Abstract
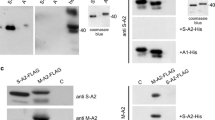
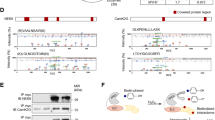
Muscle-specific mechanosensors Ankrd2/Arpp (ankyrin repeat protein 2) and Ankrd1/CARP (cardiac ankyrin repeat protein) have an important role in transcriptional regulation, myofibrillar assembly, cardiogenesis and myogenesis. In skeletal muscle myofibrils, Ankrd2 has a structural role as a component of a titin associated stretch-sensing complex, while in the nucleus it exerts regulatory function as transcriptional co-factor. It is also involved in myogenic differentiation and coordination of myoblast proliferation. Although expressed in the heart, the role of Ankrd2 in the cardiac muscle is completely unknown. Recently, we have shown that hypertrophic and dilated cardiomyopathy pathways are altered upon Ankrd2 silencing suggesting the importance of this protein in cardiac tissue. Here we provide the underlying basis for the functional investigation of Ankrd2 in the heart. We confirmed reduced Ankrd2 expression levels in human heart in comparison with Ankrd1 using RNAseq and Western blot. For the first time we demonstrated that, apart from the sarcomere and nucleus, both proteins are localized to the intercalated disks of human cardiomyocytes. We further tested the expression and localization of endogenous Ankrd2 in rat neonatal cardiomyocytes, a well-established model for studying cardiac-specific proteins. Ankrd2 was found to be expressed in both the cytoplasm and nucleus, independently from maturation status of cardiomyocytes. In contrast to Ankrd1, it is not responsive to the cardiotoxic drug Doxorubicin, suggesting that different mechanisms govern their expression in cardiac cells.







Similar content being viewed by others
References
Aihara Y, Kurabayashi M, Saito Y, Ohyama Y, Tanaka T, Takeda S, Tomaru K, Sekiguchi K, Arai M, Nakamura T, Nagai R (2000a) Cardiac ankyrin repeat protein is a novel marker of cardiac hypertrophy: role of m-cat element within the promoter. Hypertension 36:48–53
Aihara Y, Kurabayashi M, Tanaka T, Takeda SI, Tomaru K, Sekiguchi KI, Ohyama Y, Nagai R (2000b) Doxorubicin represses carp gene transcription through the generation of oxidative stress in neonatal rat cardiac myocytes: possible role of serine/threonine kinase-dependent pathways. J Mol Cell Cardiol 32:1401–1414
Arimura T, Bos JM, Sato A, Kubo T, Okamoto H, Nishi H, Harada H, Koga Y, Moulik M, Doi YL, Towbin JA, Ackerman MJ, Kimura A (2009) Cardiac ankyrin repeat protein gene (ankrd1) mutations in hypertrophic cardiomyopathy. J Am Coll Cardiol 54:334–342
Bang ML, Gu Y, Dalton ND, Peterson KL, Chien KR, Chen J (2014) The muscle ankyrin repeat proteins carp, ankrd2, and darp are not essential for normal cardiac development and function at basal conditions and in response to pressure overload. PLoS One 9:e93638
Barash IA, Mathew L, Ryan AF, Chen J, Lieber RL (2004) Rapid muscle-specific gene expression changes after a single bout of eccentric contractions in the mouse. Am J Physiol Cell Physiol 286:C355–C364
Belgrano A, Rakicevic L, Mittempergher L, Campanaro S, Martinelli VC, Mouly V, Valle G, Kojic S, Faulkner G (2011) Multi-tasking role of the mechanosensing protein ankrd2 in the signaling network of striated muscle. PLoS One 6:e25519
Burkhard P, Stetefeld J, Strelkov SV (2001) Coiled coils: a highly versatile protein folding motif. Trends Cell Biol 11:82–88
Campagna D, Albiero A, Bilardi A, Caniato E, Forcato C, Manavski S, Vitulo N, Valle G (2009) Pass: a program to align short sequences. Bioinformatics 25:967–968
Cenni V, Bavelloni A, Beretti F, Tagliavini F, Manzoli L, Lattanzi G, Maraldi NM, Cocco L, Marmiroli S (2011) Ankrd2/arpp is a novel akt2 specific substrate and regulates myogenic differentiation upon cellular exposure to h(2)o(2). Mol Biol Cell 22:2946–2956
