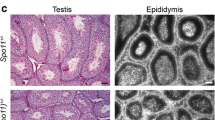
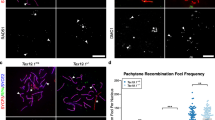
Abstract
Lack of crossing-over in meiosis can trigger an apoptotic response at metaphase I by the spindle assembly checkpoint (SAC). In contrast to females, segregation of sex chromosomes in males poses a particular challenge as recombination and chiasma formation is restricted to the pseudoautosomal region, the small region of homology between X and Y chromosomes. Existing data indicate that low levels of crossover failure in male meiosis can be tolerated without compromising fertility, while high levels of X-Y dissociation (in ≥70 % of cells) result in widespread apoptosis and subsequent infertility, demonstrated earlier, e.g., in Spo11β-only mice. Here, we explore the threshold of X-Y recombination failure frequency that is compatible with fertility. We show that in Spo11β-onlymb mice with a mixed genetic background, in contrast to Spo11β-only mice with a C57BL/6 background, X-Y pairing fails in ~50 % of cells but this still allows for sperm production without any overt impact on fertility. We also review data on apoptosis and fertility from other achiasmate mouse models and propose that the incidence of homolog dissociation that can be tolerated in vivo without compromising male fertility lies between 50 and 70 %.



Similar content being viewed by others
References
Baker SM et al (1996) Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over. Nature Genet 13:336–342. doi:10.1038/ng0796-336
Barchi M, Roig I, Di Giacomo M, de Rooij DG, Keeney S, Jasin M (2008) ATM promotes the obligate XY crossover and both crossover control and chromosome axis integrity on autosomes. PLoS Genet 4:e1000076. doi:10.1371/journal.pgen.1000076
Baudat F, Manova K, Yuen JP, Jasin M, Keeney S (2000) Chromosome synapsis defects and sexually dimorphic meiotic progression in mice lacking Spo11. Mole Cell 6:989–998
Bellani MA, Boateng KA, McLeod D, Camerini-Otero RD (2010) The expression profile of the major mouse SPO11 isoforms indicates that SPO11beta introduces double strand breaks and suggests that SPO11alpha has an additional role in prophase in both spermatocytes and oocytes. Mole Cell Biol 30:4391–4403. doi:10.1128/MCB.00002-10
Burgoyne PS, Mahadevaiah SK, Sutcliffe MJ, Palmer SJ (1992) Fertility in mice requires X-Y pairing and a Y-chromosomal “spermiogenesis” gene mapping to the long arm. Cell 71:391–398
Davisson MT, Akeson EC (1993) Recombination suppression by heterozygous Robertsonian chromosomes in the mouse. Genetics 133:649–667
Dobles M, Liberal V, Scott ML, Benezra R, Sorger PK (2000) Chromosome missegregation and apoptosis in mice lacking the mitotic checkpoint protein Mad2. Cell 101:635–645
Eaker S, Pyle A, Cobb J, Handel MA (2001) Evidence for meiotic spindle checkpoint from analysis of spermatocytes from Robertsonian-chromosome heterozygous mice. J Cell Sci 114:2953–2965
Eaker S, Cobb J, Pyle A, Handel MA (2002) Meiotic prophase abnormalities and metaphase cell death in MLH1-deficient mouse spermatocytes: insights into regulation of spermatogenic progress. Deve Biol 249:85–95
Hunt P, LeMaire R, Embury P, Sheean L, Mroz K (1995) Analysis of chromosome behavior in intact mammalian oocytes: monitoring the segregation of a univalent chromosome during female meiosis. Hum Mole Genet 4:2007–2012
Kauppi L, Barchi M, Baudat F, Romanienko PJ, Keeney S, Jasin M (2011) Distinct properties of the XY pseudoautosomal region crucial for male meiosis. Science 331:916–920. doi:10.1126/science.1195774
Kauppi L, Jasin M, Keeney S (2012) The tricky path to recombining X and Y chromosomes in meiosis. Ann N Y Acad Sci 1267:23. doi:10.1111/j.1749-6632.2012.06593.x
Keeney S, Giroux CN, Kleckner N (1997) Meiosis-specific DNA double-strand breaks are catalyzed by Spo11, a member of a widely conserved protein family. Cell 88:375–384
Keeney S et al (1999) A mouse homolog of the Saccharomyces cerevisiae meiotic recombination DNA transesterase Spo11p. Genomics 61:170–182. doi:10.1006/geno.1999.5956
