Abstract
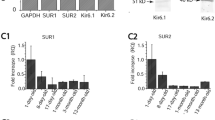
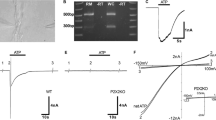
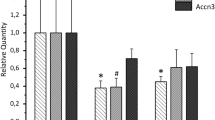
Using the double whole-cell patch-clamp technique, we found that the absence of intracellular ATP led to gap junction uncoupling in cochlear-supporting Hensen cells. The uncoupling was observed as a progressive reduction of the gap junctional electrical conductance from a starting value of approximately 40 nS to less than 0.04 nS within 10–20 min. The conductance rundown was partly avoided by at least 3 mM ATP and completely suppressed by 5 mM ATP or 5ʹ-adenylyl-imidodiphosphate (AMP-PNP), the non-hydrolysable ATP analog, in the pipette filling solution, suggesting that ATP was needed as ligand and not as a hydrolysable energy supplier or substrate for enzymatic reactions. The effect of intracellular ATP was mimicked by the external application of barium, a nonselective blocker of inwardly rectifying K+ (Kir) channels, and glibenclamide, an inhibitor of the ATP-sensitive Kir channels (KATP). Moreover a Ba2+-sensitive whole-cell inward current was observed in absence of internal ATP. We propose that the internal ATP kept the KATP channels in a closed state, thereby maintaining the gap junction coupling of Hensen cells. The immunostaining of guinea pig cochlear tissue revealed for the first time the expression of the KATP channel subunits Kir6.1 and SUR1 in Hensen cells and supported the proposed hypothesis. The results suggest that KATP channels, as regulator of the gap junction coupling in Hensen cells, could be the physiological link between the metabolic state of the supporting cells and K+ recycling in the organ of Corti.




Similar content being viewed by others
References
Blödow A, Ngezahayo A, Ernst A, Kolb HA (2003) Calmodulin antagonists suppress gap junctional coupling in isolated Hensen cells of the guinea pig cochlea. Pflugers Arch 446:36–41
Bruzzone R, White TW, Paul DL (1996) Connections with connexins: the molecular basis of direct intercellular signalling. Eur J Biochem 244:1–27
Chen J, Zhao HB (2014) The role of an inwardly rectifying K+ channel (Kir4.1) in the inner ear and hearing loss. Neuroscience 0:137–146
Cohen-Salmon M, Ott T, Michel V, Hardelin JP, Perfettini I, Eybalin M, Wu T, Marcus DC, Wangemann P, Willecke K, Petit C (2002) Targeted ablation of connexin26 in the inner ear epithelium gpa junction network causes hearing impairment and cell death. Curr Biol 12:1006–1111
Donaldson PJ, Dong Y, Roos M, Green C, Goodenough DA, Kistler J (1995) Changes in lens connexin expression lead to increased gap junctional voltage dependence and conductance. Am J Physiol 69:C590–C600
Evans HW, Martin PE (2002) Gap junctions: structure and function (review). Mol Membr Biol 19:121–136
Flock A, Flock B, Fridberger A, Scarfone E, Ulfendahl M (1999) Gap junctions and connexin expression in the inner ear. Novartis Found Symp 219:134–136
Forge A, Becker D, Casalotti S, Edwards J, Marziano N, Nickel R (2002) Connexins and gap junctions in the inner ear. Audiol Neurootol 7:141–145
Gale JE, Jagger DJ (2010) Cochlear supporting cells. In: Fuchs PA (ed) The Oxford handbook of auditory science: the ear. Oxford UP, Oxford, pp 307–327
Granda B, Tabernero A, Sanchez-Abarca LI, Medina JM (1998) The K-ATP channel regulates the effect of Ca2+ on gap junction permeability in cultured astrocytes. FEBS Lett 427:41–45
Grifa A, Wagner CA, D’Ambrosio L, Melchionda S, Bernardi F, Lopez-Bigas N, Rabionet R, Arbones M, Monica MD, Estivill X, Zelante L, Lang F, Gasparini P (1999) Mutations in GJB6 cause nonsyndromic autosomal dominant deafness at DFNA3 locus. Nat Genet 23:16–18
