Abstract
Introduction
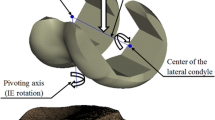
The application of a defect-size metal implant for the treatment of focal articular cartilage lesions of the femoral condyle is of potential concern resulting in cartilage damage to opposing biological structures. This in vitro study aims to determine the tibiofemoral contact pressure with a contoured articular partial femoral resurfacing device under continuous dynamic pressure loads.
Methods
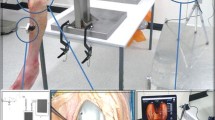
Peak and area contact pressures were determined in eight fresh-frozen cadaveric specimens using a pressure-sensitive sensor placed in the medial compartment above the menisci. All knees were tested in the untreated condition and after implantation of the prosthetic device in the weight-bearing area of the medial femoral condyle. A robotic knee simulator was used to test each knee under continuous pressure load for 400 s during 40 dynamic knee bending cycles (5°–45° flexion) with body weight ground reaction force (GRF). The GRF was adjusted to the living body weight of the cadaver donor and maintained throughout all cycles.
Results
Comparison of the untreated condition to focal inlay resurfacing showed no statistically significant differences (P ≤ 0.05) between all testing conditions. The average maximum peak contact pressure across all 40 flexion cycles increased by 5.1% after resurfacing compared to the untreated knees. The average area contact pressure essentially stayed the same (+0.9%).
Conclusion
The data suggest that resurfacing with the contoured articular prosthetic device does not pose any immediate deleterious effects to the opposing surfaces based on peak and area contact pressure in a continuous dynamic in vitro application. However, long-term in vivo effects remain to be evaluated.




Similar content being viewed by others
References
Becher C, Heyse TJ, Kron N, Ostermeier S, Hurschler C, Schofer MD, Fuchs-Winkelmann S, Tibesku CO (2009) Posterior stabilized TKA reduce patellofemoral contact pressure compared with cruciate retaining TKA in vitro. Knee Surg Sports Traumatol Arthrosc 17:1159–1165
Becher C, Huber R, Thermann H, Paessler HH, Skrbensky G (2008) Effects of a contoured articular prosthetic device on tibiofemoral peak contact pressure: a biomechanical study. Knee Surg Sports Traumatol Arthrosc 16:56–63
Beck PR, Thomas AL, Farr J, Lewis PB, Cole BJ (2005) Trochlear contact pressures after anteromedialization of the tibial tubercle. Am J Sports Med 33:1710–1715
Bingham JT, Papannagari R, Van de Velde SK, Gross C, Gill TJ, Felson DT, Rubash HE, Li G (2008) In vivo cartilage contact deformation in the healthy human tibiofemoral joint. Rheumatology (Oxford) 47:1622–1627
Bobic V (1996) Arthroscopic osteochondral autograft transplantation in anterior cruciate ligament reconstruction: a preliminary clinical study. Knee Surg Sports Traumatol Arthrosc 3:262–264
Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L (1994) Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med 331:889–895
Brown TD, Pope DF, Hale JE, Buckwalter JA, Brand RA (1991) Effects of osteochondral defect size on cartilage contact stress. J Orthop Res 9:559–567
Cicuttini F, Ding C, Wluka A, Davis S, Ebeling PR, Jones G (2005) Association of cartilage defects with loss of knee cartilage in healthy, middle-age adults: a prospective study. Arthritis Rheum 52:2033–2039
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31, 516 knee arthroscopies. Arthroscopy 13:456–460
Custers RJ, Creemers LB, van Rijen MH, Verbout AJ, Saris DB, Dhert WJ (2009) Cartilage damage caused by metal implants applied for the treatment of established localized cartilage defects in a rabbit model. J Orthop Res 27:84–90
Custers RJ, Dhert WJ, van Rijen MH, Verbout AJ, Creemers LB, Saris DB (2007) Articular damage caused by metal plugs in a rabbit model for treatment of localized cartilage defects. Osteoarthr Cartil 15:937–945
Custers RJ, Saris DB, Dhert WJ, Verbout AJ, van Rijen MH, Mastbergen SC, Lafeber FP, Creemers LB (2009) Articular cartilage degeneration following the treatment of focal cartilage defects with ceramic metal implants and compared with microfracture. J Bone Joint Surg Am 91:900–910
