Abstract
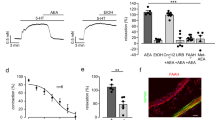
This study was aimed to investigate the role of eicosapentaenoic acid monoacylglyceride (MAG-EPA) and 17,18-epoxyeicosatetraenoic acid (17,18-EpETE) on the regulation of contractile reactivity and nuclear protein expression in 72-h-cultured and TNF-α-treated guinea pig tracheal rings. Tension measurements performed on native tissues demonstrated that the cytochrome P-450 epoxygenase (CYP450)-dependent EPA metabolite, 17,18-EpETE, displayed a higher potency than MAG-EPA in inhibiting U-46619-induced tone. Calphostin C (a PKC inhibitor), whether in association or not with MAG-EPA or 17,18-EpETE, had no further effect, while 17,18-EpETE and Y-27632 (a Rho kinase inhibitor) yielded additive effects. Of note, MAG-EPA and 17,18-EpETE pre-treatments normalized the contractile responses to broncho-constrictive agents in 72-h-cultured trachea. The enhanced expression of TNF-α, P-p65-nuclear factor kappaB (NF)-κB, c-fos and c-Jun in 72-h-cultured tissues likely contributed to the hyperresponsiveness. β-Escin-permeabilized preparations demonstrated that 17,18-EpETE abolished Ca2+ hypersensitivity, suggesting a blunting of PKC and/or Rho kinase activation. Lastly, activation of NF-κB and activating protein-1 (AP-1) signalling by exogenous TNF-α markedly increased the contractile response to MCh, through an increase in 17-kDa PKC-potentiated inhibitory protein of PP1 (CPI-17) phosphorylation and IκBα degradation. Dual incubation of 17,18-EpETE with calphostin C or Y-27632 induced cumulative inhibitory effects on MCh responses in TNF-α-incubated tracheal rings. 17,18-EpETE also reduced the detection level of P-p65-NF-κB and AP-1 subunits. The present data provide evidence that MAG-EPA, through its bioactive metabolite, represents a prospective pharmacological target in respiratory diseases.







Similar content being viewed by others
Abbreviations
- AHR:
-
Airway hyperresponsiveness
- HT:
-
Hyperreactive trachea
- MAG-EPA:
-
Eicosapentaenoic acid monoacylglyceride
- AP-1:
-
Activating protein-1
- PUFA:
-
Polyunsaturated fatty acid
- TNF-α:
-
Tumour necrosis factor alpha
References
Benoit C, Renaudon B, Salvail D, Rousseau E (2001) EETs relax airway smooth muscle via an EpDHF effect: BK(Ca) channel activation and hyperpolarization. Am J Physiol Lung Cell Mol Physiol 280(5):L965–L973
Brightling C, Berry M, Amrani Y (2008) Targeting TNF-alpha: a novel therapeutic approach for asthma. J Allergy Clin Immunol 121(1):5–10
Bruns RF, Miller FD, Merriman RL, Howbert JJ, Heath WF, Kobayashi E, Takahashi I, Tamaoki T, Nakano H (1991) Inhibition of protein kinase C by calphostin C is light-dependent. Biochem Biophys Res Commun 176(1):288–293
Carlin S, Yang KX, Donnelly R, Black JL (1999) Protein kinase C isoforms in human airway smooth muscle cells: activation of PKC-zeta during proliferation. Am J Physiol 276(3):L506–L512
Chang JH, Pratt JC, Sawasdikosol S, Kapeller R, Burakoff SJ (1998) The small GTP-binding protein Rho potentiates AP-1 transcription in T cells. Mol Cell Biol 18(9):4986–4993
Chang CS, Sun HL, Lii CK, Chen HW, Chen PY, Liu KL (2010) Gamma-linolenic acid inhibits inflammatory responses by regulating NF-kappaB and AP-1 activation in lipopolysaccharide-induced RAW 264.7 macrophages. Inflammation 33(1):46–57
Chiba Y, Misawa M (2004) The role of RhoA-mediated Ca2+ sensitization of bronchial smooth muscle contraction in airway hyperresponsiveness. J Smooth Muscle Res 40(4–5):155–678
Cleary RA, Wang R, Wang T, Tang DD (2013) Role of Abl in airway hyperresponsiveness and airway remodeling. Respir Res 11:14–105
Dakshinamurti S, Mellow L, Stephens NL (2005) Regulation of pulmonary arterial myosin phosphatase activity in neonatal circulatory transition and in hypoxic pulmonary hypertension: a role for CPI-17. Pediatr Pulmonol 40:398–407
Diaz Encarnacion MM, Warner GM, Cheng J, Gray CE, Nath KA, Grande JP (2011) n-3 fatty acids block TNF-α-stimulated MCP-1 expression in rat mesangial cells. Am J Physiol Ren Physiol 300(5):F1142–F1151
Eto M, Bock R, Brautigan DL, Linden DJ (2002) Cerebellar long-term synaptic depression requires PKC-mediated activation of CPI-17, a myosin/moesin phosphatase inhibitor. Neuron 36:1145–1158
