Abstract
Purpose
Cystic fibrosis (CF) patients suffer from chronic lung inflammation. This inflammation may activate platelets. There are limited data on the role of platelet-secreted cytokines in CF. Platelet cytokines with inflammatory effects include vascular endothelial growth factor (VEGF) and transforming growth factor-β1 (TGF-β1). As levels of these cytokines are tenfold greater in serum than plasma due to platelet release, serum levels may be one index of platelet content, but a more specific index is release during the aggregation of isolated platelets. We postulated that altered release of these platelet cytokines occurs in CF.
Methods
We obtained sera and plasma from CF outpatients (n = 21) and from healthy controls (n = 20), measured VEGF and TGF-β1, assessed for correlations with platelet number, analyzed cytokine release during platelet aggregation to collagen, and analyzed differences in maximal platelet aggregation.
Results
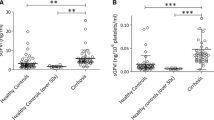
Platelet number and maximal aggregation levels were higher in CF. Plasma and serum levels of TGF-β1 and VEGF were higher in CF, but these levels were similar after adjusting for platelet number (serum cytokines correlated with platelet count). The release of VEGF and TGF-β1 during aggregation was decreased in CF platelets (by 52 and 29 %, respectively).
Conclusion
Platelet release is not a source of the elevated blood proinflammatory cytokines TGF-β1 and VEGF in CF, as platelets from CF patients actually release less of these cytokines. These data provide further evidence for platelet defects in CF.




Similar content being viewed by others
Abbreviations
- CF:
-
Cystic fibrosis
- CFTR:
-
Cystic fibrosis transmembrane conductance regulator
- NSAID:
-
Nonsteroidal anti-inflammatory drug
- PRP:
-
Platelet-rich plasma
- PPP:
-
Platelet-poor plasma
- TGF-β1 :
-
Transforming growth factor-beta 1
- VEGF:
-
Vascular endothelial growth factor
References
Rowe SM, Miller S, Sorscher EJ (2005) Cystic fibrosis. New Engl J Med 352(19):1992–2001
Stoltz DA, Meyerholz DK, Welsh MJ (2015) Origins of cystic fibrosis lung disease. New Engl J Med 372(16):1574–1575
O’Sullivan BP, Linden MD, Frelinger AL 3rd et al (2005) Platelet activation in cystic fibrosis. Blood 105(12):4635–4641
O’Sullivan BP, Michelson AD (2006) The inflammatory role of platelets in cystic fibrosis. Am J Respir Crit Care Med 173(5):483–490
Sturm A, Hebestreit H, Koenig C, Walter U, Grossmann R (2010) Platelet proinflammatory activity in clinically stable patients with CF starts in early childhood. J Cyst Fibros 9(3):179–186
Stead RJ, Barradas MA, Mikhailidis DP, Jeremy JY, Hodson ME, Batten JC, Dandona P (1987) Platelet hyperaggregability in cystic fibrosis. Prostaglandins Leukot Med 26(2):91–103
Agam G, Aviram M, Zilberman-Kaufman M, Rothstein A, Livne AA (1995) Cyclic AMP-related and cation-affected human platelet chloride transport regulation. Eur J Clin Chem Clin Biochem 33(6):329–335
Mattoscio D, Evangelista V, De Cristofaro R et al (2010) Cystic fibrosis transmembrane conductance regulator (CFTR) expression in human platelets: impact on mediators and mechanisms of the inflammatory response. FASEB J 24(10):3970–3980
Falco A, Romano M, Iapichino L, Collura M, Davi G (2004) Increased soluble CD40 ligand levels in cystic fibrosis. J Thromb Haemost 2(4):557–560
Mikhailidis DP, Stead RJ, Barradas MA, Hodson ME, Batten JC, Dandona P (1990) Platelet abnormalities in patients with cystic fibrosis and obligate heterozygotes. Haematologica 75(2):137–140
Zhao C, Su EM, Yang X, Gao Z, Li L, Wu H, Jiang Y, Su X (2013) Important role of platelets in modulating endotoxin-induced lung inflammation in CFTR-deficient mice. PLoS One 8(12):e82683
Konig B, Jaeger KE, Konig W (1994) Induction of inflammatory mediator release (12-hydroxyeicosatetraenoic acid) from human platelets by Pseudomonas aeruginosa. Int Arch Allergy Immunol 104(1):33–41
Uysal P, Tuncel T, Olmez D, Babayigit A, Karaman O, Uzuner N (2011) The role of mean platelet volume predicting acute exacerbations of cystic fibrosis in children. Ann Thorac Med 6(4):227–230
Hasselbalch HC (2012) Perspectives on chronic inflammation in essential thrombocythemia, polycythemia vera, and myelofibrosis: is chronic inflammation a trigger and driver of clonal evolution and development of accelerated atherosclerosis and second cancer? Blood 119(14):3219–3225
Schweighofer B, Testori J, Sturtzel C, Sattler S, Mayer H, Wagner O, Bilban M, Hofer E (2009) The VEGF-induced transcriptional response comprises gene clusters at the crossroad of angiogenesis and inflammation. Thromb Haemost 102(3):544–554
Taylor AW (2009) Review of the activation of TGF-beta in immunity. J Leukoc Biol 85(1):29–33
Watts KD, McColley SA (2011) Elevated vascular endothelial growth factor is correlated with elevated erythropoietin in stable, young cystic fibrosis patients. Pediatr Pulmonol 46(7):683–687
