217077
Grassmann J, Hippeli S, Elstner EF (2002) Plant’s defence and its benefit for animals and medicine: role of phenolics and terpenoids in avoiding oxygen stress. Plant Physiol Biochem 40:471–478. doi:10.1016/S0981-9428(02)01395-5
Hameed I, Hussain F (2011) Stomatal studies of some selected medicinal plants of family Solanaceae. J Med Plant Res 5:4525–4529
Harisha CR, Switu J (2013) Pharmacognostical study on trichomes of Solanaceae and its significance. Univers J Pharm 2:100–104
Joy PP, Thomas J, Mathew S, Skaria BP (1998) Medicinal plants. Aromatic and Med Plants Res Station. Kerala Agricultural University. p 211
Khan B, Ahmad SF, Bani S, Kaul A, Suri KA, Satti NK, Athar M, Qai GN (2006) Augmentation and proliferation of T lymphocytes and Th-1 cytokines by Withania somnifera in stressed mice. Int Immunopharmacol 6:1394–1403. doi:10.1016/j.intimp.2006.04.001
Kim HJ, Han J, Kim S, Lee HR, Shin J, Kim J, Cho J, Kim YH, Lee HJ, Kim B, Choi D (2011) Trichome density of main stem is tightly linked to PepMoV resistance in chili pepper (Capsicum annuum L.). Int J Plant Breed Res 122:1051–1058. doi:10.1007/s00122-010-1510-7
Köhler RH, Cao J, Zipfel WR, Webb WW, Hanson MR (1997) Exchange of protein molecules through connections between higher plant plastids. Science 276:2039–2042. doi:10.1126/science.276.5321.2039
Kumar S, Kumar V (2011) Evaluation of Withania somnifera L. (Dunal). (Solanaceae) leaf and root extracts as an antimicrobial agent-highly medicinal plants in India. Asian J Exp Biol Sci 2:155–157
Kumar MS, Aslam A, Kumar DV, Ramachandran A, Shajahan A (2010a) Comparative studies on leaf-epidermal features of W. somnifera and W. obtusifolia—highly medicinal species of India. Adv Biotechnol 10:3
Kumar MS, Kumar DV, Ramachandran A, Shajahan A (2010b) Anatomical studies in Withania somnifera L. (Dunal)—an important medicinal plant. Adv Biotechnol 10:3
Lange BM, Wildung MR, Stauber EJ, Sanchez C, Pouchnik D, Croteau R (2000) Probig essential oil biosynthesis and secretion by functional evaluation of expressed sequence tags from mint glandular trichomes. Proc Nat Acad Sci 97:2934–2939. doi:10.1073/pnas.97.6.2934
Levin DA (1973) The role of trichomes in plant defense. Q Rev Biol 48:3–15
Mahesh B, Satish S (2008) Antimicrobial activity of some important medicinal plant against plant and human pathogens. World J Agric Sci 4:839–843
Marin M, Ascensão L, Lakusie B (2012) Trichomes of Satureja horvatii Silic (Lamiaceae): micromorphology and histochemistry. Arch Biol Sci Bel 64:995–1000. doi:10.1080/0972060X.2014.901604
Mirrett S (1982) Acridine orange stain. Inf Control 3:250–252
Negro C, Tomasi L, Miceli A (2003) Phenolic compounds and antioxidant activity from red grape marc extracts. Bioresour Tech 87:41–44. doi:10.1016/S0960-8524(02)00202-X
Pearse AGE (1968) Histochemistry: theoretical and applied 3rd edn vol 1. Churchill Livingstone, London, pp. 1–759
Peiffer M, Tooker JF, Luthe DS, Felton GW (2009) Plants on early alert: glandular trichomes as sensors for insect herbivores. New Phytol 184:644–656. doi:10.1111/j.1469-8137.2009.03002.x
Pickard WF (2008) Laticifers and secretory ducts: two other tube systems in plants. New Phytol 177:877–888. doi:10.1111/j.1469-8137.2007.02323.x
Pyke KA, Howells CA (2002) Plastid and stromule morphogenesis in tomato. Ann Bot 90:559–566. doi:10.1093/aob/mcf235
Rahman A, Shahwar D, Naz A, Choudhary MI (2003) Withanolides from Withania coagulans. Phytochemistry 63:387–390. doi:10.5923/j.als.20120201.02
Ram H, Kumar A, Sharma SK, Ojha A, Rao SR (2012) Meiotic studies in Withania somnifera (L) Dunal.: a threatened medicinal herb of Indian Thar Desert. Am J Plant Sci 3:185–189. doi:10.13140/2.1.2308.6729
