Abstract
Introduction
A restful sleep is essential for regenerative processes and remains crucial for patients recovering from stressful periods in the intensive care unit. The current study aimed to assess sleep quality in critically ill patients receiving invasive mechanical ventilation within a specialized weaning unit in hospital.
Methods
Tracheotomized subjects undergoing prolonged weaning from mechanical ventilation were included in the study. Polysomnography and gas exchange monitoring was performed during nocturnal ventilation. Subjective evaluation of sleep quality and health-related quality of life were also assessed.
Results
Nineteen subjects completed the study protocol. Sleep architecture was highly heterogeneous across individual subjects. Mean total sleep time (TST) was 273 ± 114 min, sleep efficacy 70 ± 23%, slow-wave sleep 25.7 ± 18.4%/TST, rapid eye movement sleep 9.6 ± 7.5%/TST, and arousal index 18.7 ± 12.4/h. No significant difference in sleep quality was found between subjects with successful (N = 7) or unsuccessful (N = 12) weaning. Bicarbonate levels were negatively correlated both with sleep efficacy and sleep quality, that latter of which was subjectively assessed by the subjects using a visual analogue scale.
Conclusion
Subjects who were undergoing prolonged weaning from mechanical ventilation and admitted to a specialized weaning unit, showed reduced sleep quality with preservation of high amounts of slow-wave sleep.
Similar content being viewed by others
Introduction
Weaning from mechanical ventilation is essential for proper care of critically ill patients receiving invasive mechanical ventilation [1]. According to the difficulty and duration of the weaning process, patients can be classified into three groups: simple weaning, difficult weaning, and prolonged weaning [1]. In particular, prolonged weaning can last several weeks, depending on medical conditions and underlying disease of the patient [2], and survival is known to be impaired [3].
Sleep quality in ICU patients can be compromised by critical illness, as well as environmental factors such as staff number, background noise, and frequency of interventions [4–8]. Furthermore, it is known that sleep deprivation is associated with deterioration of respiratory muscle strength [9]. However, there is only limited information available about sleep quality in the setting of prolonged weaning. Fanfulla et al. found that in a mixed cohort of patients, with or without mechanical ventilation, treated on a step-down unit after initial ICU admission still showed sleep abnormalities [10].
During the last decades, specialized weaning units have been implemented in several countries to improve medical care and weaning outcome, to reduce costs and relieve pressure on the demand for limited ICU beds [11, 12]. Compared to ICUs, weaning units provide an environment more similar to that at home (e.g., less noise, privacy, daytime activity, longer visiting hours) [2]. To our knowledge this is the first trial to assess and describe sleep quality in subjects undergoing prolonged weaning from invasive mechanical ventilation in a specialized weaning unit.
Materials and Methods
The study protocol was approved by Local Ethics Committee (Ärztekammer Nordrhein, Germany) and performed in accordance with ethical standards laid down in the Declaration of Helsinki, 2013. The study was registered on German Clinical Trials Register under DRKS00004736. Informed written consent was obtained from all subjects or their legal guardians.
Subjects
The study was performed in tracheostomized subjects undergoing prolonged weaning from mechanical ventilation who were admitted to a specialized weaning unit (Department of Pneumology, Cologne-Merheim Hospital, University of Witten/Herdecke). All subjects were fully conscious, investigated during a stable state (respiratory rate <35/min, pH >7.30, no clinical signs for infection), and fulfilled the criteria for readiness to wean [1, 12]. Subjects were excluded if they received vasopressors or continuous sedative medication, anxiolytics, and analgetics, or if they showed clinical signs of delirium.
Once subjects were discharged from the weaning unit, weaning outcome was documented. Subjects were categorized into the following groups: (I) successful weaning with or without transfer to long-term non-invasive positive pressure ventilation (NPPV) or (II) unsuccessful weaning with the need for ongoing invasive mechanical ventilation outside the hospital.
