Abstract
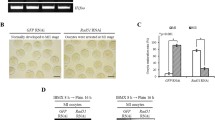
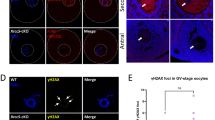
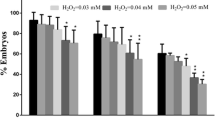
The maintenance of genomic integrity and stability is essential for the survival of every organism. Unfortunately, DNA is vulnerable to attack by a variety of damaging agents. Oxidative stress is a major cause of DNA damage because reactive oxygen species (ROS) are produced as by-products of normal cellular metabolism. Cells have developed eloquent antioxidant defense systems to protect themselves from oxidative damage along with aerobic metabolism. Here, we show that catalase (CAT) is present in mouse oocytes to protect the genome from oxidative damage during meiotic maturation. CAT was expressed in the nucleus to form unique vesicular structures. However, after nuclear envelope breakdown, CAT was redistributed in the cytoplasm with particular focus at the chromosomes. Inhibition of CAT activity increased endogenous ROS levels, but did not perturb meiotic maturation. In addition, CAT inhibition produced chromosomal defects, including chromosome misalignment and DNA damage. Therefore, our data suggest that CAT is required not only to scavenge ROS, but also to protect DNA from oxidative damage during meiotic maturation in mouse oocytes.



Similar content being viewed by others
Abbreviations
- ROS:
-
Reactive oxygen species
- DDR:
-
DNA damage response
- CAT:
-
Catalase
- SOD:
-
Superoxide dismutase
- GPX:
-
Glutathione peroxidase
- 3-AT:
-
3-amino-1,2,4-triazole
- PMSG:
-
Pregnant mare’s serum gonadotropin
- IBMX:
-
3-Isobutyl-1-methylxanthine
- DHR-123:
-
Dihydrorhodamine 123
- DSB:
-
DNA double-strand break
References
Ali AA, Bilodeau JF, Sirard MA (2003) Antioxidant requirements for bovine oocytes varies during in vitro maturation, fertilization and development. Theriogenology 59(3–4):939–949
Angermuller S, Islinger M, Volkl A (2009) Peroxisomes and reactive oxygen species, a lasting challenge. Histochem Cell Biol 131(4):459–463. doi:10.1007/s00418-009-0563-7
Brigelius-Flohe R (1999) Tissue-specific functions of individual glutathione peroxidases. Free Radic Biol Med 27(9–10):951–965
Bulitta C, Ganea C, Fahimi HD, Volkl A (1996) Cytoplasmic and peroxisomal catalases of the guinea pig liver: evidence for two distinct proteins. Biochim Biophys Acta 1293(1):55–62
Carbone MC, Tatone C, Delle Monache S, Marci R, Caserta D, Colonna R, Amicarelli F (2003) Antioxidant enzymatic defences in human follicular fluid: characterization and age-dependent changes. Mol Hum Reprod 9(11):639–643
Chang JY, Schroeder WA (1972) Reaction of 3-amino-1:2:4-triazole with bovine liver catalase and human erythrocyte catalase. Arch Biochem Biophys 148(2):505–508
Chelikani P, Fita I, Loewen PC (2004) Diversity of structures and properties among catalases. Cell Mol Life Sci 61(2):192–208. doi:10.1007/s00018-003-3206-5
Choi WJ, Banerjee J, Falcone T, Bena J, Agarwal A, Sharma RK (2007) Oxidative stress and tumor necrosis factor-alpha-induced alterations in metaphase II mouse oocyte spindle structure. Fertil Steril 88(4 Suppl):1220–1231. doi:10.1016/j.fertnstert.2007.02.067
Ciccia A, Elledge SJ (2010) The DNA damage response: making it safe to play with knives. Mol Cell 40(2):179–204. doi:10.1016/j.molcel.2010.09.019
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J 17(10):1195–1214. doi:10.1096/fj.02-0752rev
Dalton CM, Carroll J (2013) Biased inheritance of mitochondria during asymmetric cell division in the mouse oocyte. J Cell Sci 126(Pt 13):2955–2964. doi:10.1242/jcs.128744
de Matos DG, Furnus CC (2000) The importance of having high glutathione (GSH) level after bovine in vitro maturation on embryo development effect of beta-mercaptoethanol, cysteine and cystine. Theriogenology 53(3):761–771. doi:10.1016/S0093-691X(99)00278-2
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82(1):47–95. doi:10.1152/physrev.00018.2001
El Mouatassim S, Guerin P, Menezo Y (1999) Expression of genes encoding antioxidant enzymes in human and mouse oocytes during the final stages of maturation. Mol Hum Reprod 5(8):720–725
Eppig JJ, Hosoe M, O’Brien MJ, Pendola FM, Requena A, Watanabe S (2000) Conditions that affect acquisition of developmental competence by mouse oocytes in vitro: FSH, insulin, glucose and ascorbic acid. Mol Cell Endocrinol 163(1–2):109–116
Gupta S, Choi A, Yu HY, Czerniak SM, Holick EA, Paolella LJ, Agarwal A, Combelles CM (2011) Fluctuations in total antioxidant capacity, catalase activity and hydrogen peroxide levels of follicular fluid during bovine folliculogenesis. Reprod Fertil Dev 23(5):673–680. doi:10.1071/RD10270
Harvey MB, Arcellana-Panlilio MY, Zhang X, Schultz GA, Watson AJ (1995) Expression of genes encoding antioxidant enzymes in preimplantation mouse and cow embryos and primary bovine oviduct cultures employed for embryo coculture. Biol Reprod 53(3):532–540
