Abstract
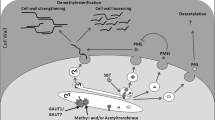
Pectin methylesterases (PMEs) are ubiquitous enzymes present in the plant cell wall. They catalyse the demethylesterification of homogalacturonic acid units of pectins, which, in turn, can be associated with different physiological phenomena. In this study, different flax (Linum usitatissimum L.) PME isoforms were observed: neutral (pI 7.0 and 7.5, MW: 110 kDa), basic (pI 8.3 and 8.5, MW: 110 kDa) and very basic (pI>9.5, MW: 38 kDa). In an attempt to identify most of the expressed cell wall LuPME isoforms, polyclonal antibodies were raised against a conserved region of PME. These antibodies allowed the purification of the very basic PME isoform (pI 9.5, MW: 36 kDa) from flax cells, designated LuPME5. This isoform corresponds to the Lupme5 cDNA isolated, at the same time, from flax hypocotyls, by using the RACE–PCR technique. Semi-quantitative PCR experiments showed that the Lupme5 transcript was highly expressed in the hypocotyl zones where elongation is being achieved. Thus, this enzyme may be involved in cell wall stiffening.





Similar content being viewed by others
Abbreviations
- GST :
-
Glutathione S-transferase
- IEF :
-
Isoelectric focusing
- pI :
-
Isoelectric point
- MALDI–TOF :
-
Matrix-assisted laser desorption/ionisation–time of flight
- PME :
-
Pectin methylesterase
- RACE :
-
Rapid amplification of cDNA ends
- RT–PCR:
-
Reverse transcription–polymerase chain reaction
References
Albani D, Altosaar I, Arnison PG, Fabijanski SF (1991) A gene showing similarity to pectin esterase is specifically expressed in developing pollen of Brassica napus. Sequences in its 5′ flanking region are conserved in other pollen-specific promoters. Plant Mol Biol 16:501–513
Alexandre F, Morvan O, Gaffé J, Mareck A, Jauneau A, Dauchel H, Balangé AP, Morvan C (1997) Pectin methylesterase pattern in flax seedlings during their development. Plant Physiol Biochem 35:427–436
Ausubel FM, Brent R, Kingston RE, Moore D, Smith JA, Seidman JG, Struhl K (1988) Current protocols in molecular biology. Analysis of proteins. Greene and Wiley, New York, pp 1000–1017
Aviv H, Leder P (1972) Purification of biologically active globin messenger RNA by chromatography on olithymidylic acid–cellulose. Proc Natl Acad Sci USA 69:1408–1412
Baron-Epel O, Gharyl PK, Graig JWT (1988) Pectins as wall mediators of wall porosity in soybean cells. Planta 175:389–395
Berthe T, Klein-Eude D, Balangé AP (2003) Study of 5-aminolevulinate dehydratase in radish seedlings: are there housekeeping and light-induced enzymes? Plant Sci 164:395–405
Bertheau Y, Madgidi-Hervan E, Kotoujanski A, Nguyen-The C, Andro T, Coleno A (1984) Detection of depolymerase isoenzymes after electrophoresis or electrofocusing, or in titration curves. Anal Biochem 139:383–389
Bordenave M, Breton C, Goldberg R, Huet JC, Perez S, Pernollet JC (1996) Pectinmethylesterase isoforms from Vigna radiata and molecular cloning of a cDNA encoding the most alkaline isoform. Plant Mol Biol 31:1039–1049
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Carpita NC, Gibeaut DM (1993) Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physiological properties of the walls during growth. Plant J 3:1–30
Chen MH, Sheng J, Hind G, Handa AK, Citousky V (2000) Interaction between the tobacco mosaic virus movement protein and host cell pectin methylesterases is required for viral cell-to-cell movement. EMBO J 19:913–920
Chirgwin JM, Przybyla AE, MacDonald RJ, Rutter WJ (1979) Isolation of biologically active ribonucleic acid from sources enriched in ribonucleases. Am Chem Soc 18:5294–5299
Christensen TMIE, Nielsen JE, Kreiberg JD, Rasmussen P, Mikkelsen JD (1998) Pectin methylesterase from orange fruit: characterization and localization by in-situ hybridization and immunohistochemestry. Planta 206:493–503
Demarty M, Morvan C, Thellier M (1984) Calcium and the cell wall. Plant Cell Environ 7:441–448
Dorokhov YL, Mäkinen K, Frolova OY, Merits A, Saarinen J, Kalkkinen N, Atabekov JG, Saarma M (1999) A novel function for a ubiquitous plant enzyme pectin methylesterase: the host-cell receptor for the tobacco mosaic virus movement protein. FEBS Lett 461:223–228
Ebbelar MEM, Tucker GA, Laats MM, Dijk CV, Stolle-Smits T, Recourt K (1996) Characterization of pectinases and pectin methylesterase cDNAs in pods of green beans (Phaseolus vulgaris L.). Plant Mol Biol 31:1141–1151
Frohman MA, Dush MK, Martin GR (1988) Rapid production of full-length cDNAs from rare transcripts: amplification using a single gene-specific oligonucleotide primer. Proc Natl Acad Sci USA 85:8998–9002
Gaffé J, Morvan C, Jauneau A, Demarty M (1992) Partial purification of flax cell wall pectin methylesterase. Phytochemistry 31:761–765
Gaffé J, Tieman DM, Handa AK (1994) Pectin methylesterase isoforms in tomato Lycopersicum esculentum tissue. Effects of expression of a pectin methylesterase antisense gene. Plant Physiol 105:199–203
Gaffé J, Tiznado ME, Handa AK (1997) Characterization and functional expression of a ubiquitously expressed tomato pectin methylesterase. Plant Physiol 114:1547–1556