Hibino H, Horio Y, Ianobe A, Doi K, Ito M, Yamada M, Gotow T, Uchiyama Y, Kawamura M, Kubo T, Kurachi Y (1997) An ATP-dependent inwardly rectifying potassium channel, KAB-2 (Kir4.1), in cochlear stria vascularis of inner ear: its specific subcellular localization and correlation with the formation of endocochlear potential. J Neurosci 17:4711–4721
Hibino H, Inanobe A, Furutani K, Murakami S, Findlay I, Kurachi Y (2010) Inwardly rectifying potassium channels: their structure, function, and physiological roles. Physiol Rev 90:291–366
Jiang K, Wang J, Zhao C, Feng M, Shen Z, Yu Z, Xia Z (2011) Regulation of gap junctional communication by astrocytic mitochondrial K(ATP) channels following neurotoxin administration in in vitro and in vivo models. Neurosignals 19:63–74
Kelsell DP, Dunlop J, Stevens HP, Lench NJ, Liang JN, Parry G, Mueller RF, Leigh IM (1997) Connexin 26 mutations in heredirary nonsyndromic sensorineural deafness. Nature 387:80–83
Kikuchi T, Adams JC, Miyabe Y, So E, Kobayashi (2000) Potassium ion recycling pathway via gap junction systems in the mammalian cochlea and its interruption in hereditary nonsyndromic deafness. Med Electron Microsc 33:51–56
Kitano I, Mori N, Matsunaga T (1995) Role of ATP-sensitive K+ channels in anoxia-sensitive negative potential of endolymph. Hear Res 90:24–30
Kumar NM, Gilula NB (1996) The gap junction communication channel. Cell 84:381–388
Lautermann J, ten Cate WJF, Altenhoff P, Gruemmer R, Traub O, Frank HG, Jahnke K, Winterhager E (1998) Expression of the gap-junction connexins 26 and 30 in the rat cochlea. Cell Tissue Res 294:415–420
Marcus DC, Wu T, Wangemann P, Kofuji P (2002) KCNJ10 (Kir4.1) potassium channel knockout abolishes endocochlear potential. Am J Physiol 282:C403–C407
Naitoh K, Ichikawa Y, Miura T, Nakamura Y, Miki T, Ikeda Y, Kobayashi H, Nishihara M, Ohori K, Shimamoto K (2006) MitoKATP channel activation suppresses gap junction permeability in the ischemic myocardium by an ERK-dependent mechanism. Cardiovasc Res 70:374–383
Nenov AP, Chen C, Bobbin RP (1998) Outward rectifying potassium currents are the dominant voltage activated currents present in Deiters’ cells. Hear Res 123:168–182
Neyton J, Trautmann A (1985) Single-channel currents of an intercellular junction. Nature 317:331–335
Ngezahayo A, Altmann B, Kolb HA (2003) Regulation of ion fluxes, cell volume and gap junctional coupling by cGMP in GFSHR-17 granulosa cells. J Membrane Biol 194:165–176
Nichols CG (2006) KATP channels as molecular sensors of cellular metabolism. Nature 440:470–476
Rocheleau JV, Remedi MS, Granada B, Head WS, Koster JC, Nichols CG, Piston DW (2006) Critical role of gap junction coupled KATP channel activity for regulated insulin secretion. PLoS Biol 4(2):e26, Epub 2006 Jan 17
Saez JC, Spray DC, Nairn AC, Hertzberg E, Greengard P, Bennett MVL (1986) cAMP increases junctional conductance and stimulates phosphorylation of the 27-kDa principal gap junction polypeptide. Proc Natl Acad Sci 83:2473–2477
Santos-Sacchi J (1985) The effects of cytoplasmic acidification upon electrical coupling in the organ of Corti. Hear Res 19:207–215
Santos-Sacchi J (1986) The temperature dependence of electrical coupling in the organ of Corti. Hear Res 21:205–211
Santos-Sacchi J (2000) Cell coupling in the organ of Corti. Brain Res Rev 32:167–171
Sato Y, Santos-Sacchi J (1994) Cell coupling in the supporting cells of Corti’s organ: sensitivity to intracellular H+ and Ca++. Hear Res 80:21–24
