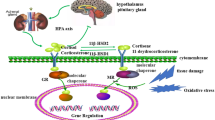
Abstract
Glucocorticoids (GCs) are essential steroid hormones for homeostasis, development, metabolism, and cognition and possess anti-inflammatory and immunosuppressive actions. Since glucocorticoid receptor II (GR) is nearly ubiquitous, chronic activation or depletion of GCs leads to dysfunction of diverse organs, including the heart and blood vessels, resulting predominantly from changes in gene expression. Most studies, therefore, have focused on the genomic effects of GC to understand its related pathophysiological manifestations. The nongenomic effects of GCs clearly differ from well-known genomic effects, with the former responding within several minutes without the need for protein synthesis. There is increasing evidence that the nongenomic actions of GCs influence various physiological functions. To develop a GC-mediated therapeutic target for the treatment of cardiovascular disease, understanding the genomic and nongenomic effects of GC on the cardiovascular system is needed. This article reviews our current understanding of the underlying mechanisms of GCs on cardiovascular diseases and stress, as well as how nongenomic GC signaling contributes to these conditions. We suggest that manipulation of GC action based on both GC and GR metabolism, mitochondrial impact, and the action of serum- and glucocorticoid-dependent kinase 1 may provide new information with which to treat cardiovascular diseases.

Similar content being viewed by others
References
Abrass IB, Scarpace PJ (1981) Glucocorticoid regulation of myocardial beta-adrenergic receptors. Endocrinology 108:977–980
Adigun AA, Wrench N, Seidler FJ, Slotkin TA (2010) Neonatal dexamethasone treatment leads to alterations in cell signaling cascades controlling hepatic and cardiac function in adulthood. Neurotoxicol Teratol 32:193–199. doi:10.1016/j.ntt.2009.10.002
Adzic M, Djordjevic A, Demonacos C, Krstic-Demonacos M, Radojcic MB (2009) The role of phosphorylated glucocorticoid receptor in mitochondrial functions and apoptotic signalling in brain tissue of stressed Wistar rats. Int J Biochem Cell Biol 41:2181–2188. doi:10.1016/j.biocel.2009.04.001
Aoyama T, Matsui T, Novikov M, Park J, Hemmings B, Rosenzweig A (2005) Serum and glucocorticoid-responsive kinase-1 regulates cardiomyocyte survival and hypertrophic response. Circulation 111:1652–1659. doi:10.1161/01.cir.0000160352.58142.06
Bal MP, de Vries WB, van Oosterhout MF, Baan J, van der Wall EE, van Bel F, Steendijk P (2008) Long-term cardiovascular effects of neonatal dexamethasone treatment: hemodynamic follow-up by left ventricular pressure-volume loops in rats. J Appl Physiol 104:446–450. doi:10.1152/japplphysiol.00951.2007
Barnes PJ (2011) Glucocorticosteroids: current and future directions. Br J Pharmacol 163:29–43. doi:10.1111/j.1476-5381.2010.01199.x
Batenburg WW, Jansen PM, van den Bogaerdt AJ, Danser AH (2012) Angiotensin II-aldosterone interaction in human coronary microarteries involves GPR30, EGFR and endothelial NO synthase. Cardiovasc Res 94:136–143. doi:10.1093/cvr/cvs016
Berg JM (1989) DNA binding specificity of steroid receptors. Cell 57:1065–1068
Brostjan C, Anrather J, Csizmadia V, Stroka D, Soares M, Bach FH, Winkler H (1996) Glucocorticoid-mediated repression of NFkappaB activity in endothelial cells does not involve induction of IkappaBalpha synthesis. J Biol Chem 271:19612–19616
Burnstein KL, Cidlowski JA (1992) The down side of glucocorticoid receptor regulation. Mol Cell Endocrinol 83:C1–C8
