Abstract
Background
Age-related macular degeneration is the primary cause of blindness in developed countries. Current treatments of this degenerative disease mainly include laser, photodynamic therapy with verteporfin and administration of anti-vascular endothelial growth factors. The LUEUR (LUcentis® En Utilisation Réelle) study is composed of a cross-sectional part (LUEUR1), which examined the current management of wet AMD in France, and a follow-up part (LUEUR2), which will assess the development of patients treated for wet AMD over 4 years. Here we describe the results of LUEUR1.
Methods
Patients with wet AMD were enrolled during a routine medical examination in LUEUR1, a cross-sectional, observational, prospective, multicentre study. Investigators recorded patient demographics, visual acuity, characteristics of wet AMD lesions, date of AMD diagnosis, comorbidities, previous treatments, treatments prescribed at inclusion, and low vision rehabilitation.
Results
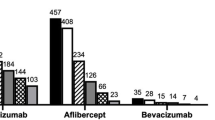
A total of 72 investigators recruited 1,019 patients with wet AMD, corresponding to 1,405 eyes affected by the disease. The mean age of patients was 78.7 ± 7.3 years. Most were female (62.3%) and non-smokers (66.9%). The mean visual acuity was 49.12 ± 24.18 Early Treatment Diabetic Retinopathy Study letters. Most eyes showed occult (52.8%) and subfoveal (84.6%) choroidal neovascularisation. Bilateral wet AMD affected 37.9% of patients. The median time since diagnosis was 12 months. Ranibizumab-based therapy (67.3%) and photodynamic therapy (29.8%) were the most frequent previous treatments. Prior to inclusion, 5.6% of patients had low vision rehabilitation. When a treatment was prescribed on the day of inclusion, it was most often ranibizumab (89.0% of all treatments at inclusion).
Conclusions
The results of this study illustrate the impact of anti-vascular endothelial growth factor therapies on the treatment of wet AMD in a real-life context. Specifically, ranibizumab-based therapy appears to have largely replaced laser photocoagulation and verteporfin-based photodynamic therapy.


Similar content being viewed by others
Abbreviations
- AMD:
-
age-related macular degeneration
- CNV:
-
choroidal neovascularisation
- PDT:
-
photodynamic therapy
- VEGF:
-
vascular endothelial growth factor
- ETDRS:
-
Early Treatment Diabetic Retinopathy Study
References
Resnikoff S, Pascolini D, Etya'ale D, Kocur I, Pararajasegaram R, Pokharel GP, Mariotti SP (2004) Global data on visual impairment in the year 2002. Bull World Health Organ 82:844–851
Friedman DS, O'Colmain BJ, Muñoz B, Tomany SC, McCarty C, de Jong PTVM, Nemesure B, Mitchell P, Kempen J (2004) Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol 122:564–572
Schmidt-Erfurth UM, Richard G, Augustin A, Aylward WG, Bandello F, Corcòstegui B, Cunha-Vaz J, Gaudric A, Leys A, Schlingemann RO (2007) Guidance for the treatment of neovascular age-related macular degeneration. Acta Ophthalmol Scand 85:486–494
de Jong PTVM (2006) Age-related macular degeneration. N Engl J Med 355:1474–1485
Evans JR (2001) Risk factors for age-related macular degeneration. Prog Retin Eye Res 20:227–253
Baird PN, Hageman GS, Guymer RH (2009) New era for personalized medicine: the diagnosis and management of age-related macular degeneration. Clin Experiment Ophthalmol 37:814–821
Leveziel N, Zerbib J, Richard F, Querques G, Morineau G, Fremeaux-Bacchi V, Coscas G, Soubrane G, Benlian P, Souied EH (2008) Genotype-phenotype correlations for exudative age-related macular degeneration associated with homozygous HTRA1 and CFH genotypes. Invest Ophthalmol Vis Sci 49:3090–3094
Ferrara N, Gerber H-P, LeCouter J (2003) The biology of VEGF and its receptors. Nat Med 9:669–676
Hubschman JP, Reddy S, Schwartz SD (2009) Age-related macular degeneration: current treatments. Clin Ophthalmol 3:155–166
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431
Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY, Sy JP, Schneider S (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355:1432–1444
Beutel J, Rudolf M, Grisanti S (2009) Current and future therapies for age-related macular degeneration. Expert Opin Emerg Drugs 14:341–362
Zanlonghi X, Avital L, Pedelahore C, Robin C, Baty F (2003) Comment explorer la vision dans un contexte de conduite automobile? Conduite et age. Octopus Multimédia, Paris, pp 101–105