Söhl G, Willecke K (2004) Gap junctions and the connexin protein family. Cardiovasc Res 62:228–232
Solan JL, Lampe PD (2005) Connexin phosphorylation as a regulatory event linked to gap junction channel assembly. Biochim Biophys Acta 1711:154–163
Spicer SS, Schulte BA (1996) The fine structure of spiral ligament cells relates to ion return to the stria and varies with place-frequency. Hear Res 100:80–100
Spitzer N, Sammons GS, Price EM (2011) Autofluorescent cells in rat brain can be convincing impostors in green fluorescent reporter studies. J Neurosci Methods 197:48–55
Todt I, Ngezahayo A, Ernst A, Kolb HA (1999) Inhibition of gap junctional coupling in cochlear supporting cells by gentamicin. Pflugers Arch 438:865–867
Todt I, Ngezahayo A, Ernst A, Kolb HA (2001) Hydrogenperoxide inhibits gap junctional coupling and modulates intracellular free calcium in cochlear Hensen-cells. J Membr Biol 181:107–114
Velasco A, Tabernero A, Granda B, Medina JM (2000) ATP-sensitive potassium channel regulates astrocytic gap junction permeability by a Ca2+-independent mechanism. J Neurochem 74:1249–1256
Vera B, Sanchez-Abarca LI, Bolanos JP, Medina JM (1996) Inhibition of astrocyte gap junctional communication by ATP depletion is revered by calcium sequestration. FEBS 392:225–228
Willecke K, Eiberger J, Degen J, Eckhardt D, Romualdi A, Güldenagel M, Deutsch U, Söhl U (2002) Structural and functional diversity of connexin genes in the mouse and human genome. Biol Chem 383:725–737
Wollbold J, Jaster R, Müller S, Rateitschak K, Wolkenhauer O (2014) Anti-inflammatory effects of reactive oxygen species—a multi-valued logical model validated by formal concept analysis. BMC Syst Biol 8:101
Xia JH, Liu CY, Tang BS, Pan Q, Huang L, Dai HP, Zhang BR, Xie W, Hu DX, Zheng D, Shi XL, Wang DA, Xia K, Yu KP, Liao XD, Feng Y, Yang YF, Xiao JY, Xie DH, Huang JZ (1998) Mutations in the gene encoding gap junction protein beta-3 associated with autosomal dominant hearing impairment. Nat Genet 20:370–373
Yang J, Wang J (2002) Possible function of outward potassium currents in isolated Deiters’cells of guinea pig cochlea. Chin Med J 115:264–267
Zelante L, Gasparini P, Estivill X, Melchionda S, D’Agruma L, Govea N, Milá M, Monica MD, Lutfi J, Shohat M, Mansfield E, Delgrosso K, Rappaport E, Surrey S, Fortina P (1997) Connexin26 mutations associated with the most common form of non-syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum Mol Genet 6:1605–1609
Zhao HB, Santos-Sacchi J (1998) Effect of membrane tension on gap junctional conductance of supporting cells in Corti’s organ. J Gen Physiol 112:447–455
Zhao HB, Yu N, Fleming CR (2005) Gap junctional hemichannel-mediated ATP release and hearing controls in the inner ear. Proc Natl Acad Sci U S A 102:18724–18729
Zhao HB, Kikuchi T, Ngezahayo A, White TW (2006) Gap junction and cochlear homeostasis. J Membr Biol 209:177–186
Acknowledgments
The authors thank Ina G. Siller for the help with imaging the cochlear slides. We thank Dr. Kathrin Rübensam, ZTL, MH Hannover, and Dr. Henning Vogt, HNO-Klinik, MH Hannover for their kind help with the animals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blödow, A., Begandt, D., Bader, A. et al. ATP-sensitive K+ channels (Kir6.1/SUR1) regulate gap junctional coupling in cochlear-supporting cells. Pflugers Arch - Eur J Physiol 468, 1215–1222 (2016). https://doi.org/10.1007/s00424-016-1815-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-016-1815-8