Eckstein F, Lemberger B, Gratzke C, Hudelmaier M, Glaser C, Englmeier KH, Reiser M (2005) In vivo cartilage deformation after different types of activity and its dependence on physical training status. Ann Rheum Dis 64:291–295
Guettler JH, Demetropoulos CK, Yang KH, Jurist KA (2004) Osteochondral defects in the human knee: influence of defect size on cartilage rim stress and load redistribution to surrounding cartilage. Am J Sports Med 32:1451–1458
Hangody L, Fules P (2003) Autologous osteochondral mosaicplasty for the treatment of full-thickness defects of weight-bearing joints: ten years of experimental and clinical experience. J Bone Joint Surg Am A 85(Suppl 2):25–32
Hangody L, Kish G, Karpati Z, Udvarhelyi I, Szigeti I, Bely M (1998) Mosaicplasty for the treatment of articular cartilage defects: application in clinical practice. Orthopedics 21:751–756
Herberhold C, Stammberger T, Faber S, Putz R, Englmeier KH, Reiser M, Eckstein F (1998) An MR-based technique for quantifying the deformation of articular cartilage during mechanical loading in an intact cadaver joint. Magn Reson Med 39:843–850
Hjelle K, Solheim E, Strand T, Muri R, Brittberg M (2002) Articular cartilage defects in 1,000 knee arthroscopies. Arthroscopy 18:730–734
Jackson DW, Lalor PA, Aberman HM, Simon TM (2001) Spontaneous repair of full-thickness defects of articular cartilage in a goat model. A preliminary study. J Bone Joint Surg Am 83-A:53–64
Kirker-Head CA, Van Sickle DC, Ek SW, McCool JC (2006) Safety of, and biological and functional response to, a novel metallic implant for the management of focal full-thickness cartilage defects: preliminary assessment in an animal model out to 1 year. J Orthop Res 24:1095–1108
Kreuz PC, Erggelet C, Steinwachs MR, Krause SJ, Lahm A, Niemeyer P, Ghanem N, Uhl M, Sudkamp N (2006) Is microfracture of chondral defects in the knee associated with different results in patients aged 40 years or younger? Arthroscopy 22:1180–1186
Lee SJ, Aadalen KJ, Malaviya P, Lorenz EP, Hayden JK, Farr J, Kang RW, Cole BJ (2006) Tibiofemoral contact mechanics after serial medial meniscectomies in the human cadaveric knee. Am J Sports Med 34:1334–1344
Mithoefer K, Williams RJ 3rd, Warren RF, Potter HG, Spock CR, Jones EC, Wickiewicz TL, Marx RG (2005) The microfracture technique for the treatment of articular cartilage lesions in the knee. A prospective cohort study. J Bone Joint Surg Am 87:1911–1920
Nehrer S, Domayer S, Dorotka R, Schatz K, Bindreiter U, Kotz R (2006) Three-year clinical outcome after chondrocyte transplantation using a hyaluronan matrix for cartilage repair. Eur J Radiol 57:3–8
O’Driscoll SW (1998) The healing and regeneration of articular cartilage. J Bone Joint Surg Am 80:1795–1812
Pena E, Calvo B, Martinez MA, Doblare M (2007) Effect of the size and location of osteochondral defects in degenerative arthritis. A finite element simulation. Comput Biol Med 37:376–387
Peterson L, Brittberg M, Kiviranta I, Akerlund EL, Lindahl A (2002) Autologous chondrocyte transplantation. Biomechanics and long-term durability. Am J Sports Med 30:2–12
Steadman JR, Rodkey WG, Rodrigo JJ (2001) Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin Orthop 391:S362–S369
Tondu B, Lopez P (2000) Modeling and control of McKibben artificial muscle robot actuators. IEEE Control Syst 20:15–38
von Skrbensky G, Huber R (2006) Vienna 2.2 knee joint simulator for long-term in vitro testing. J Biomech 39:S50
Wang Y, Ding C, Wluka AE, Davis S, Ebeling PR, Jones G, Cicuttini FM (2006) Factors affecting progression of knee cartilage defects in normal subjects over 2 years. Rheumatology (Oxf) 45:79–84
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Becher, C., Huber, R., Thermann, H. et al. Effects of a surface matching articular resurfacing device on tibiofemoral contact pressure: results from continuous dynamic flexion–extension cycles. Arch Orthop Trauma Surg 131, 413–419 (2011). https://doi.org/10.1007/s00402-010-1201-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-010-1201-5