Hensley K, Robinson KA, Gabbita SP, Salsman S, Floyd RA (2000) Reactive oxygen species, cell signaling, and cell injury. Free Radic Biol Med 28(10):1456–1462
Hossein MS, Kim MK, Jang G, Oh HJ, Koo O, Kim JJ, Kang SK, Lee BC, Hwang WS (2007) Effects of thiol compounds on in vitro maturation of canine oocytes collected from different reproductive stages. Mol Reprod Dev 74(9):1213–1220. doi:10.1002/mrd.20674
Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461(7267):1071–1078. doi:10.1038/nature08467
Jacquet P, Adriaens I, Buset J, Neefs M, Vankerkom J (2005) Cytogenetic studies in mouse oocytes irradiated in vitro at different stages of maturation, by use of an early preantral follicle culture system. Mutat Res 583(2):168–177. doi:10.1016/j.mrgentox.2005.03.008
Johnson F, Giulivi C (2005) Superoxide dismutases and their impact upon human health. Mol Aspects Med 26(4–5):340–352. doi:10.1016/j.mam.2005.07.006
Luberda Z (2005) The role of glutathione in mammalian gametes. Reprod Biol 5(1):5–17
Luciano AM, Goudet G, Perazzoli F, Lahuec C, Gerard N (2006) Glutathione content and glutathione peroxidase expression in in vivo and in vitro matured equine oocytes. Mol Reprod Dev 73(5):658–666. doi:10.1002/mrd.20469
Marangos P, Carroll J (2012) Oocytes progress beyond prophase in the presence of DNA damage. Curr Biol 22(11):989–994. doi:10.1016/j.cub.2012.03.063
Margoliash E, Novogrodsky A (1958) A study of the inhibition of catalase by 3-amino-1:2:4:-triazole. Biochem J 68(3):468–475
Margoliash E, Novogrodsky A, Schejter A (1960) Irreversible reaction of 3-amino-1:2:4-triazole and related inhibitors with the protein of catalase. Biochem J 74:339–348
Masui Y, Clarke HJ (1979) Oocyte maturation. Int Rev Cytol 57:185–282
Mates JM, Perez-Gomez C, Nunez de Castro I (1999) Antioxidant enzymes and human diseases. Clin Biochem 32(8):595–603
Pandey AN, Chaube SK (2014) A moderate increase of hydrogen peroxide level is beneficial for spontaneous resumption of meiosis from diplotene arrest in rat oocytes cultured in vitro. Biores Open Access 3(4):183–191. doi:10.1089/biores.2014.0013
Schieber M, Chandel NS (2014) ROS function in redox signaling and oxidative stress. Curr Biol 24(10):R453–R462. doi:10.1016/j.cub.2014.03.034
Tamura H, Takasaki A, Miwa I, Taniguchi K, Maekawa R, Asada H, Taketani T, Matsuoka A, Yamagata Y, Shimamura K, Morioka H, Ishikawa H, Reiter RJ, Sugino N (2008) Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J Pineal Res 44(3):280–287. doi:10.1111/j.1600-079X.2007.00524.x
Tao Y, Zhou B, Xia G, Wang F, Wu Z, Fu M (2004) Exposure to L-ascorbic acid or alpha-tocopherol facilitates the development of porcine denuded oocytes from metaphase I to metaphase II and prevents cumulus cells from fragmentation. Reprod Domest Anim 39(1):52–57
Tatemoto H, Muto N, Sunagawa I, Shinjo A, Nakada T (2004) Protection of porcine oocytes against cell damage caused by oxidative stress during in vitro maturation: role of superoxide dismutase activity in porcine follicular fluid. Biol Reprod 71(4):1150–1157. doi:10.1095/biolreprod.104.029264
Tease C (1983) X-ray-induced chromosome aberrations in dictyate oocytes of young and old female mice. Mutat Res 119(2):191–194
Van Blerkom J, Runner MN (1984) Mitochondrial reorganization during resumption of arrested meiosis in the mouse oocyte. Am J Anat 171(3):335–355. doi:10.1002/aja.1001710309
Van Remmen H, Ikeno Y, Hamilton M, Pahlavani M, Wolf N, Thorpe SR, Alderson NL, Baynes JW, Epstein CJ, Huang TT, Nelson J, Strong R, Richardson A (2003) Life-long reduction in MnSOD activity results in increased DNA damage and higher incidence of cancer but does not accelerate aging. Physiol Genomics 16(1):29–37. doi:10.1152/physiolgenomics.00122.2003
Whitaker BD, Knight JW (2008) Mechanisms of oxidative stress in porcine oocytes and the role of anti-oxidants. Reprod Fertil Dev 20(6):694–702
Yamamoto K, Volkl A, Hashimoto T, Fahimi HD (1988) Catalase in guinea pig hepatocytes is localized in cytoplasm, nuclear matrix and peroxisomes. Eur J Cell Biol 46(1):129–135
Acknowledgments
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI12C0737), and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2013R1A1A1004766).
Author’s contributions
JO conceived and designed the experiments; Y.P, S.Y and S.C performed the experiments; Y.P and J.O analyzed the data; H. J, S.L, D.C and J.K contributed reagents, materials, analysis and tools; and J.O wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, Y.S., You, S.Y., Cho, S. et al. Eccentric localization of catalase to protect chromosomes from oxidative damages during meiotic maturation in mouse oocytes. Histochem Cell Biol 146, 281–288 (2016). https://doi.org/10.1007/s00418-016-1446-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-016-1446-3