Abstract
Purpose
To analyze the structures of schisis in eyes with hereditary juvenile retinoschisis using en-face optical coherence tomography (OCT) imaging.
Methods
In this retrospective observational study, we reviewed the medical records of patients with hereditary juvenile retinoschisis who underwent comprehensive ophthalmic examinations including swept-source OCT.
Results
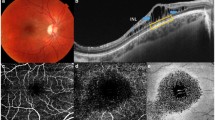
OCT images were obtained from 16 eyes of nine boys (mean age ± standard deviation, 10.6 ± 4.0 years). The horizontal OCT images at the fovea showed inner nuclear layer (INL) schisis in one eye (6.3 %), ganglion cell layer (GCL) and INL schisis in 12 eyes (75.0 %), INL and outer plexiform layer (OPL) schisis in two eyes (12.5 %), and GCL, INL, and OPL schisis in one eye (6.3 %). En-face OCT images showed characteristic schisis patterns in each retinal layer, which were represented by multiple hyporeflective holes in the parafoveal region in the GCL, a spoke-like pattern in the foveal region, a reticular pattern in the parafoveal region in the INL, and multiple hyporeflective polygonal cavities with partitions in the OPL.
Conclusions
Our results using en-face OCT imaging clarified different patterns of schisis formation among the GCL, INL, and OPL, which lead to further recognition of structure in hereditary juvenile retinoschisis.



Similar content being viewed by others
References
Kellner U, Brummer S, Foerster MH, Wessing A (1990) X-linked congenital retinoschisis. Graefes Arch Clin Exp Ophthalmol 228:432–437
George ND, Yates JR, Moore AT (1995) X linked retinoschisis. Br J Ophthalmol 79:697–702
George ND, Yates JR, Moore AT (1996) Clinical features in affected males with X-linked retinoschisis. Arch Ophthalmol 114:274–280
Sauer CG, Gehrig A, Warneke-Wittstock R, Marquardt A, Ewing CC, Gibson A et al (1997) Positional cloning of the gene associated with X-linked juvenile retinoschisis. Nat Genet 17:164–170
Molday RS, Kellner U, Weber BH (2012) X-linked juvenile retinoschisis: clinical diagnosis, genetic analysis, and molecular mechanisms. Prog Retin Eye Res 31:195–212
Choma M, Sarunic M, Yang C, Izatt J (2003) Sensitivity advantage of swept source and Fourier domain optical coherence tomography. Opt Express 11:2183–2189
Gerth C, Zawadzki RJ, Werner JS, Heon E (2008) Retinal morphological changes of patients with X-linked retinoschisis evaluated by Fourier-domain optical coherence tomography. Arch Ophthalmol 126:807–811
Gregori NZ, Berrocal AM, Gregori G, Murray TG, Knighton RW, Flynn HW Jr et al (2009) Macular spectral-domain optical coherence tomography in patients with X-linked retinoschisis. Br J Ophthalmol 93:373–378
Yu J, Ni Y, Keane PA, Jiang C, Wang W, Xu G (2010) Foveomacular schisis in juvenile X-linked retinoschisis: an optical coherence tomography study. Am J Ophthalmol 149(973-978):e972
Liang H, Cid M, Cucu R, Dobre G, Podoleanu A, Pedro J et al (2005) En-face optical coherence tomography — a novel application of non-invasive imaging to art conservation. Opt Express 13:6133–6144
Cozzi M, Pagliarini S (2016) Retinoschisis microstructure visualization with En face spectral domain optical coherence tomography. Retina 36:227–228
Samara WA, Shahlaee A, Sridhar J, Khan MA, Ho AC (2016) En face optical coherence tomography of bilateral myopic macular retinoschisis. Retina 36:e91–e92
Ono S, Takahashi A, Mase T, Nagaoka T, Yoshida A (2016) En face swept-source optical coherence tomographic analysis of X-linked juvenile retinoschisis. Am J Ophthalmol Case Rep 2:30–32
Katagiri S, Yokoi T, Mikami M, Nishina S, Azuma N (2016) Outer retinal deformity detected by optical coherence tomography in eyes with foveal hypoplasia. Graefes Arch Clin Exp Ophthalmol. doi:10.1007/s00417-016-3385-z
Yokoi T, Nishina S, Fukami M, Ogata T, Hosono K, Hotta Y et al (2016) Genotype-phenotype correlation of PAX6 gene mutations in aniridia. Hum Genome Var 3:15052