Specialized Weaning Unit
The weaning unit from the Department of Pneumology, Cologne-Merheim Hospital, University of Witten/Herdecke, is a respiratory ICU that specializes in the treatment of patients undergoing prolonged weaning. All patients are accommodated in separate rooms that can be darkened. Telemetry is used for continuous monitoring of ECG and pulse oximetry. Background noise and nursing interventions are reduced to a minimum at night to sustain circadian rhythm.
Study Design
Nocturnal measurements were taken while subjects were continuously ventilated via a cuffed tracheostomy tube attached to a personalized ventilator. Subjective sleep quality and health-related quality of life (HRQL) using the severe respiratory insufficiency (SRI) questionnaire [13, 14] were assessed the following morning after termination of nocturnal measurements.
Methods
Polysomnographic Measurements
Full-night polysomnography was performed with Alice®LE (Philips Respironics, Murrysville, US) between 10 p.m. and 6 a.m. A trained investigator positioned two electrodes at locations C3 and C4, and two reference electrodes at mastoids A1 and A2. Electrooculograms, a submental electromyogram, and ECG were all recorded. Respiratory air flow was monitored with a flow transducer connected to a tracheostomy tube. Abdominal and thoracic plethysmography (respiratory effort belts) and pulse oximetry were recorded. EEG signals were amplified, recorded at 200 Hz sampling frequency, and filtered (0.5–30 Hz). Sleep recordings were scored manually in 30 s epochs. Sleep stages were classified according to the criteria described by Rechtschaffen and Kales [15]. Total sleep time (TST) was defined as total time asleep from beginning to the end of the recording period. Sleep efficiency (SE) was defined as the ratio of TST–sleep period time (SPT), which is the time from first sleep until final awakening. Sleep onset-latency time was defined as the length of time for transition from full wakefulness at the beginning of recording until sleep, and rapid eye movement (REM) latency from the beginning of recording to the first occurrence of REM sleep. Arousals were defined according to the criteria by Rechtschaffen and Kales [15]. Arousal index was defined as the number of arousals/h of sleep. The investigator scoring polysomnographic data was blinded to ventilator settings and subjective evaluation.
Gas Exchange Monitoring
\({\text{S}}{{\text{p}}_{{{\text{O}}_2}}}\)was continuously measured with a pulse oximeter using a finger-type probe. Minimum and mean \({\text{S}}{{\text{p}}_{{{\text{O}}_2}}}\) oxygen desaturation index (transient desaturation defined as a decrease in \({\text{S}}{{\text{p}}_{{{\text{O}}_2}}}\) of ≥4%), and time of desaturations <90% were recorded. Transcutaneous \({P_{{\text{C}}{{\text{O}}_2}}}\) (\({\text{Pt}}{{\text{c}}_{{\text{C}}{{\text{O}}_2}}}\)) was monitored between 10 p.m. and 6 a.m using SenTec digital monitor. The sensor was attached to the forehead. Correction of technical drift was calculated by the device, as previously described [16]. Here, minimal, mean, maximal, and time of \({\text{Pt}}{{\text{c}}_{{\text{C}}{{\text{O}}_2}}}\) >55 mmHg were recorded. In addition, arterial blood samples were taken from the arterialized earlobe at 6 a.m. for analysis (ALB800 Flex, Radiometer) [17].
Subjective Evaluation of Sleep Quality and HRQL
Using the visual analogue scale ranging from 0 to 100, subjects were asked to score their own sleep quality, in terms of restfulness and disturbance, during the sleep period. Higher values were deemed to represent better sleep quality. Following four questions were addressed: (1) How restful was your sleep? (2) Do you have the feeling of being well rested this morning? (3) To what extent did mechanical ventilation disrupt your sleep quality? (4) To what extent did ambient noise disrupt your sleep quality?
The SRI questionnaire was used to measure HRQL in subjects with severe respiratory failure [13, 14]. Finally, subject-specific data were collected, namely, the primary disease that led to mechanical ventilation on the ICU and prolonged weaning, co-morbidities, ventilator settings, and current medication.