Chen B, Zhong L, Roush SF, Pentassuglia L, Peng X, Samaras S, Davidson JM, Sawyer DB, Lim CC (2012) Disruption of a gata4/ankrd1 signaling axis in cardiomyocytes leads to sarcomere disarray: implications for anthracycline cardiomyopathy. PLoS One 7:e35743
Chlopcíková S, Psotová J, Miketová P (2001) Neonatal rat cardiomyocytes—a model for the study of morphological, biochemical and electrophysiological characteristics of the heart. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 145:49–55
Clark KA, McElhinny AS, Beckerle MC, Gregorio CC (2002) Striated muscle cytoarchitecture: an intricate web of form and function. Annu Rev Cell Dev Biol 18:637–706
Cooper ST, Lo HP, North KN (2003) Single section western blot: improving the molecular diagnosis of the muscular dystrophies. Neurology 61:93–97
Duboscq-Bidot L, Charron P, Ruppert V, Fauchier L, Richter A, Tavazzi L, Arbustini E, Wichter T, Maisch B, Komajda M, Isnard R, Villard E, Network EHF (2009) Mutations in the ankrd1 gene encoding carp are responsible for human dilated cardiomyopathy. Eur Heart J 30:2128–2136
Dwyer J, Pluess M, Iskratsch T, Dos Remedios CG, Ehler E (2014) The formin fhod1 in cardiomyocytes. Anat Rec 297:1560–1570
Eble DM, Cadre BM, Qi M, Bers DM, Samarel AM (1998) Contractile activity modulates atrial natriuretic factor gene expression in neonatal rat ventricular myocytes. J Mol Cell Cardiol 30:55–60
Giepmans BN (2004) Gap junctions and connexin-interacting proteins. Cardiovasc Res 62:233–245
Hayashi C, Ono Y, Doi N, Kitamura F, Tagami M, Mineki R, Arai T, Taguchi H, Yanagida M, Hirner S, Labeit D, Labeit S, Sorimachi H (2008) Multiple molecular interactions implicate the connectin/titin n2a region as a modulating scaffold for p94/calpain 3 activity in skeletal muscle. J Biol Chem 283:14801–14814
Hoshida S, Nishida M, Yamashita N, Igarashi J, Aoki K, Hori M, Kuzuya T, Tada M (1996) Heme oxygenase-1 expression and its relation to oxidative stress during primary culture of cardiomyocytes. J Mol Cell Cardiol 28:1845–1855
Hoshijima M (2006) Mechanical stress–strain sensors embedded in cardiac cytoskeleton: Z disk, titin, and associated structures. Am J Physiol Heart Circ Physiol 290:H1313–H1325
Ishiguro N, Baba T, Ishida T, Takeuchi K, Osaki M, Araki N, Okada E, Takahashi S, Saito M, Watanabe M, Nakada C, Tsukamoto Y, Sato K, Ito K, Fukayama M, Mori S, Ito H, Moriyama M (2002) Carp, a cardiac ankyrin-repeated protein, and its new homologue, arpp, are differentially expressed in heart, skeletal muscle, and rhabdomyosarcomas. Am J Pathol 160:1767–1778
Jeyaseelan R, Poizat C, Baker RK, Abdishoo S, Isterabadi LB, Lyons GE, Kedes L (1997) A novel cardiac-restricted target for doxorubicin. Carp, a nuclear modulator of gene expression in cardiac progenitor cells and cardiomyocytes. J Biol Chem 272:22800–22808
Kemp TJ, Sadusky TJ, Saltisi F, Carey N, Moss J, Yang SY, Sassoon DA, Goldspink G, Coulton GR (2000) Identification of ankrd2, a novel skeletal muscle gene coding for a stretch-responsive ankyrin-repeat protein. Genomics 66:229–241
Kimura A (2010) Molecular basis of hereditary cardiomyopathy: abnormalities in calcium sensitivity, stretch response, stress response and beyond. J Hum Genet 55:81–90
Kojic S, Medeot E, Guccione E, Krmac H, Zara I, Martinelli V, Valle G, Faulkner G (2004) The ankrd2 protein, a link between the sarcomere and the nucleus in skeletal muscle. J Mol Biol 339:313–325
Kojic S, Medeot E, Faulkner G (2009) Characterization of antibodies directed against the ankrd2 human muscle protein. Arch Biol Sci 61:9
Kojic S, Nestorovic A, Rakicevic L, Belgrano A, Stankovic M, Divac A, Faulkner G (2010) A novel role for cardiac ankyrin repeat protein ankrd1/carp as a co-activator of the p53 tumor suppressor protein. Arch Biochem Biophys 502:60–67