Eto M, Senba S, Morita F, Yazawa M (1997) Molecular cloning of a novel phosphorylation-dependent inhibitory protein of protein phosphatase-1 (CPI-17) in smooth muscle: its specific localization in smooth muscle. FEBS Lett 410:356–360
Gallos G, Townsend E, Yim P, Virag L, Zhang Y, Xu D, Bacchetta M, Emala CW (2012) Airway epithelium is a predominant source of endogenous airway GABA and contributes to relaxation of airway smooth muscle tone. Am J Physiol Lung Cell Mol Physiol 304(3):L191–L197
Henderson WR Jr, Chi EY, Teo JL, Nguyen C, Kahn MA (2002) Small molecule inhibitor of redox-regulated NF-kappa B and activator protein-1 transcription blocks allergic airway inflammation in a mouse asthma model. J Immunol 169(9):5294–5299
Henry PJ, Mann TS, Goldie RG (2005) A Rho kinase inhibitor, Y-27632 inhibits pulmonary eosinophilia, bronchoconstriction and airways hyperresponsiveness in allergic mice. Pulm Pharmacol Ther 18(1):67–74
Holgate ST (2011) The sentinel role of the airway epithelium in asthma pathogenesis. Immunol Rev 242(1):205–219
Hoover WC, Zhang W, Xue Z, Gao H, Chernoff J, Clapp DW, Gunst SJ, Tepper RS (2012) Inhibition of p21 activated kinase (PAK) reduces airway responsiveness in vivo and in vitro in murine and human airways. PLoS One 7(8):e42601
Hu W, Mahavadi S, Li F, Murthy KS (2007) Upregulation of RGS4 and downregulation of CPI-17 mediate inhibition of colonic muscle contraction by interleukin-1beta. Am J Physiol Cell Physiol 293:C1991–C2000
Huang XS, Chen LY, Doerner AM, Pan WW, Smith L, Huang S, Papadimos TJ, Pan ZK (2009) An atypical protein kinase C (PKC zeta) plays a critical role in lipopolysaccharide-activated NF-κB in human peripheral blood monocytes and macrophages. J Immunol 182(9):5810–5815
Hunter I, Cobban HJ, Vandenabeele P, Macewan DJ, Nixon GF (2003) Tumor necrosis factor-alpha-induced activation of Rho A in airway smooth muscle cells: role in the Ca2+ sensitization and myosin light chain20 phosphorylation. Mol Pharmacol 63(3):714–721
Im DS (2012) Omega-3 fatty acids in anti-inflammation (pro-resolution) and GPCRs. Prog Lipid Res 51(3):232–237
Ismailoglu UB, Van Scott MR, Fedan JS (2009) Effects of cytokines on mechanical and epithelial bioelectric responses to methacholine and hyperosmolarity in guinea-pig airways: an in vitro study. Eur J Pharmacol 612(1–3):115–121
Kagoshima M, Ito K, Cosio B, Adcock IM (2003) Glucocorticoid suppression of nuclear factor-kappa B: a role for histone modifications. Biochem Soc Trans 31(Pt1):60–65
Kubota T, Arita M, Isobe Y, Iwamoto R, Goto T, Yoshioka T, Urabe D, Inoue M, Arai H (2014) Eicosapentanoic acid is converted via ω-3 epoxygenation to the anti-inflammatory metabolite 12-hydroxy-17,18-epoxyeicosatetrenoic acid. FASEB J 28(2):586–593
Lauterbach B, Barbosa-Sicard E, Wang MH, Honeck H, Kärgel E, Theuer J, Schwartzman ML, Haller H, Luft FC, Gollasch M, Schunck WH (2002) Cytochrome P450-dependent eicosapentaenoic acid metabolites are novel BK channel activators. Hypertension 39:609–613
Manerba A, Vizzardi E, Metra M, Dei Cas L (2010) n-3 PUFAs and cardiovascular disease prevention. Futur Cardiol 6(3):343–350
Morin C, Fortin S, Cantin AM, Rousseau É (2013) MAG-EPA resolves lung inflammation in an allergic model of asthma. Clin Exp Allergy 43(9):1071–1082
Morin C, Fortin S, Rousseau E (2012) Bronchial inflammation induced PKC zeta over-expression: involvement in mechanical properties of airway smooth muscle. Can J Physiol Pharmacol 90(2):261–269
Morin C, Proteau S, Rousseau E, Brayden J (2005) Organ-cultured airway explants: a new model of airway hyper responsiveness. Exp Lung Res 31(7):719–744
Morin C, Sirois M, Echavé V, Albadine R, Rousseau E (2010) 17,18-epoxyeicosatetraenoic acid targets PPARγ and p38 mitogen-activated protein kinase to mediate its anti-inflammatory effects in the lung: role of soluble epoxide hydrolase. Am J Respir Cell Mol Biol 43(5):564–575
Morin C, Sirois M, Echave V, Gomes MM, Rousseau E (2008) EET displays anti-inflammatory effects in TNF-alpha stimulated human bronchi: putative role of CPI-17. Am J Respir Cell Mol Biol 38(2):192–201