Analysis
All data were subjected to normality testing using the Shapiro–Wilk test; this was valid for both whole group and subgroup comparisons, respectively. Here, subjects with successful weaning, with or without continuance of long-term non-invasive ventilation, were compared to subjects who could not be weaned from invasive ventilation.
All normally distributed data are presented as mean ± standard deviation. Non-normally distributed data (Shapiro–Wilk with P-value <0.05) are given as median values with 25th–75th percentiles. For group comparisons, paired t-tests were used for normally distributed data, while a non-parametric test (Wilcoxon–Mann–Whitney rank sum test) was applied to non-normally distributed data. Group effects were estimated with 95% confidence intervals and tested with a two-sided level of 0.05. Binary data Fisher’s exact test was used to compare the proportions between different weaning categories.
Spearman’s rank correlation coefficient was determined among the following variables: TST, SE, REM sleep, slow-wave sleep (NREM 3 + 4), arousal index, SRI summary scale score, mean \({\text{Pt}}{{\text{c}}_{{\text{C}}{{\text{O}}_2}}},\) bicarbonate, and subjective sleep quality.
Results
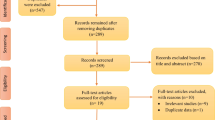
Twenty-one subjects receiving invasive mechanical ventilation and undergoing prolonged weaning were consecutively enrolled between May 2013 and April 2014. Polysomnographic measurements were unsuccessful in two subjects due to technical limitations (Fig. 1). Therefore, a total of 19 subjects completed the study protocol and 13 of these subjects suffered primarily from COPD. The remaining subjects suffered from neuromuscular diseases (N = 4; 2 with ALS, 1 with spinal cord injury, 1 with critical illness polyneuropathy), kyphoscoliosis (N = 1), and cardiac as well as renal insufficiency with severe pulmonary congestion (N = 1). All subjects were initially intubated and treated in ICU for acute respiratory failure; they then underwent prolonged weaning from the respirator. Three subjects (2 with COPD, 1 with ALS) were included during a follow-up visit for re-evaluation of potential weaning after the establishment of long-term invasive home mechanical ventilation.
At discharge from the weaning unit, four had been successfully weaned from mechanical ventilation, while another three were transferred to NPPV. The remaining 12 subjects could not be weaned and therefore continued invasive mechanical ventilation outside the hospital. Demographic data and ventilator settings of subjects with successful and unsuccessful weaning are given in Table 1.
Sleep Analysis
TST among all subjects (N = 19) was 273 ± 114 min, with a sleep efficacy of 71 ± 23%, a sleep onset-latency time of 53 ± 40 min, a REM-latency time of 130 ± 76 min, and an arousal index of 18.7 ± 12.4/h. Distribution of sleep stages is illustrated in Fig. 2 for both subgroups (successfully and unsuccessfully weaned), and in Fig. 3 for each individual. No significant differences between successfully and unsuccessfully weaned subjects were seen in sleep quantity, sleep architecture, arousals, or nocturnal heart rates (Table 2).
Individual distribution of sleep stages, total sleep time, and levels of bicarbonate during invasive mechanical ventilation in subjects undergoing prolonged weaning (N = 19). Data are presented in ascending order of sleep efficiency. \(HCO_3^ -\) bicarbonate, NREM non-rapid eye movement sleep, MVT movement time, REM rapid eye movement sleep, SPT sleep period time, TST total sleep time
Respiratory Analysis and Gas Exchange
Flow measurements using a pressure transducer connected to the tracheostomy tube showed no sleep-disordered breathing in all subjects during invasive mechanical ventilation. Nocturnal gas exchange did not differ significantly between subjects with successful versus unsuccessful weaning (Table 2). Of note, bicarbonate levels were moderately correlated with mean \({\text{Pt}}{{\text{c}}_{{\text{C}}{{\text{O}}_2}}}\) (R = 0.49; P 0.04), and were also negatively correlated with sleep efficacy (R = −0.48; P 0.036; Fig. 3).