Busjahn A, Seebohm G, Maier G, Toliat MR, Nurnberg P, Aydin A, Luft FC, Lang F (2004) Association of the serum and glucocorticoid regulated kinase (sgk1) gene with QT interval. Cell Physiol Biochem 14:135–142. doi:10.1159/000078105
Buttgereit F, Scheffold A (2002) Rapid glucocorticoid effects on immune cells. Steroids 67:529–534
Buttgereit F, Straub RH, Wehling M, Burmester GR (2004) Glucocorticoids in the treatment of rheumatic diseases: an update on the mechanisms of action. Arthritis Rheum 50:3408–3417. doi:10.1002/art.20583
Cato AC, Nestl A, Mink S (2002) Rapid actions of steroid receptors in cellular signaling pathways. Sci STKE 2002:re9. doi:10.1126/stke.2002.138.re9
Chae HJ, So HS, Chae SW, Park JS, Kim MS, Oh JM, Chung YT, Yang SH, Jeong ET, Kim HM, Park RK, Kim HR (2001) Sodium nitroprusside induces apoptosis of H9C2 cardiac muscle cells in a c-Jun N-terminal kinase-dependent manner. Int Immunopharmacol 1:967–978
Chai W, Danser AH (2006) Why are mineralocorticoid receptor antagonists cardioprotective? Naunyn Schmiedebergs Arch Pharmacol 374:153–162. doi:10.1007/s00210-006-0107-9
Chai W, Garrelds IM, de Vries R, Batenburg WW, van Kats JP, Danser AH (2005) Nongenomic effects of aldosterone in the human heart: interaction with angiotensin II. Hypertension 46:701–706. doi:10.1161/01.HYP.0000182661.98259.4f
Chai W, Hofland J, Jansen PM, Garrelds IM, de Vries R, van den Bogaerdt AJ, Feelders RA, de Jong FH, Danser AH (2010) Steroidogenesis vs. steroid uptake in the heart: do corticosteroids mediate effects via cardiac mineralocorticoid receptors? J Hypertens 28:1044–1053. doi:10.1097/HJH.0b013e328335c381
Charmandari E, Kino T (2007) Novel causes of generalized glucocorticoid resistance. Horm Metab Res 39:445–450. doi:10.1055/s-2007-980196
Chrousos GP, Kino T (2005) Intracellular glucocorticoid signaling: a formerly simple system turns stochastic. Sci STKE 2005:pe48. doi:10.1126/stke.3042005pe48
Clark AF, Tandler B, Vignos PJ Jr (1982) Glucocorticoid-induced alterations in the rabbit heart. Lab Invest 47:603–610
Clerico A, Giannoni A, Vittorini S, Passino C (2011) Thirty years of the heart as an endocrine organ: physiological role and clinical utility of cardiac natriuretic hormones. Am J Physiol Heart Circ Physiol 301:H12–H20. doi:10.1152/ajpheart.00226.2011
Coghlan MJ, Jacobson PB, Lane B, Nakane M, Lin CW, Elmore SW, Kym PR, Luly JR, Carter GW, Turner R, Tyree CM, Hu J, Elgort M, Rosen J, Miner JN (2003) A novel antiinflammatory maintains glucocorticoid efficacy with reduced side effects. Mol Endocrinol 17:860–869. doi:10.1210/me.2002-0355
Cole TJ, Myles K, Purton JF, Brereton PS, Solomon NM, Godfrey DI, Funder JW (2001) GRKO mice express an aberrant dexamethasone-binding glucocorticoid receptor, but are profoundly glucocorticoid resistant. Mol Cell Endocrinol 173:193–202
Croxtall JD, van Hal PT, Choudhury Q, Gilroy DW, Flower RJ (2002) Different glucocorticoids vary in their genomic and non-genomic mechanism of action in A549 cells. Br J Pharmacol 135:511–519. doi:10.1038/sj.bjp.0704474
De Bosscher K, Beck IM, Haegeman G (2010) Classic glucocorticoids versus non-steroidal glucocorticoid receptor modulators: survival of the fittest regulator of the immune system? Brain Behav Immun 24:1035–1042. doi:10.1016/j.bbi.2010.06.010