Delcourt C, Michel F, Colvez A, Lacroux A, Delage M, Vernet MH, POLA Study Group (2001) Associations of cardiovascular disease and its risk factors with age-related macular degeneration: the POLA study. Ophthalmic Epidemiol 8:237–249
Zawinka C, Ergun E, Stur M (2005) Prevalence of patients presenting with neovascular age-related macular degeneration in an urban population. Retina 25:324–331
Wong TY, Wong T, Chakravarthy U, Klein R, Mitchell P, Zlateva G, Buggage R, Fahrbach K, Probst C, Sledge I (2008) The natural history and prognosis of neovascular age-related macular degeneration: a systematic review of the literature and meta-analysis. Ophthalmology 115:116–126
Bonastre J, Le Pen C, Soubrane G, Quentel G (2003) The burden of age-related macular degeneration: results of a cohort study in two French referral centres. Pharmacoeconomics 21:181–190
Ergun E, Zawinka C, Stur M (2004) Incidence of laser photocoagulation and photodynamic therapy with verteporfin at a tertiary retinal center. Retina 24:13–18
Cohen SY, Creuzot-Garcher C, Darmon J, Desmettre T, Korobelnik JF, Levrat F, Quentel G, Paliès S, Sanchez A, de Gendre AS, Schluep H, Weber M, Delcourt C (2007) Types of choroidal neovascularisation in newly diagnosed exudative age-related macular degeneration. Br J Ophthalmol 91:1173–1176
Macular Photocoagulation Study Group (1997) Risk factors for choroidal neovascularization in the second eye of patients with juxtafoveal or subfoveal choroidal neovascularization secondary to age-related macular degeneration. Arch Ophthalmol 115:741–747
Solomon SD, Jefferys JL, Hawkins BS, Bressler NM (2007) Incident choroidal neovascularization in fellow eyes of patients with unilateral subfoveal choroidal neovascularization secondary to age-related macular degeneration. SST report No. 20 from the Submacular Surgery Trials Research Group. Arch Ophthalmol 125:1323–1330
Chang TS, Bressler NM, Fine JT, Dolan CM, Ward J, Klesert TR (2007) Improved vision-related function after Ranibizumab treatment of neovascular age-related macular degeneration: results of a randomized clinical trial. Arch Ophthalmol 125:1460–1469
Bressler NM, Chang TS, Fine JT, Dolan CM, Ward J (2009) Improved vision-related function after ranibizumab vs photodynamic therapy: a randomized clinical trial. Arch Ophthalmol 127:13–21
van Velthoven MEJ, de Smet MD, Schlingemann RO, Magnani M, Verbraak FD (2006) Added value of OCT in evaluating the presence of leakage in patients with age-related macular degeneration treated with PDT. Graefes Arch Clin Exp Ophthalmol 244:1119–1123
Fung AE, Lalwani GA, Rosenfeld PJ, Dubovy SR, Michels S, Feuer WJ, Puliafito CA, Davis JL, Flynn HW, Esquiabro M (2007) An optical coherence tomography-guided, variable dosing regimen with intravitreal ranibizumab (Lucentis) for neovascular age-related macular degeneration. Am J Ophthalmol 143:566–583
ANAES report. [Traitements de la dégénérescence maculaire liée à l'âge]. http://www.has-sante.fr/portail/upload/docs/application/pdf/DMLA.rap.pdf (accessed 08 Feb 2010)
Acknowledgments
We thank the physicians who kindly participated in this study. The study was conducted by EZUS, University of Lyon, France for Novartis Pharma and by the study scientific committee (SY Cohen, G De Pouvourville, G Dupeyron, M Lievre, E Souied and M Weber). All members of the scientific committee had a direct role in study design, analysis and interpretation of data as well as in the writing of and decision to submit this manuscript. We also thank 4Clinics (Waterloo, Belgium) for assistance in writing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funding and competing interests
This study was sponsored by Novartis Pharma SAS, France. SY Cohen, G De Pouvourville, G Dupeyron, M Lievre, E Souied and M Weber are consultants for Novartis Pharma, France. A Ponthieux is an employee of Novartis Pharma, France. The authors have full control of all primary data, and they agree to allow Graefe's Archive for Clinical and Experimental Ophthalmology to review their data upon request.
Rights and permissions
About this article
Cite this article
Cohen, SY., Souied, E.H., Weber, M. et al. Patient characteristics and treatment of neovascular age-related macular degeneration in France: the LUEUR1 observational study. Graefes Arch Clin Exp Ophthalmol 249, 521–527 (2011). https://doi.org/10.1007/s00417-010-1553-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-010-1553-0