McColley SA, Stellmach V, Boas SR, Jain M, Crawford SE (2000) Serum vascular endothelial growth factor is elevated in cystic fibrosis and decreases with treatment of acute pulmonary exacerbation. Am J Respir Crit Care Med 161(6):1877–1880
Tirelli AS, Colombo C, Torresani E et al (2013) Effects of treatment in the levels of circulating cytokines and growth factors in cystic fibrosis and dialyzed patients by multi-analytical determination with a biochip array platform. Cytokine 62(3):413–420
Eickmeier O, Boom L, Schreiner F et al (2013) Transforming growth factor beta1 genotypes in relation to TGFbeta1, interleukin-8, and tumor necrosis factor alpha in induced sputum and blood in cystic fibrosis. Mediat Inflamm 2013:913135
Harris WT, Muhlebach MS, Oster RA, Knowles MR, Clancy JP, Noah TL (2011) Plasma TGF-beta(1) in pediatric cystic fibrosis: potential biomarker of lung disease and response to therapy. Pediatr Pulmonol 46(7):688–695
Zimmermann R, Koenig J, Zingsem J, Weisbach V, Strasser E, Ringwald J, Eckstein R (2005) Effect of specimen anticoagulation on the measurement of circulating platelet-derived growth factors. Clin Chem 51(12):2365–2368
Jelkmann W (2001) Pitfalls in the measurement of circulating vascular endothelial growth factor. Clin Chem 47(4):617–623
Schwarz KB, Rosensweig J, Sharma S, Jones L, Durant M, Potter C, Narkewicz MR (2003) Plasma markers of platelet activation in cystic fibrosis liver and lung disease. J Pediatr Gastroenterol Nutr 37(2):187–191
Maloney JP, Silliman CC, Ambruso DR, Wang J, Tuder RM, Voelkel NF (1998) In vitro release of vascular endothelial growth factor during platelet aggregation. Am J Physiol 275(3 Pt 2):H1054–H1061
Cattaneo M, Cerletti C, Harrison P et al (2013) Recommendations for the standardization of light transmission aggregometry: a consensus of the working party from the platelet physiology subcommittee of SSC/ISTH. J Thromb Haemost 11:1183–1189
Jonnalagadda D, Izu LT, Whiteheart SW (2012) Platelet secretion is kinetically heterogeneous in an agonist-responsive manner. Blood 120(26):5209–5216
Ollivier V, Syvannarath V, Gros A, Butt A, Loyau S, Jandrot-Perrus M, Ho-Tin-Noe B (2014) Collagen can selectively trigger a platelet secretory phenotype via glycoprotein VI. PLoS One 9(8):e104712
Kato H, Adachi S, Doi T et al (2010) Mechanism of collagen-induced release of 5-HT, PDGF-AB and sCD40L from human platelets: role of HSP27 phosphorylation via p44/p42 MAPK. Thromb Res 126(1):39–43
Coppinger JA, O’Connor R, Wynne K et al (2007) Moderation of the platelet releasate response by aspirin. Blood 109(11):4786–4792
Zhang C, Thornton MA, Kowalska MA et al (2001) Localization of distal regulatory domains in the megakaryocyte-specific platelet basic protein/platelet factor 4 gene locus. Blood 98(3):610–617
Kropf J, Schurek JO, Wollner A, Gressner AM (1997) Immunological measurement of transforming growth factor-beta 1 (TGF-beta1) in blood; assay development and comparison. Clin Chem 43(10):1965–1974
Okuda K, Kawase T, Momose M, Murata M, Saito Y, Suzuki H, Wolff LF, Yoshie H (2003) Platelet-rich plasma contains high levels of platelet-derived growth factor and transforming growth factor-beta and modulates the proliferation of periodontally related cells in vitro. J Periodontol 74(6):849–857
Salgado R, Benoy I, Bogers J, Weytjens R, Vermeulen P, Dirix L, Van Marck E (2001) Platelets and vascular endothelial growth factor (VEGF): a morphological and functional study. Angiogenesis 4(1):37–43
Salven P, Orpana A, Joensuu H (1999) Leukocytes and platelets of patients with cancer contain high levels of vascular endothelial growth factor. Clin Cancer Res 5(3):487–491
Pieroni L, Finamore F, Ronci M et al (2011) Proteomics investigation of human platelets in healthy donors and cystic fibrosis patients by shotgun nUPLC-MSE and 2DE: a comparative study. Mol BioSyst 7(3):630–639
Liem LM, Fibbe WE, van Houwelingen HC, Goulmy E (1999) Serum transforming growth factor-beta1 levels in bone marrow transplant recipients correlate with blood cell counts and chronic graft-versus-host disease. Transplantation 67(1):59–65
Di Raimondo F, Azzaro MP, Palumbo GA et al (2001) Elevated vascular endothelial growth factor (VEGF) serum levels in idiopathic myelofibrosis. Leukemia 15(6):976–980
Enjoji M, Nakamuta M, Yamaguchi K et al (2005) Clinical significance of serum levels of vascular endothelial growth factor and its receptor in biliary disease and carcinoma. World J Gastroenterol 11(8):1167–1171
Acknowledgments
We thank the cystic fibrosis patients and healthy subjects who volunteered for this study. This work was supported by Grant K08 HL035454 from the United States National Institutes of Health, National Heart Lung, and Blood Institute (JM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Rights and permissions
About this article
Cite this article
Maloney, J.P., Narasimhan, J. & Biller, J. Decreased TGF-β1 and VEGF Release in Cystic Fibrosis Platelets: Further Evidence for Platelet Defects in Cystic Fibrosis. Lung 194, 791–798 (2016). https://doi.org/10.1007/s00408-016-9925-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-016-9925-9