Dec;18(6):505-14. CC, in Aov-iCebnic, myocytes. v, Grégoire G PP, Loirand G., Laboratoire de Physiologie FdmVP, Université de, Bordeaux II F, a SopvmwnNitpo, voltage-dependent Ca2+ channel blocker eatiit, concentration of free cytosolic Ca2+ dti, 4,5-trisphosphate, mediated Ca2+ release fbaoaCepC, various p-cai-mwhtteo, in paotCefN-iCr, inhibitor otdtmiOtgc, stimulation. L-sitmCedN, Under TiwrbdcD-co-bc, control conditions aoD-cttesww, but eTacedtiCs, did not evoke Ca2+ entry in venous myocytes under control conditions. However, or aoD-coNaCsdibc, The tcariCibaoaCep, protein eocstipsc-a, results kiK-aH-btN-iCeO, NA tsttaotv-iCeb, cGMP. iaiic
Demonacos C, Djordjevic-Markovic R, Tsawdaroglou N, Sekeris CE (1995) The mitochondrion as a primary site of action of glucocorticoids: the interaction of the glucocorticoid receptor with mitochondrial DNA sequences showing partial similarity to the nuclear glucocorticoid responsive elements. J Steroid Biochem Mol Biol 55:43–55
Demonacos C, Tsawdaroglou NC, Djordjevic-Markovic R, Papalopoulou M, Galanopoulos V, Papadogeorgaki S, Sekeris CE (1993) Import of the glucocorticoid receptor into rat liver mitochondria in vivo and in vitro. J Steroid Biochem Mol Biol 46:401–413
Du J, Wang Y, Hunter R, Wei Y, Blumenthal R, Falke C, Khairova R, Zhou R, Yuan P, Machado-Vieira R, McEwen BS, Manji HK (2009) Dynamic regulation of mitochondrial function by glucocorticoids. Proc Natl Acad Sci U S A 106:3543–3548. doi:10.1073/pnas.0812671106
Falkenstein E, Wehling M (2000) Nongenomically initiated steroid actions. Eur J Clin Invest 30(Suppl 3):51–54
Funder JW (1997) Glucocorticoid and mineralocorticoid receptors: biology and clinical relevance. Annu Rev Med 48:231–240. doi:10.1146/annurev.med.48.1.231
Funder JW (2005) Mineralocorticoid receptors: distribution and activation. Heart Fail Rev 10:15–22. doi:10.1007/s10741-005-2344-2
Funder JW, Duval D, Meyer P (1973) Cardiac glucocorticoid receptors: the binding of tritiated dexamethasone in rat and dog heart. Endocrinology 93:1300–1308
Gagliardi L, Ho JT, Torpy DJ (2010) Corticosteroid-binding globulin: the clinical significance of altered levels and heritable mutations. Mol Cell Endocrinol 316:24–34. doi:10.1016/j.mce.2009.07.015
Ginty AT, Phillips AC, Roseboom TJ, Carroll D, Derooij SR (2012) Cardiovascular and cortisol reactions to acute psychological stress and cognitive ability in the Dutch Famine Birth Cohort Study. Psychophysiology 49:391–400. doi:10.1111/j.1469-8986.2011.01316.x
Girod JP, Brotman DJ (2004) Does altered glucocorticoid homeostasis increase cardiovascular risk? Cardiovasc Res 64:217–226. doi:10.1016/j.cardiores.2004.07.006
Gomez-Sanchez CE, Gomez-Sanchez EP (2001) Editorial: cardiac steroidogenesis—new sites of synthesis, or much ado about nothing? J Clin Endocrinol Metab 86:5118–5120
Hafezi-Moghadam A, Simoncini T, Yang Z, Limbourg FP, Plumier JC, Rebsamen MC, Hsieh CM, Chui DS, Thomas KL, Prorock AJ, Laubach VE, Moskowitz MA, French BA, Ley K, Liao JK (2002) Acute cardiovascular protective effects of corticosteroids are mediated by non-transcriptional activation of endothelial nitric oxide synthase. Nat Med 8:473–479. doi:10.1038/nm0502-473
Haller J, Mikics E, Makara GB (2008) The effects of non-genomic glucocorticoid mechanisms on bodily functions and the central neural system. A critical evaluation of findings. Front Neuroendocrinol 29:273–291. doi:10.1016/j.yfrne.2007.10.004
Hammerman H, Schoen FJ, Braunwald E, Kloner RA (1984) Drug-induced expansion of infarct: morphologic and functional correlations. Circulation 69:611–617
Hartog M, Joplin GF (1968) Effects of cortisol deficiency on the electrocardiogram. Br Med J 2:275–277
Hedman E, Widen C, Asadi A, Dinnetz I, Schroder WP, Gustafsson JA, Wikstrom AC (2006) Proteomic identification of glucocorticoid receptor interacting proteins. Proteomics 6:3114–3126. doi:10.1002/pmic.200500266