Kojic S, Radojkovic D, Faulkner G (2011) Muscle ankyrin repeat proteins: their role in striated muscle function in health and disease. Crit Rev Clin Lab Sci 48:269–294
Kuo H, Chen J, Ruiz-Lozano P, Zou Y, Nemer M, Chien KR (1999) Control of segmental expression of the cardiac-restricted ankyrin repeat protein gene by distinct regulatory pathways in murine cardiogenesis. Development 126:4223–4234
Kurabayashi M, Jeyaseelan R, Kedes L (1993) Antineoplastic agent doxorubicin inhibits myogenic differentiation of c2 myoblasts. J Biol Chem 268:5524–5529
Kurabayashi M, Dutta S, Jeyaseelan R, Kedes L (1995) Doxorubicin-induced id2a gene transcription is targeted at an activating transcription factor/cyclic amp response element motif through novel mechanisms involving protein kinases distinct from protein kinase c and protein kinase a. Mol Cell Biol 15:6386–6397
Martinelli V, Cellot G, Toma FM, Long CS, Caldwell JH, Zentilin L, Giacca M, Turco A, Prato M, Ballerini L, Mestroni L (2012) Carbon nanotubes promote growth and spontaneous electrical activity in cultured cardiac myocytes. Nano Lett 12:1831–1838
Martinelli VC, Kyle WB, Kojic S, Vitulo N, Li Z, Belgrano A, Maiuri P, Banks L, Vatta M, Valle G, Faulkner G (2014) Zasp interacts with the mechanosensing protein ankrd2 and p53 in the signalling network of striated muscle. PLoS One 9:e92259
Miller MK, Bang ML, Witt CC, Labeit D, Trombitas C, Watanabe K, Granzier H, McElhinny AS, Gregorio CC, Labeit S (2003) The muscle ankyrin repeat proteins: carp, ankrd2/arpp and darp as a family of titin filament-based stress response molecules. J Mol Biol 333:951–964
Moriyama M, Tsukamoto Y, Fujiwara M, Kondo G, Nakada C, Baba T, Ishiguro N, Miyazaki A, Nakamura K, Hori N, Sato K, Shomori K, Takeuchi K, Satoh H, Mori S, Ito H (2001) Identification of a novel human ankyrin-repeated protein homologous to carp. Biochem Biophys Res Commun 285:715–723
Moulik M, Vatta M, Witt SH, Arola AM, Murphy RT, McKenna WJ, Boriek AM, Oka K, Labeit S, Bowles NE, Arimura T, Kimura A, Towbin JA (2009) Ankrd1, the gene encoding cardiac ankyrin repeat protein, is a novel dilated cardiomyopathy gene. J Am Coll Cardiol 54:325–333
Nagueh SF, Shah G, Wu Y, Torre-Amione G, King NM, Lahmers S, Witt CC, Becker K, Labeit S, Granzier HL (2004) Altered titin expression, myocardial stiffness, and left ventricular function in patients with dilated cardiomyopathy. Circulation 110:155–162
Nakada C, Tsukamoto Y, Oka A, Nonaka I, Sato K, Mori S, Ito H, Moriyama M (2004) Altered expression of arpp protein in skeletal muscles of patients with muscular dystrophy, congenital myopathy and spinal muscular atrophy. Pathobiology 71:43–51
Nakamura K, Nakada C, Takeuchi K, Osaki M, Shomori K, Kato S, Ohama E, Sato K, Fukayama M, Mori S, Ito H, Moriyama M (2002) Altered expression of cardiac ankyrin repeat protein and its homologue, ankyrin repeat protein with pest and proline-rich region, in atrophic muscles in amyotrophic lateral sclerosis. Pathobiology 70:197–203
Nestorovic A, Jasnic-Savovic J, Faulkner G, Radojkovic D, Kojic S (2014) Ankrd1-mediated signaling is supported by its interaction with zonula occludens-1. Arch Biol Sci 66:10
Pallavicini A, Kojić S, Bean C, Vainzof M, Salamon M, Ievolella C, Bortoletto G, Pacchioni B, Zatz M, Lanfranchi G, Faulkner G, Valle G (2001) Characterization of human skeletal muscle ankrd2. Biochem Biophys Res Commun 285:378–386
Pearson WR, Lipman DJ (1988) Improved tools for biological sequence comparison. Proc Natl Acad Sci USA 85:2444–2448
Rechsteiner M, Rogers SW (1996) Pest sequences and regulation by proteolysis. Trends Biochem Sci 21:267–271