Subjective Evaluation of Sleep Quality and HRQL
Results of the subjective evaluation of sleep quality and SRI questionnaire are given in Table 3. Mean SRI summary score (N = 19) was 49 ± 14. Correlation analyses between questions about subjective sleep quality and (i) SRI summary score, (ii) objective measurements of sleep quality, and (iii) nocturnal gas exchange, are given in Table 4.
Discussion
This is the first study to provide data on sleep quality in subjects undergoing prolonged weaning from invasive mechanical ventilation in a specialized weaning unit. The main findings are (1) mean slow-wave sleep was within normal range, despite sleep architecture showing an elevated arousal index, a decreased amount of REM stage, and reductions in both SE and TST, (2) sleep quantity and sleep architecture were highly heterogeneous across individual subjects, (3) no significant difference in sleep quality and HRQL was found between subjects with successful versus unsuccessful weaning.
Previous studies investigating sleep in invasive mechanically ventilated subjects mainly included patients who were treated in the ICU, and reported that most subjects suffer from a predominance of stage 1 sleep, with deficient amounts of slow-wave and REM sleep [4–8]. Of note, most of the subjects in these studies were sedated, had more serious impairments caused by the severity of their underlying diseases, and the surroundings were more likely to be noisy and regularly disrupted by patient care activities. In contrast to these findings, the subjects in the current investigation were treated in a specialized weaning unit and presented with slow-wave sleep within a normal range. This difference can be explained by the following reasons: firstly, subjects in the current study had already been mechanically ventilated for a notable period of time. Secondly, the specialized weaning unit provides single rooms, and all activities are aimed at maintaining day–night rhythms, with sleep-disturbing activities being reduced as much as possible; this is in contrast to the conditions in the busy ICU. Accordingly, the arousal index in the present study remained below 20 arousals/h. Bonnet and Arand even reported similar values for arousal index (22/h) for normal subjects aged 50–70 years [18]. Fanfulla et al. found similar results in a mixed cohort of subjects with or without mechanical ventilation in a step-down unit after treatment in the ICU for acute respiratory failure [10]. Here, the arousal index was 21 arousals/h in subjects with mechanical ventilation and 22 arousals/h in subjects without mechanical ventilation [10]. In contrast, Parthasarathy and Tobin reported a twofold higher arousal index in critically ill subjects treated in the ICU [8]. The study by Fanfulla et al. also showed that illumination, noise, and patient care only contributed minimally to sleep impairment [10]. Similarly, subjects in the current study reported that ambient noise did not subjectively disturb their sleep.
Only a small number of studies have previously investigated sleep quality in subjects with long-term invasive mechanical ventilation under stable conditions. In line with the current findings, Nardi et al. reported a similar arousal index in subjects with Duchenne muscular dystrophy who had been mechanically ventilated via a tracheostoma for an average of almost 100 months [19]. These results gained after unintentional leaks were minimized by the implementation of low-pressure cuffed tubes, which also improved gas exchange. Of note, subjects initially received mechanical ventilation via uncuffed tubes with high amounts of leakage, and this was associated with a twofold higher arousal index. This clearly indicates the vulnerability of sleep to the side-effects of mechanical ventilation via tracheostoma. Interestingly, in the study by Nardi et al., the TST, the amount of REM stage, and SE were all substantially higher than the same parameters in the present study. This can be explained by the fact that subjects enrolled in the aforementioned study were comparatively younger, and the duration of mechanical ventilation was considerably longer. Subjects in the current investigation represent those with severe chronic respiratory failure and impaired HRQL, as revealed by the results of the SRI. These results by the SRI were similar to the recently published data for HRQL of patients with long-term home mechanical ventilation [20]. From this observation, it can be concluded that habituation to mechanical ventilation improves sleep quality in subjects who are dependent on this form of ventilation.
Previous studies have shown that mechanical ventilation itself can cause sleep disruption, although the precise mechanism has not yet been defined [4, 8, 19]. In the current study, the majority of subjects were ventilated in the assist-controlled mode with a high backup rate. Polysomnographic flow measurements using a flow transducer connected to a tracheostomy tube showed no sleep-disordered breathing in all the subjects. In addition, subjects reported this mode of invasive ventilation as non-disruptive to their sleep. In contrast, Parthasarathy and Tobin reported that switching from assist-controlled to pressure-supported ventilation increases sleep fragmentation related to central apneas [8]. As a result, respiratory events can have a substantial impact on the nocturnal sleep quality during invasive mechanical ventilation.