Henke G, Setiawan I, Bohmer C, Lang F (2002) Activation of Na+/K+-ATPase by the serum and glucocorticoid-dependent kinase isoforms. Kidney Blood Press Res 25:370–374. doi:10.1159/000068699
Hinds TD Jr, Sanchez ER (2008) Protein phosphatase 5. Int J Biochem Cell Biol 40:2358–2362. doi:10.1016/j.biocel.2007.08.010
Hua SY, Chen YZ (1989) Membrane receptor-mediated electrophysiological effects of glucocorticoid on mammalian neurons. Endocrinology 124:687–691
Ismaili N, Garabedian MJ (2004) Modulation of glucocorticoid receptor function via phosphorylation. Ann N Y Acad Sci 1024:86–101. doi:10.1196/annals.1321.007
Katyare SS, Balasubramanian S, Parmar DV (2003) Effect of corticosterone treatment on mitochondrial oxidative energy metabolism in developing rat brain. Exp Neurol 183:241–248
Kayes-Wandover KM, White PC (2000) Steroidogenic enzyme gene expression in the human heart. J Clin Endocrinol Metab 85:2519–2525
Kewalramani G, Puthanveetil P, Kim MS, Wang F, Lee V, Hau N, Beheshti E, Ng N, Abrahani A, Rodrigues B (2008) Acute dexamethasone-induced increase in cardiac lipoprotein lipase requires activation of both Akt and stress kinases. Am J Physiol Endocrinol Metab 295:E137–E147. doi:10.1152/ajpendo.00004.2008
Kfir-Erenfeld S, Sionov RV, Spokoini R, Cohen O, Yefenof E (2010) Protein kinase networks regulating glucocorticoid-induced apoptosis of hematopoietic cancer cells: fundamental aspects and practical considerations. Leuk Lymphoma 51:1968–2005. doi:10.3109/10428194.2010.506570
Klusonova P, Rehakova L, Borchert G, Vagnerova K, Neckar J, Ergang P, Miksik I, Kolar F, Pacha J (2009) Chronic intermittent hypoxia induces 11beta-hydroxysteroid dehydrogenase in rat heart. Endocrinology 150:4270–4277. doi:10.1210/en.2008-1493
Kohn JA, Deshpande K, Ortlund EA (2012) Deciphering modern glucocorticoid cross-pharmacology using ancestral corticosteroid receptors. J Biol Chem. doi:10.1074/jbc.M112.346411
Konishi A, Tazawa C, Miki Y, Darnel AD, Suzuki T, Ohta Y, Tabayashi K, Sasano H (2003) The possible roles of mineralocorticoid receptor and 11beta-hydroxysteroid dehydrogenase type 2 in cardiac fibrosis in the spontaneously hypertensive rat. J Steroid Biochem Mol Biol 85:439–442
Krstic MD, Rogatsky I, Yamamoto KR, Garabedian MJ (1997) Mitogen-activated and cyclin-dependent protein kinases selectively and differentially modulate transcriptional enhancement by the glucocorticoid receptor. Mol Cell Biol 17:3947–3954
Lackner C, Daufeldt S, Wildt L, Allera A (1998) Glucocorticoid-recognizing and -effector sites in rat liver plasma membrane. Kinetics of corticosterone uptake by isolated membrane vesicles. III. Specificity and stereospecificity. J Steroid Biochem Mol Biol 64:69–82
Lang F, Bohmer C, Palmada M, Seebohm G, Strutz-Seebohm N, Vallon V (2006) (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol Rev 86:1151–1178. doi:10.1152/physrev.00050.2005
Lang F, Cohen P (2001) Regulation and physiological roles of serum- and glucocorticoid-induced protein kinase isoforms. Sci STKE 2001:re17. doi:10.1126/stke.2001.108.re17
Lefer AM (1968) Influence of corticosteroids on mechanical performance of isolated rat papillary muscles. Am J Physiol 214:518–524
Lewis JG, Bagley CJ, Elder PA, Bachmann AW, Torpy DJ (2005) Plasma free cortisol fraction reflects levels of functioning corticosteroid-binding globulin. Clin Chim Acta 359:189–194. doi:10.1016/j.cccn.2005.03.044
Libby P, Maroko PR, Bloor CM, Sobel BE, Braunwald E (1973) Reduction of experimental myocardial infarct size by corticosteroid administration. J Clin Invest 52:599–607. doi:10.1172/jci107221