Benhamou N, Massin P, Haouchine B, Erginay A, Gaudric A (2002) Macular retinoschisis in highly myopic eyes. Am J Ophthalmol 133:794–800
Gass JD (1999) Muller cell cone, an overlooked part of the anatomy of the fovea centralis: hypotheses concerning its role in the pathogenesis of macular hole and foveomacualr retinoschisis. Arch Ophthalmol 117:821–823
Mooy CM, Van Den Born LI, Baarsma S, Paridaens DA, Kraaijenbrink T, Bergen A et al (2002) Hereditary X-linked juvenile retinoschisis: a review of the role of Muller cells. Arch Ophthalmol 120:979–984
Reid SN, Yamashita C, Farber DB (2003) Retinoschisin, a photoreceptor-secreted protein, and its interaction with bipolar and muller cells. J Neurosci 23:6030–6040
Tang J, Rivers MB, Moshfeghi AA, Flynn HW, Chan CC (2010) Pathology of macular foveoschisis associated with degenerative myopia. J Ophthalmol. doi:10.1155/2010/175613
Rashid A, Grossniklaus HE (2014) Update on the pathology of pathological myopia. In: Spaide FR, Ohno-Matsui K, Yannuzzi AL (eds) Pathologic myopia. Springer, New York, NY, pp 83–95
Hendrickson A, Possin D, Vajzovic L, Toth CA (2012) Histologic development of the human fovea from midgestation to maturity. Am J Ophthalmol 154(5):767.e2–778.e2
Kafieh R, Rabbani H, Kermani S (2013) A review of algorithms for segmentation of optical coherence tomography from retina. J Med Signals Sens 3:45–60
Acknowledgments
We thank the patients for participating in this study. This work was supported by grants from the Ministry of Health, Labour, and Welfare (H24-Nanchi-Ippan-031) and the National Centre for Child Health and Development 25-7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Ethical approval
The Institutional Review Board of the National Center for Child Health and Development approved this retrospective observational case series, which adhered to the tenets of the Declaration of Helsinki.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
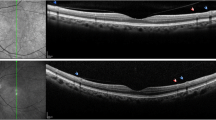
Pairs of horizontal optical coherence tomography (OCT) images of each retinal layer from 512 A-scans × 256 B-scans in a 3 × 3-millimeter square. a A randomly selected pair of upper and lower horizontal OCT images corresponding to the holes in the ganglion cell layer (GCL) on en-face OCT clearly show several schisis cavities in the GCL, while the B-scan images do not detect a round expanding schisis cavity (right eye of case 5). b A randomly selected pair of upper and lower horizontal OCT images corresponding to the reticular pattern in the inner nuclear layer (INL) on en-face OCT show similar structures (right eye of case 3). c A randomly selected pair of upper and lower horizontal OCT images corresponding to a spoke-wheel pattern in the INL on en-face OCT show similar structures (right eye of case 6). d A randomly selected pair of upper and lower horizontal OCT images corresponding to polygonal hyporeflective cavities with hyperreflective partitions in the outer plexiform layer on en-face OCT show similar structures (right eye of case 8). a–d In each column, yellow dashed lines in the upper panels indicate the position at which the B-scan images in the middle and the lower panels are obtained. The orange arrows in the upper panels indicating the cavity in each en-face OCT image correspond to the orange arrows in the middle panels indicating the cavity in each B-scan image. The yellow arrows in the upper panels indicating the cavity in each en-face OCT image correspond to the yellow arrows in the lower panels indicating the cavity in each B-scan image. (GIF 99 kb)
Rights and permissions
About this article
Cite this article
Yoshida-Uemura, T., Katagiri, S., Yokoi, T. et al. Different foveal schisis patterns in each retinal layer in eyes with hereditary juvenile retinoschisis evaluated by en-face optical coherence tomography. Graefes Arch Clin Exp Ophthalmol 255, 719–723 (2017). https://doi.org/10.1007/s00417-016-3552-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3552-2