Interestingly, bicarbonate levels were negatively correlated with both objective and subjective parameters of sleep quality. Elevated bicarbonate levels in patients with chronic respiratory failure typically indicate metabolic compensation of respiratory acidosis resulting from hypercapnia. However, mean pH values were alkalotic rather than acidotic, and \({\text{P}}{{\text{a}}_{{\text{C}}{{\text{O}}_2}}}\) values in the morning following nocturnal ventilation were relatively normal. However, since metabolic adaptation occurs to some degree, elevated bicarbonate values could simply reflect intermittent increases in \({\text{P}}{{\text{a}}_{{\text{C}}{{\text{O}}_2}}}\) during daytime spontaneous breathing periods, as typically performed in patients on a prolonged weaning program [1, 11]. In this regard, one could speculate that excessive spontaneous breathing periods that result in elevated bicarbonate levels in prolonged weaning patients could also eventually have a negative impact on sleep quality, even if subsequent nocturnal mechanical ventilation efficiently improves alveolar ventilation. Interestingly, Fanfulla et al. found an association between higher pH and reduced sleep quantity and quality [10], possibly caused by increased neuronal excitability. In contrast, results showed elevated \({\text{Pt}}{{\text{c}}_{{\text{C}}{{\text{O}}_2}}}\)-levels >55 mmHg at least in some patients given by a continuous measurement, which has shown to provide a more complete picture of ventilation compared to single blood gas analysis [16]. However, since bicarbonate levels were not specifically investigated in this study, further studies are needed to understand the correlation and mechanisms between metabolic alterations and sleep.
There are some limitations to this study: firstly, this study was performed as a pilot trial in a mixed cohort, without the definition of a primary endpoint and sample size calculation. Consequently, while important ideas can be extracted from these results, further studies are still needed to provide a more systematic and conclusive assessment of the influence of sleep quality on weaning outcome. Secondly, TST was recorded at night between 10 p.m. and 6 a.m., but sleep periods during the day were not analyzed, despite previous studies reporting a significant impact of daytime sleep on the recovery in the subjects similar to those investigated here. For example, several studies have demonstrated that approximately 50% of sleep occurs during daytime in ICU patients [5–7]. Thirdly, further important disturbances such as ventilator–patient asynchronies, alarms, or nursing care manipulations were not systematically observed in the current investigation.
In conclusion, subjects who were submitted to a specialized weaning unit while undergoing prolonged weaning from mechanical ventilation showed impaired sleep quality. Of note, sleep architecture is suggested to be highly heterogeneous across individual weaning patients, even though slow-wave sleep is well preserved in most of these subjects, a finding in clear contrast to previous studies on ICU subjects.