Morin C, Sirois M, Echave V, Rizcallah E, Rousseau E (2009) Relaxing effects of 17(18)-EpETE on arterial and airway smooth muscles in human lung. Am J Physiol Lung Cell Mol Physiol 296(1):L130–L139
Ohama T, Hori M, Sato K, Ozaki H, Karaki H (2003) Chronic treatment with interleukin-1beta attenuates contractions by decreasing the activities of CPI-17 and MYPT-1 in intestinal smooth muscle. J Biol Chem 278:48794–48804
Pelaia G, Renda T, Gallelli L, Vatrella A, Busceti MT, Agati S, Caputi M, Cazzola M, Maselli R, Marsico SA (2008) Molecular mechanisms underlying airway smooth muscle contraction and proliferation: implications for asthma. Respir Med 102(8):1173–1181
Pyne S, Pyne NJ (1993) Bradykinin stimulates phospholipase D in primary cultures of guinea-pig tracheal smooth muscle. Biochem Pharmacol 45(3):593–603
Ratz PH, Meehl JT, Eddinger TJ (2002) RhoA kinase and protein kinase C participate in regulation of rabbit stomach fundus smooth muscle contraction. Br J Pharmacol 137(7):983–992
Redhu NS, Saleh A, Halayko AJ, Ali AS, Gounni AS (2011) Essential role of NF-κB and AP-1 transcription factors in TNF-α-induced TSLP expression in human airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol 300(3):L479–L485
Renzetti LM, Paciorek PM, Tannu SA, Rinaldi NC, Tocker JE, Wasserman MA, Gater PR (1996) Pharmacological evidence for tumor necrosis factor as a mediator of allergic inflammation in the airways. J Pharmacol Exp Ther 278(2):847–853
Revermann M, Schloss M, Mieth A, Babelova A, Schröder K, Neofitidou S, Buerkl J, Kirschning T, Schermuly RT, Hofstetter C, Brandes RP (2011) Levosimendan attenuates pulmonary vascular remodeling. Intensive Care Med 37(8):1368–1377
Ricciardolo FL, Nijkamp F, De Rose V, Folkerts G (2008) The guinea pig as an animal model for asthma. Curr Drug Targets 9(6):452–465
Ruan YC, Zhou W, Chan HC (2011) Regulation of smooth muscle contraction by the epithelium: Role of prostaglandins. Physiology 26:156–170
Sakai H, Chiba Y, Hirano T, Misawa M (2005) Possible involvement of CPI-17 in augmented bronchial smooth muscle contraction in antigen-induced airway hyper-responsive rats. Mol Pharmacol 68(1):145–151
Senouvo FY, Tabet Y, Morin C, Albadine R, Sirois C, Rousseau E (2011) Improved bioavailability of epoxyeicosatrienoic acids reduces TP-receptor agonist-induced tension in human bronchi. Am J Physiol Lung Cell Mol Physiol 301(5):L675–L682
Shaulian E, Karin M (2002) AP-1 as a regulator of cell life and death. Nat Cell Biol 4(5):E131–E136
Shea-Donohue T, Notari L, Sun R, Zhao A (2012) Mechanisms of smooth muscle responses to inflammation. Neurogastroenterol Motil 24(9):802–811
Thomas PS, Yates DH, Barnes PJ (1995) Tumor necrosis factor-alpha increases airway responsiveness and sputum neutrophilia in normal human subjects. Am J Respir Crit Care Med 152:76–80
Wang T, Kendig DM, Smolock EM, Moreland RS (2009) Carbachol induced rabbit bladder smooth muscle contraction: roles of protein kinase C and Rho kinase. Am J Physiol Ren Physiol 297(6):F1534–F1542
Yamamoto Y, Gaynor RB (2001) Therapeutic potential of inhibition of the NF-kappaB pathway in the treatment of inflammation and cancer. J Clin Invest 107(2):135–142
Zhao Y, Chen LH (2005) Eicosapentanoic acid prevents lipopolysaccharide-stimulated DNA binding of activator protein-1 and c-Jun N-terminal kinase activity. J Nutr Biochem 16(2):78–84
Acknowledgments
We wish to thank Dr. Caroline Morin and Dr. Samuel Fortin from SCF Pharma for the gift of MAG-EPA. The authors wish to thank Mr. Pierre Pothier, Ms. Stephanie Corriveau and Elyse Burt for critical review of the manuscript. This work is supported by a CIHR grant MOP-111112 to ER who is a member of the Respiratory Health Network of the FRQS: www.rsr-frqs.ca. We are especially grateful to the members of the animal care facility at the Faculty of Medicine and Health Sciences for their precious collaboration, as well as Mr. Yacine Tabet for logistic support.
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khaddaj-Mallat, R., Rousseau, É. MAG-EPA and 17,18-EpETE target cytoplasmic signalling pathways to reduce short-term airway hyperresponsiveness. Pflugers Arch - Eur J Physiol 467, 1591–1605 (2015). https://doi.org/10.1007/s00424-014-1584-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-014-1584-1