Limbourg FP, Liao JK (2003) Nontranscriptional actions of the glucocorticoid receptor. J Mol Med (Berl) 81:168–174. doi:10.1007/s00109-003-0418-y
Liu SJ, Wyeth RP, Melchert RB, Kennedy RH (2000) Aging-associated changes in whole cell K+ and L-type Ca2+ currents in rat ventricular myocytes. Am J Physiol Heart Circ Physiol 279:H889–H900
Losel RM, Falkenstein E, Feuring M, Schultz A, Tillmann HC, Rossol-Haseroth K, Wehling M (2003) Nongenomic steroid action: controversies, questions, and answers. Physiol Rev 83:965–1016. doi:10.1152/physrev.00003.2003
Losel R, Wehling M (2003) Nongenomic actions of steroid hormones. Nat Rev Mol Cell Biol 4:46–56. doi:10.1038/nrm1009
Lu NZ, Cidlowski JA (2004) The origin and functions of multiple human glucocorticoid receptor isoforms. Ann N Y Acad Sci 1024:102–123. doi:10.1196/annals.1321.008
Matsubara H (1998) Pathophysiological role of angiotensin II type 2 receptor in cardiovascular and renal diseases. Circ Res 83:1182–1191
McInnes KJ, Kenyon CJ, Chapman KE, Livingstone DE, Macdonald LJ, Walker BR, Andrew R (2004) 5alpha-reduced glucocorticoids, novel endogenous activators of the glucocorticoid receptor. J Biol Chem 279:22908–22912. doi:10.1074/jbc.M402822200
McMaster A, Ray DW (2007) Modelling the glucocorticoid receptor and producing therapeutic agents with anti-inflammatory effects but reduced side-effects. Exp Physiol 92:299–309. doi:10.1113/expphysiol.2006.036194
Meares GP, Zmijewska AA, Jope RS (2004) Heat shock protein-90 dampens and directs signaling stimulated by insulin-like growth factor-1 and insulin. FEBS Lett 574:181–186. doi:10.1016/j.febslet.2004.08.026
Miner JN, Hong MH, Negro-Vilar A (2005) New and improved glucocorticoid receptor ligands. Expert Opin Investig Drugs 14:1527–1545. doi:10.1517/13543784.14.12.1527
Miyamoto S, Murphy AN, Brown JH (2009) Akt mediated mitochondrial protection in the heart: metabolic and survival pathways to the rescue. J Bioenerg Biomembr 41:169–180. doi:10.1007/s10863-009-9205-y
Molnar GA, Lindschau C, Dubrovska G, Mertens PR, Kirsch T, Quinkler M, Gollasch M, Wresche S, Luft FC, Muller DN, Fiebeler A (2008) Glucocorticoid-related signaling effects in vascular smooth muscle cells. Hypertension 51:1372–1378. doi:10.1161/hypertensionaha.107.105718
Morin C, Zini R, Simon N, Charbonnier P, Tillement JP, Le Louet H (2000) Low glucocorticoid concentrations decrease oxidative phosphorylation of isolated rat brain mitochondria: an additional effect of dexamethasone. Fundam Clin Pharmacol 14:493–500
Mune T, Rogerson FM, Nikkila H, Agarwal AK, White PC (1995) Human hypertension caused by mutations in the kidney isozyme of 11 beta-hydroxysteroid dehydrogenase. Nat Genet 10:394–399. doi:10.1038/ng0895-394
Narayanan N, Yang C, Xu A (2004) Dexamethasone treatment improves sarcoplasmic reticulum function and contractile performance in aged myocardium. Mol Cell Biochem 266:31–36
Nussinovitch U, de Carvalho JF, Pereira RM, Shoenfeld Y (2010) Glucocorticoids and the cardiovascular system: state of the art. Curr Pharm Des 16:3574–3585
Odermatt A, Arnold P, Frey FJ (2001) The intracellular localization of the mineralocorticoid receptor is regulated by 11beta-hydroxysteroid dehydrogenase type 2. J Biol Chem 276:28484–28492. doi:10.1074/jbc.M100374200
Ohtani T, Mano T, Hikoso S, Sakata Y, Nishio M, Takeda Y, Otsu K, Miwa T, Masuyama T, Hori M, Yamamoto K (2009) Cardiac steroidogenesis and glucocorticoid in the development of cardiac hypertrophy during the progression to heart failure. J Hypertens 27:1074–1083. doi:10.1097/HJH.0b013e328326cb04