Roberts A, Pimentel H, Trapnell C, Pachter L (2011) Identification of novel transcripts in annotated genomes using rna-seq. Bioinformatics 27:2325–2329
Rogers S, Wells R, Rechsteiner M (1986) Amino acid sequences common to rapidly degraded proteins: the pest hypothesis. Science 234:364–368
Sheikh F, Ross RS, Chen J (2009) Cell–cell connection to cardiac disease. Trends Cardiovasc Med 19:182–190
Singal PK, Deally CM, Weinberg LE (1987) Subcellular effects of adriamycin in the heart: a concise review. J Mol Cell Cardiol 19:817–828
Singal PK, Li T, Kumar D, Danelisen I, Iliskovic N (2000) Adriamycin-induced heart failure: mechanism and modulation. Mol Cell Biochem 207:77–86
Sreejit P, Kumar S, Verma RS (2008) An improved protocol for primary culture of cardiomyocyte from neonatal mice. In Vitro Cell Dev Biol Anim 44:45–50
Tee JM, Peppelenbosch MP (2010) Anchoring skeletal muscle development and disease: the role of ankyrin repeat domain containing proteins in muscle physiology. Crit Rev Biochem Mol Biol 45:318–330
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by rna-seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515
Tsukamoto Y, Senda T, Nakano T, Nakada C, Hida T, Ishiguro N, Kondo G, Baba T, Sato K, Osaki M, Mori S, Ito H, Moriyama M (2002) Arpp, a new homolog of carp, is preferentially expressed in type 1 skeletal muscle fibers and is markedly induced by denervation. Lab Invest 82:645–655
Tsukamoto Y, Hijiya N, Yano S, Yokoyama S, Nakada C, Uchida T, Matsuura K, Moriyama M (2008) Arpp/ankrd2, a member of the muscle ankyrin repeat proteins (marps), translocates from the i-band to the nucleus after muscle injury. Histochem Cell Biol 129:55–64
Wu TD, Nacu S (2010) Fast and snp-tolerant detection of complex variants and splicing in short reads. Bioinformatics 26:873–881
Zhu W, Soonpaa MH, Chen H, Shen W, Payne RM, Liechty EA, Caldwell RL, Shou W, Field LJ (2009) Acute doxorubicin cardiotoxicity is associated with p53-induced inhibition of the mammalian target of rapamycin pathway. Circulation 119:99–106
Zolk O, Frohme M, Maurer A, Kluxen FW, Hentsch B, Zubakov D, Hoheisel JD, Zucker IH, Pepe S, Eschenhagen T (2002) Cardiac ankyrin repeat protein, a negative regulator of cardiac gene expression, is augmented in human heart failure. Biochem Biophys Res Commun 293:1377–1382
Zou Y, Evans S, Chen J, Kuo HC, Harvey RP, Chien KR (1997) Carp, a cardiac ankyrin repeat protein, is downstream in the nkx2-5 homeobox gene pathway. Development 124:793–804
Acknowledgments
The excellent technical assistance of B Anicic is gratefully acknowledged. We would like to thank M Schwirtlich for connexin 43 and β-catenin antibodies. This study was supported by grants from the Ministry of Education and Science of Serbia (Project No. 173008 to DR), Collaborative Research Programme, ICGEB, Italy (grant CRP/YUG-05-01 to SK), Fondazione Cariparo, Italy (Progetto Eccellenza 2010 CHROMUS to GV and GF) and the Italian Ministry of Research (PRIN 20108XYHJS to GV).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All procedures performed in this study involving human participants were in accordance with the ethical standards of the Faculty of Medicine, University of Belgrade, 1964 Helsinki declaration and the Law on Health Care of the Republic of Serbia. For this type of study, formal consent is not required. All applicable international and national guidelines for the care and use of animals were followed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jasnic-Savovic, J., Nestorovic, A., Savic, S. et al. Profiling of skeletal muscle Ankrd2 protein in human cardiac tissue and neonatal rat cardiomyocytes. Histochem Cell Biol 143, 583–597 (2015). https://doi.org/10.1007/s00418-015-1307-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-015-1307-5