References
Boles J, Bion J, Connors A, Herridge M, Marsh B, Melot C et al (2007) Weaning from mechanical ventilation. Eur Respir J 29:1033–1056
Schönhofer B, Euteneuer S, Nava S, Suchi S, Köhler D (2002) Survival of mechanically ventilated patients admitted to a specialised weaning centre. Intensive Care Med 28:908–916
Funk G, Anders S, Breyer M, Burghuber OC, Edelmann G, Heindl W et al (2010) Incidence and outcome of weaning from mechanical ventilation according to new categories. Eur Respir J 35:88–94
Cabello B, Thille AW, Drouot X, Galia F, Mancebo J, d’Ortho MP, Brochard L (2008) Sleep quality in mechanically ventilated patients: comparison of three ventilatory modes. Crit Care Med 36:1749–1755
Cooper AB, Thornley KS, Young GB, Slutsky AS, Stewart TE, Hanly PJ (2000) Sleep in critically ill patients requiring mechanical ventilation. Chest 117:809–818
Freedman NS, Gazendam J, Levan L, Pack AI, Schwab RJ (2001) Abnormal sleep/wake cycles and the effect of environmental noise on sleep disruption in the intensive care unit. Am J Respir Crit Care Med 163:451–457
Gabor JY, Cooper AB, Crombach SA, Lee B, Kadikar N, Bettger HE, Hanly PJ (2003) Contribution of the intensive care unit environment to sleep disruption in mechanically ventilated patients and healthy subjects. Am J Respir Crit Care Med 167:708–715
Parthasarathy S, Tobin MJ (2002) Effect of ventilator mode on sleep quality in critically ill patients. Am J Respir Crit Care Med 166:1423–1429
Chen HI, Tang YR (1989) Sleep loss impairs inspiratory muscle endurance. Am Rev Respir Dis 140(4):907–909
Fanfulla F, Ceriana P, D’Artavilla Lupo N, Trentin R, Frigerio F, Nava S (2011) Sleep disturbances in patients admitted to a step-down unit after ICU discharge: the role of mechanical ventilation. Sleep 34:355–362
Polverino E, Nava S, Ferrer M, Ceriana P, Clini E, Spada E et al (2010) Patients’ characterization, hospital course and clinical outcomes in five Italian respiratory intensive care units. Intensive Care Med 36:137–142
Schönhofer B, Geiseler J, Dellweg D, Moerer O, Barchfeld T, Fuchs H et al (2014) Prolonged weaning: S2k-guideline published by the German Respiratory Society. Pneumologie 68:19–75
Windisch W, Freidel K, Schucher B, Baumann H, Wiebel M, Matthys H, Petermann F (2003) The Severe Respiratory Insufficiency (SRI) Questionnaire: a specific measure of health-related quality of life in patients receiving home mechanical ventilation. J Clin Epidemiol 56:752–759
Windisch W, Budweiser S, Heinemann F, Pfeifer M, Rzehak P (2008) The Severe Respiratory Insufficiency (SRI) Questionnaire was valid for patients with COPD. J Clin Epidemiol 61:848–853
Rechtschaffen A, Kales A (1968) A manual of standardized terminology, techniques and scoring system for sleep stages in human subjects. NIH publication 204. National Institutes of Health, Washington, DC
Storre JH, Magnet FS, Dreher M, Windisch W (2011) Transcutaneous monitoring as a replacement for arterial PCO(2) monitoring during nocturnal non-invasive ventilation. Respir Med 105:143–150
Ekkernkamp E, Welte L, Schmoor C, Huttmann SE, Dreher M, Windisch W, Storre JH (2015) Spot check analysis of gas exchange: invasive versus non-invasive methods. Respiration 89:294–303
Bonnet MH, Arand DL (2007) EEG arousal norms by age. J Clin Sleep Med 3:271–274
Nardi J, Prigent H, Garnier B, Lebargy F, Quera-Salva MA, Orlikowski D, Lofaso F (2012) Efficiency of invasive mechanical ventilation during sleep in Duchenne muscular dystrophy. Sleep Med 13:1056–1065
Huttmann SE, Windisch W, Storre JH (2015) Invasive home mechanical ventilation: living conditions and health-related quality of life. Respiration 89(4):312–321
Acknowledgements
We thank the study subjects for the effort they devoted to this study, Dr. Abdel Hakim Bayarassou, and Axel Muth for their support and Dr. Sandra Dieni, PhD for helpful comments on the manuscript prior to submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The study group received an Open Research Grant from Weinmann Geräte für Medizin GmbH and Co. KG and VIVISOL Deutschland GmbH. All authors state that none of the discussed issues in the submitted manuscript was dependent on or influenced support and funding.
Rights and permissions
About this article
Cite this article
Huttmann, S.E., Wilms, K., Hamm, C. et al. Assessment of Sleep in Patients Receiving Invasive Mechanical Ventilation in a Specialized Weaning Unit. Lung 195, 361–369 (2017). https://doi.org/10.1007/s00408-017-9988-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-017-9988-2