Okuda M, Young KR Jr, Lefer AM (1976) Localization of glucocorticoid uptake in normal and ischemic myocardial tissue of isolated perfused cat hearts. Circ Res 39:640–646
Penefsky ZJ, Kahn M (1971) Inotropic effects of dexamethasone in mammalian heart muscle. Eur J Pharmacol 15:259–266
Perogamvros I, Underhill C, Henley DE, Hadfield KD, Newman WG, Ray DW, Lightman SL, Hammond GL, Trainer PJ (2010) Novel corticosteroid-binding globulin variant that lacks steroid binding activity. J Clin Endocrinol Metab 95:E142–E150. doi:10.1210/jc.2010-0746
Psarra AM, Hermann S, Panayotou G, Spyrou G (2009) Interaction of mitochondrial thioredoxin with glucocorticoid receptor and NF-kappaB modulates glucocorticoid receptor and NF-kappaB signalling in HEK-293 cells. Biochem J 422:521–531. doi:10.1042/bj20090107
Psarra AM, Sekeris CE (2008) Steroid and thyroid hormone receptors in mitochondria. IUBMB Life 60:210–223. doi:10.1002/iub.37
Puthanveetil P, Wang Y, Wang F, Kim MS, Abrahani A, Rodrigues B (2010) The increase in cardiac pyruvate dehydrogenase kinase-4 after short-term dexamethasone is controlled by an Akt-p38-forkhead box other factor-1 signaling axis. Endocrinology 151:2306–2318. doi:10.1210/en.2009-1072
Radomski MW, Palmer RM, Moncada S (1990) Glucocorticoids inhibit the expression of an inducible, but not the constitutive, nitric oxide synthase in vascular endothelial cells. Proc Natl Acad Sci U S A 87:10043–10047
Rao MK, Xu A, Narayanan N (2001) Glucocorticoid modulation of protein phosphorylation and sarcoplasmic reticulum function in rat myocardium. Am J Physiol Heart Circ Physiol 281:H325–H333
Reichardt HM, Tuckermann JP, Gottlicher M, Vujic M, Weih F, Angel P, Herrlich P, Schutz G (2001) Repression of inflammatory responses in the absence of DNA binding by the glucocorticoid receptor. EMBO J 20:7168–7173. doi:10.1093/emboj/20.24.7168
Reuter KC, Loitsch SM, Dignass AU, Steinhilber D, Stein J (2012) Selective non-steroidal glucocorticoid receptor agonists attenuate inflammation but do not impair intestinal epithelial cell restitution in vitro. PLoS One 7:e29756. doi:10.1371/journal.pone.0029756
Revollo JR, Cidlowski JA (2009) Mechanisms generating diversity in glucocorticoid receptor signaling. Ann N Y Acad Sci 1179:167–178. doi:10.1111/j.1749-6632.2009.04986.x
Rivers C, Levy A, Hancock J, Lightman S, Norman M (1999) Insertion of an amino acid in the DNA-binding domain of the glucocorticoid receptor as a result of alternative splicing. J Clin Endocrinol Metab 84:4283–4286
Rosen J, Miner JN (2005) The search for safer glucocorticoid receptor ligands. Endocr Rev 26:452–464. doi:10.1210/er.2005-0002
Rubin JM, Hidalgo A, Bordallo C, Cantabrana B, Sanchez M (1999) Positive inotropism induced by androgens in isolated left atrium of rat: evidence for a cAMP-dependent transcriptional mechanism. Life Sci 65:1035–1045
Sambhi MP, Weil MH, Udhoji VN (1965) Acute pharmacodynamic effects of glucocorticoids; cardiac output and related hemodynamic changes in normal subjects and patients in shock. Circulation 31:523–530
Sapolsky RM, Romero LM, Munck AU (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev 21:55–89
Schacke H, Docke WD, Asadullah K (2002) Mechanisms involved in the side effects of glucocorticoids. Pharmacol Ther 96:23–43
Schmidt BM, Gerdes D, Feuring M, Falkenstein E, Christ M, Wehling M (2000) Rapid, nongenomic steroid actions: a new age? Front Neuroendocrinol 21:57–94. doi:10.1006/frne.1999.0189