Goldberg R, Morvan C, Jauneau A, Jarvis MC (1995) Methyl-esterification, de-esterification and gelation of pectins in the primary cell wall. In: Visser J, Voragen AGJ (eds) Pectins and pectinases. Elsevier, Wageningen, pp 151–172
Hall LN, Tucker GA, Smith CJS, Watson CF, Seymour GB, Bundlick Y, Boniwell JM, Fletcher JD, Ray JA, Schuch W, Bird CR, Grierson D (1993) Antisense inhibition of pectin esterase gene expression in transgenic tomatoes. Plant J 3:121–129
Jarvis MC (1984) Structure and properties of pectin gels in plant cell walls. Plant Cell Environ 7:153–164
Kim JB, Carpita NC (1992) Changes in esterification of uronic acid groups of cell wall polysaccharides during elongation of maize coleoptiles. Plant Physiol 98:646–653
Kim HU, Park BS, Jin YM, Chung TY (1997) Promoter sequences of two homologous pectin esterase genes from Chinese cabbage (Brassica campestris L. ssp pekinsis) and pollen-specific expression of the GUS gene driven by a promoter in tobacco plants. Mol Cell 7:21–27
Klavons JA, Bennett AD (1986) Determination of methanol using alcohol oxidase and its application to methyl ester content of pectins. J Agric Food Chem 34:597–598
Knox JP, Linstead PJ, Kong J, Cooper C, Roberts K (1990) Pectin esterification is spatially regulated both within cell walls and between developing tissues of root apices. Planta 181:512–521
Lacoux J, Klein D, Domon JM, Burel C, Lamblin F, Alexandre F, Sihachakr D, Roger D, Balangé AP, David A, Morvan C, Lainé E (2003) Antisense transgenesis of Linum usitatissimum with a pectin methylesterase cDNA. Plant Physiol Biochem 41:241–249
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 227:680–685
Lievens S, Goormatching S, Herman S, Holsters M (2002) Patterns of pectin methyl esterase transcripts in developing stem nodules of Sesbania rostrata. Mol Plant Microbe Interact 15:164–168
Mareck A, Gaffé J, Morvan O, Alexandre F, Morvan C (1995) Characterization of isoforms of pectin methylesterase of Linum usitatissimum using polyclonal antibodies. Plant Cell Physiol 36:409–417
Micheli F (2001) Pectin methylesterases: cell wall enzymes with important roles in plant physiology. Trends Plant Sci 6:414–419
Micheli F, Sundberg B, Goldberg R, Richard L (2000) Radial distribution pattern of PME across the cambial region of hybrid aspen at activity and dormancy. Plant Physiol 124:191–199
Nari J, Noat G, Diamantidis G, Woudstra M, Ricard J (1986) Electrostatic effects and the dynamics of enzymes reactions at the surface of plant cells; III. Interplay between limited cell wall autolysis, pectin methylesterase activity and electrostatic effects in soybean cell wall. Eur J Biochem 155:199–210
Ouchterlony O (1949) Antigen-antibody reactions in gels. Acta Pathol Microbiol Scan 26:507–509
Pilling J, Willmitzer L, Fishan J (2000) Expression of a Petunia inflata pectin methylesterase in Solanum tuberosum L. enhances stem elongation and modifies cation distribution. Planta 210:391–399
Roberts K (1990) Structure at the plant cell surface. Curr Cell Biol 2:920–928
Roger D, Lacoux J, Lamblin F, Gaillet D, Dauchel H, Klein D, Balangé AP, David A, Lainé E (2001) Isolation of a flax pectin methylesterase promoter and its expression in transgenic tobacco. Plant Sci 160:713–721
Schaumann A, Bruyant-Vannier MP, Goubet F, Morvan C (1993) Pectic metabolism in suspension-cultured cells of flax, Linum usitatissimum. Plant Cell Physiol 34:891–897
Shimizu Y, Aotsuka S, Hasegawa O, Kawada T, Sakuno T, Sakai F, Hayashi T (1997) Changes in levels of mRNAs for cell wall-related enzymes in growing cotton fiber cells. Plant Cell Physiol 38:375–378
Tieman DM, Harriman RW, Ramamohan G, Handa, AK (1992) An antisense pectin methylesterase gene alters pectin chemistry and soluble solids in tomato fruit. Plant Cell 4:667–679
Wakeley PR, Rogers HJ, Rozycka M, Greenland AJ, Hussey PJ (1998) A maize pectin methylesterase-like gene, ZmC5, specifically expressed in pollen. Plant Mol Biol 37:187–192
Wen F, Zhu Y, Hawes MC (1999) Effect of pectin methylesterase gene expression on pea root development. Plant Cell 11:1129–1140
Zhang GF, Staehelin LA (1992) Functional compartmentalization of the Golgi apparatus of plant cells. An immunochemical analysis of high pressure frozen and freeze substituted sycamore maple suspension-cultured cells. Plant Physiol 99:1070–1083
Acknowlegements
S. Al-Qsous and E. Carpentier contributed equally to this work. We acknowledge the région Haute-Normandie (MIIAT-BP; E.C.) and the Jordanian Kingdom (S.A.-Q.) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Accession numbers: Lupme1, AF355056; Lupme3, AF188895; Lupme5, AF355057
Rights and permissions
About this article
Cite this article
Al-Qsous, S., Carpentier, E., Klein-Eude, D. et al. Identification and isolation of a pectin methylesterase isoform that could be involved in flax cell wall stiffening. Planta 219, 369–378 (2004). https://doi.org/10.1007/s00425-004-1246-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-004-1246-1