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212. doi:10.1083/jcb.17.1.208
Robinson T (1974) Metabolism and function of alkaloids in plants. Am Assoc Adv Sci 184:430–435. doi:10.1126/science.184.4135.430
Rusydi A, Talip N, Latip J, Rahman RA, Sharif I (2013) Morphology of trichomes in Pogostemon cablin Benth. (Lamiaceae). Austral J Crop Sci 7:744–749
Sangwan RS, Chaurasiya ND, Lal P, Misra L, Tuli R, Sangwan NS (2008) Withanolide A is inherently de novo biosynthesised in roots of the medicinal plant Ashwagandha (Withania somnifera). Physiol Plant 133:278–287. doi:10.1111/j.1399-3054.2008.01076.x
Scartezzini P, Speron E (2000) Review on some plants of Indian traditional medicine with antioxidant activity. J Ethnopharmacol 71:23–43. doi:10.1016/S0378-8741(00)00213-0
Senthil K, Wasnik NG, Kim Y, Yanj D (2009) Generation and analysis of expressed sequence tags from leaf and root of Withania somnifera (Ashwagandha). Mol Biol Rep 37:893–902. doi:10.1007/s11033-009-9696-y
Singh BK, Gahoi R, Sonkar A (2010) A quality assessment and phytochemical screening of selected region of Withania somnifera Dunal. Int J Pharmaceut Sci Res 1:73–77 (icid: 913296)
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43. doi:10.1016/S0022-5320(69)90033-1
Turner GW, Gershenzon J, Croteau RB (2000) Development of peltate glandular trichomes of peppermint. Plant Physiol 124:665–679. doi:10.1104/pp.124.2.665
Uddin Q, Samiulla L, Singh VK, Jamil S (2012) Phytochemical and pharmacological profile of Withania somnifera Dunal: a review. J Appl Pharmaceut Sci 2:170–175
van Wyk BE (2008) A broad review of commercially important southern African medicinal plants. J Ethnopharmacol 119:342–355. doi:10.1016/j.jep.2008.05.029
Wagner GJ, Wang E, Shepherd RW (2004) New approaches for studying and exploiting an old protuberance, the plant trichome. Ann Bot 93:3–11. doi:10.1093/aob/mch011
Werker E (2000) Trichome diversity and development. Adv Bot Res 31:1–35. doi:10.1016/S0065-2296(00)31005-9
Werker E, Putievsky E, Ravid U, Dudai N, Katzir I (1993) Glandular hairs and essential oil in developing leaves of Ocimum basilicum L. (Lamiaceae). Ann Bot 71:43–50. doi:10.1006/anbo.1993.1005
Winter N, Kollwig G, Zhang S, Kragler F (2007) MPB2C, a microtubule-associated protein, regulates non-cell-anatomy of the homeodomain protein KNOTTED. Am Soc Plant Biol 19:3001–3018. doi:10.1105/tpc.107.044354
Wink M (1998) Alkaloids: biochemistry, ecology, and medicinal application. In: MF Roberts, M Wink. Plenum Press, New York. pp. 265–300
Yan A, Pan J, An L, Gan Y, Feng H (2012) The responses of trichome mutants to enhanced ultraviolet-B radiation in Arabidopsis thaliana. J Photochem Photobiol B Biol 113:29–35 (pmid: 22647943)
Yu H, Kolwalski SP, Steffens JC (1992) Comparison of polyphenol oxidase expression in glandular trichomes of Solanum and Lycopersicon species. Plant Physiol 100:1885–1890. doi:10.1104/pp.100.4.1885
Acknowledgments
The authors would like to thank Preyan Arumugam, Kashmira Raghu and Ashlin Munsamy for research assistance and aiding with histochemical preparation. Drs S. Gairola and C.T. Sadashiva are thanked for editing the manuscript and assistance with phytochemical and TLC studies, respectively. Funding from the National Research Foundation (NRF) is also gratefully appreciated.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Munien, P., Naidoo, Y. & Naidoo, G. Micromorphology, histochemistry and ultrastructure of the foliar trichomes of Withania somnifera (L.) Dunal (Solanaceae). Planta 242, 1107–1122 (2015). https://doi.org/10.1007/s00425-015-2341-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-015-2341-1