Kipling D et al (1996) Structural variation of the pseudoautosomal region between and within inbred mouse strains. Proc Natl Acad Sci U S A 93:171–175
Lyon MF, Cattanach BM, Charlton HM (1981) Genes affecting sex differentiation in mammals. In: Austin CR, Edwards RG (eds) Mechanisms of sex differentiation in animals and man. Academic Press, London, pp 329–386
Mahadevaiah S, Setterfield LA, Mittwoch U (1988) Univalent sex chromosomes in spermatocytes of Sxr-carrying mice. Chromosoma 97:145–153
Mahadevaiah SK, Evans EP, Burgoyne PS (2000) An analysis of meiotic impairment and of sex chromosome associations throughout meiosis in XYY mice. Cytogene Cell Genet 89:29–37
Matsuda Y, Hirobe T, Chapman VM (1991) Genetic basis of X-Y chromosome dissociation and male sterility in interspecific hybrids. Proc Natl Acad Sci U S A 88:4850–4854
Matsuda Y, Moens PB, Chapman VM (1992) Deficiency of X and Y chromosomal pairing at meiotic prophase in spermatocytes of sterile interspecific hybrids between laboratory mice (Mus domesticus) and Mus spretus. Chromosoma 101:483–492
Moens PB, Freire R, Tarsounas M, Spyropoulos B, Jackson SP (2000) Expression and nuclear localization of BLM, a chromosome stability protein mutated in Bloom’s syndrome, suggest a role in recombination during meiotic prophase. J Cell Sci 113(Pt 4):663–672
Perry J, Palmer S, Gabriel A, Ashworth A (2001) A short pseudoautosomal region in laboratory mice. Genome Res 11:1826–1832. doi:10.1101/gr.203001
Romanienko PJ, Camerini-Otero RD (1999) Cloning, characterization, and localization of mouse and human SPO11. Genomics 61:156–169. doi:10.1006/geno.1999.5955
Sutcliffe MJ, Darling SM, Burgoyne PS (1991) Spermatogenesis in XY, XYSxra and XOSxra mice: a quantitative analysis of spermatogenesis throughout puberty. Mole Reprod Dev 30:81–89. doi:10.1002/mrd.1080300202
Tease C, Cattanach BM (1989) Sex chromosome pairing patterns in male mice of novel Sxr genotypes. Chromosoma 97:390–395
Vernet N et al (2011) The Y-encoded gene Zfy2 acts to remove cells with unpaired chromosomes at the first meiotic metaphase in male mice. Curr Biol CB 21:787–793. doi:10.1016/j.cub.2011.03.057
Vernet N, Mahadevaiah SK, Yamauchi Y, Decarpentrie F, Mitchell MJ, Ward MA, Burgoyne PS (2014) Mouse Y-linked Zfy1 and Zfy2 are expressed during the male-specific interphase between meiosis I and meiosis II and promote the 2nd meiotic division. PLoS Genet 10, e1004444. doi:10.1371/journal.pgen.1004444
Acknowledgments
We are grateful to Scott Keeney, Maria Jasin, and Robert Benezra (Memorial Sloan-Kettering Cancer Center, New York) for providing mice and to Elina Hakonen from Timo Otonkoski’s Laboratory (University of Helsinki), Noora Kotaja (University of Turku), and Rocio Sotillo (EMBL, Italy) for their advice on experimental procedures. Assistance was provided by the following core facilities at the University of Helsinki: the Tissue Preparation and Histochemistry Unit, the Laboratory Animal Center, and the Biomedicum Imaging Unit.
Funding
L.K. is an Academy Research Fellow of the Academy of Finland and the recipient of a Marie Curie Career Integration Grant (European Commission Framework Programme 7). This work was supported by the Academy of Finland (funding decision number 263870), Biocentrum Helsinki, and the Sigrid Juselius Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Imrul Faisal declares that he has no conflict of interest. Liisa Kauppi declares that she has no conflict of interest.
Ethical approval
All applicable national and institutional guidelines for the care and use of animals were followed.
Additional information
This article is part of a special issue on “Recent advances in meiotic chromosome structure, recombination, and segregation” edited by Marco Barchi, Paula Cohen, and Scott Keeney
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
(PDF 70 kb)
Rights and permissions
About this article
Cite this article
Faisal, I., Kauppi, L. Sex chromosome recombination failure, apoptosis, and fertility in male mice. Chromosoma 125, 227–235 (2016). https://doi.org/10.1007/s00412-015-0542-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-015-0542-9