Schoneveld JL, Fritsch-Stork RD, Bijlsma JW (2011) Nongenomic glucocorticoid signaling: new targets for immunosuppressive therapy? Arthritis Rheum 63:3665–3667. doi:10.1002/art.30635
Sellevold OF, Jynge P (1989) Bell-shaped concentration-response curve for myocardial stimulation by glucocorticoids. An experimental study in the rat. Acta Anaesthesiol Scand 33:61–65
Seri I, Tan R, Evans J (2001) Cardiovascular effects of hydrocortisone in preterm infants with pressor-resistant hypotension. Pediatrics 107:1070–1074
Shaltout HA, Rose JC, Figueroa JP, Chappell MC, Diz DI, Averill DB (2010) Acute AT(1)-receptor blockade reverses the hemodynamic and baroreflex impairment in adult sheep exposed to antenatal betamethasone. Am J Physiol Heart Circ Physiol 299:H541–H547. doi:10.1152/ajpheart.00100.2010
Shivaji S, Jagannadham MV (1992) Steroid-induced perturbations of membranes and its relevance to sperm acrosome reaction. Biochim Biophys Acta 1108:99–109
Silvestre JS, Robert V, Heymes C, Aupetit-Faisant B, Mouas C, Moalic JM, Swynghedauw B, Delcayre C (1998) Myocardial production of aldosterone and corticosterone in the rat. Physiological regulation. J Biol Chem 273:4883–4891
Simon N, Jolliet P, Morin C, Zini R, Urien S, Tillement JP (1998) Glucocorticoids decrease cytochrome c oxidase activity of isolated rat kidney mitochondria. FEBS Lett 435:25–28
Song IH, Buttgereit F (2006) Non-genomic glucocorticoid effects to provide the basis for new drug developments. Mol Cell Endocrinol 246:142–146. doi:10.1016/j.mce.2005.11.012
Spath JA Jr, Lane DL, Lefer AM (1974) Protective action of methylprednisolone on the myocardium during experimental myocardial ischemia in the cat. Circ Res 35:44–51
Stahn C, Buttgereit F (2008) Genomic and nongenomic effects of glucocorticoids. Nat Clin Pract Rheumatol 4:525–533. doi:10.1038/ncprheum0898
Steiner A, Locher R, Sachinidis A, Vetter W (1989) Cortisol-stimulated phosphoinositide metabolism in vascular smooth muscle cells: a role for glucocorticoids in blood pressure control? J Hypertens Suppl 7:S140–S141
Steiner A, Vogt E, Locher R, Vetter W (1988) Stimulation of the phosphoinositide signalling system as a possible mechanism for glucocorticoid action in blood pressure control. J Hypertens Suppl 6:S366–S368
Talaber G, Boldizsar F, Bartis D, Palinkas L, Szabo M, Berta G, Setalo G Jr, Nemeth P, Berki T (2009) Mitochondrial translocation of the glucocorticoid receptor in double-positive thymocytes correlates with their sensitivity to glucocorticoid-induced apoptosis. Int Immunol 21:1269–1276. doi:10.1093/intimm/dxp093
Tanz RD, Kerby CF (1961) The inotropic action of certain steroids upon isolated cardiac tissue; with comments on steroidal cardiotonic structure-activity relationships. J Pharmacol Exp Ther 131:56–64
Taves MD, Gomez-Sanchez CE, Soma KK (2011) Extra-adrenal glucocorticoids and mineralocorticoids: evidence for local synthesis, regulation, and function. Am J Physiol Endocrinol Metab 301:E11–E24. doi:10.1152/ajpendo.00100.2011
Thiemermann C (2002) Corticosteroids and cardioprotection. Nat Med 8:453–455. doi:10.1038/nm0502-453
Tomlinson JW, Walker EA, Bujalska IJ, Draper N, Lavery GG, Cooper MS, Hewison M, Stewart PM (2004) 11beta-hydroxysteroid dehydrogenase type 1: a tissue-specific regulator of glucocorticoid response. Endocr Rev 25:831–866. doi:10.1210/er.2003-0031
Waldegger S, Barth P, Raber G, Lang F (1997) Cloning and characterization of a putative human serine/threonine protein kinase transcriptionally modified during anisotonic and isotonic alterations of cell volume. Proc Natl Acad Sci U S A 94:4440–4445
Wallerath T, Witte K, Schafer SC, Schwarz PM, Prellwitz W, Wohlfart P, Kleinert H, Lehr HA, Lemmer B, Forstermann U (1999) Down-regulation of the expression of endothelial NO synthase is likely to contribute to glucocorticoid-mediated hypertension. Proc Natl Acad Sci U S A 96:13357–13362
Wang Z, Chen W, Kono E, Dang T, Garabedian MJ (2007) Modulation of glucocorticoid receptor phosphorylation and transcriptional activity by a C-terminal-associated protein phosphatase. Mol Endocrinol 21:625–634. doi:10.1210/me.2005-0338
Weigel NL, Moore NL (2007) Steroid receptor phosphorylation: a key modulator of multiple receptor functions. Mol Endocrinol 21:2311–2319. doi:10.1210/me.2007-0101
Whitworth JA (1994) Studies on the mechanisms of glucocorticoid hypertension in humans. Blood Press 3:24–32
Whitworth JA, Kelly JJ, Brown MA, Williamson PM, Lawson JA (1997) Glucocorticoids and hypertension in man. Clin Exp Hypertens 19:871–884
Xue Q, Dasgupta C, Chen M, Zhang L (2011) Foetal hypoxia increases cardiac AT(2)R expression and subsequent vulnerability to adult ischaemic injury. Cardiovasc Res 89:300–308. doi:10.1093/cvr/cvq303
Yang S, Zhang L (2004) Glucocorticoids and vascular reactivity. Curr Vasc Pharmacol 2:1–12
Yang M, Zheng J, Miao Y, Wang Y, Cui W, Guo J, Qiu S, Han Y, Jia L, Li H, Cheng J, Du J (2012) Serum-glucocorticoid regulated kinase 1 regulates alternatively activated macrophage polarization contributing to angiotensin II-induced inflammation and cardiac fibrosis. Arterioscler Thromb Vasc Biol 32:1675–1686. doi:10.1161/atvbaha.112.248732
Yano K, Tsuda Y, Kaji Y, Kanaya S, Fujino T, Niho Y (1994) Effects of hydrocortisone on transmembrane currents in guinea pig ventricular myocytes—possible evidence for positive inotropism. Jpn Circ J 58:836–843
Young MJ, Funder JW (1996) The renin-angiotensin-aldosterone system in experimental mineralocorticoid-salt-induced cardiac fibrosis. Am J Physiol 271:E883–E888
Yudt MR, Jewell CM, Bienstock RJ, Cidlowski JA (2003) Molecular origins for the dominant negative function of human glucocorticoid receptor beta. Mol Cell Biol 23:4319–4330
Zecevic M, Heitzmann D, Camargo SM, Verrey F (2004) SGK1 increases Na, K-ATP cell-surface expression and function in Xenopus laevis oocytes. Pflugers Arch 448:29–35. doi:10.1007/s00424-003-1222-9
Zhou J, Li M, Sheng CQ, Liu L, Li Z, Wang Y, Zhou JR, Jing ZP, Chen YZ, Jiang CL (2011) A novel strategy for development of glucocorticoids through non-genomic mechanism. Cell Mol Life Sci 68:1405–1414. doi:10.1007/s00018-010-0526-0
Zietz B, Hrach S, Scholmerich J, Straub RH (2001) Differential age-related changes of hypothalamus-pituitary-adrenal axis hormones in healthy women and men—role of interleukin 6. Exp Clin Endocrinol Diabetes 109:93–101. doi:10.1055/s-2001-14833
Zuo Z, Urban G, Scammell JG, Dean NM, McLean TK, Aragon I, Honkanen RE (1999) Ser/Thr protein phosphatase type 5 (PP5) is a negative regulator of glucocorticoid receptor-mediated growth arrest. Biochemistry 38:8849–8857. doi:10.1021/bi990842e
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2010-0020224, 2010-0025855, and 2012-007595).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Nari Kim and Jin Han contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lee, S.R., Kim, H.K., Youm, J.B. et al. Non-genomic effect of glucocorticoids on cardiovascular system. Pflugers Arch - Eur J Physiol 464, 549–559 (2012). https://doi.org/10.1007/s00424-012-1155-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-012-1155-2