Abstract
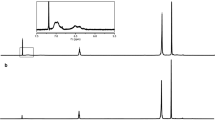
Polyisoprene (PI) is the main component of natural rubber. To imitate natural rubber and understand the function of phospholipid in natural rubber, a novel phosphatidylcholine (PC)-modified polyisoprene (PI-pc) is synthesized using a PC and a commercial PI. The PI is firstly brominated by N-bromosuccinimide, and then, diethyl malonate is introduced as a branch chain of PI. In sequence, the PC is imported into the branch chain of PI via condensation of the activated ester terminals in diethyl malonate and the amino terminals in PC to obtain the desired product PI-pc. The bulk structure of the prepared PI-pc is carefully characterized with nuclear magnetic resonance spectra (1H and 31P NMR), Fourier transform infrared spectroscopy (FT-IR), gel permeation chromatograph (GPC), and differential scanning calorimetry (DSC). The introduction of PC to the branch chain of PI increases the molecular weight and also the glass transition (Tg) of the PI. The increment of Tgs and melting enthalpy for the PI-pcs from both solution and emulsion indicates that the attached PC is beneficial to the self-assembly of PI chains and thus promotes the crystallization. Our present work provides a new method for importing PC to PI to imitate natural rubber, and also, the results could be a footstone for us to explore the effect of phospholipid on the property of natural rubber.





Similar content being viewed by others
References
Shah AA, Hasan F, Shah Z, Kanwal N, Zeb S (2013) Biodegradation of natural and synthetic rubbers: a review. Int Biodeter Biodegr 83:145–157
Tuampoemsab S, Nimpaiboon A, Sakdapipanich JT (2015) Quantitative analysis of isoprene units in natural rubber and synthetic polyisoprene using 1H-NMR spectroscopy with an internal standard. Polym Test 43:21–26
Toki S, Hsiao BS, Amnuaypornsri S, Sakdapipanich J (2009) New insights into the relationship between network structure and strain-induced crystallization in un-vulcanized and vulcanized natural rubber by synchrotron X-ray diffraction. Polymer 50:2142–2148
Kosugi K, Sutthangkul R, Chaikumpollert O, Yamamoto Y, Sakdapipanich J, Isono Y, Kawahara S (2012) Preparation and characterization of natural rubber with soft nanomatrix structure. Colloid Polym Sci 290:1457–1462
Nishi T, Nukaga H, Fujinami S, Nakajima K (2007) Nanomechanical mapping of carbon black reinforced natural rubber by atomic force microscopy. Chin J Polym Sci 25:35–41
Ouardad S, Bakleh ME, Kostjuk SV, Ganachaud F, Puskas JE, Deffieux A, Peruch F (2012) Bio-inspired cationic polymerization of isoprene and analogues: state-of-the-art. Polym Int 61:149–156
Liu J, Wu S, Tang Z, Lin T, Guo B, Huang G (2015) New evidence disclosed for networking in natural rubber by dielectric relaxation spectroscopy. Soft Matter 11:2290–2299
Siler DJ, Cornish K, Hamilton RG (1996) Absence of cross-reactivity of IgE antibodies from subjects allergic to Hevea brasiliensis latex with a new source of natural rubber latex from guayule (Parthenium argentatum). J Allergy Clin Immun 98:895–902
Mooibroek H, Cornish K (2000) Alternative sources of natural rubber. Appl Microbiol Biot 53:355–365
Tangpakdee J, Tanaka Y (1998) Long-chain polyprenols and rubber in young leaves of Hevea brasiliensis. Phytochemistry 48:447–450
Tangpakdee J, Tanaka Y (1997) Characterization of sol and gel in Hevea natural rubber. Rubber Chem Technol 70:707–713
Eng A, Tanaka Y, Gan S (1992) FTIR studies on amino groups in purified Hevea rubber. J Nat Rubber Res 7:152
Tarachiwin L, Sakdapipanich J, Ute K, Kitayama T, Bamba T, Fukusaki E-i, Kobayashi A, Tanaka Y (2005) Structural characterization of α-terminal group of natural rubber. 1. Decomposition of branch-points by lipase and phosphatase treatments. Biomacromolecules 6:1851–1857
Tarachiwin L, Sakdapipanich J, Ute K, Kitayama T, Tanaka Y (2005) Structural characterization of α-terminal group of natural rubber. 2. Decomposition of branch-points by phospholipase and chemical treatments. Biomacromolecules 6:1858–1863
Amnuaypornsri S, Tarachiwin L, Sakdapipanich JT (2010) Character of long-chain branching in highly purified natural rubber. J Appl Polym Sci 115:3645–3650
Amnuaypornsri S, Sakdapipanich J, Toki S, Hsiao BS, Ichikawa N, Tanaka Y (2008) Strain-induced crystallization of natural rubber: effect of proteins and phospholipids. Rubber Chem Technol 81:753–766
Carretero–González J, Ezquerra TA, Amnuaypornsri S, Toki S, Verdejo R, Sanz A, Sakdapipanich J, Hsiao BS, López–Manchado MA (2010) Molecular dynamics of natural rubber as revealed by dielectric spectroscopy: The role of natural cross–linking. Soft Matter 6:3636–3642
Shao F, Ni XF, Shen ZQ (2012) Preparation of amphiphilic graft copolymer with polyisoprene backbone by combination of anionic polymerization and “click” reaction. Chin Chem Lett 23:347–350
Derouet D, Tran QN, Thuc HH (2007) Synthesis of N,N-diethyldithiocarbamate functionalized 1,4-polyisoprene, from natural rubber and synthetic 1,4-polyisoprene. Eur Polym J 43:1806–1824
Georgopanos P, Rangou S, Haenelt TG, Abetz C, Meyer A, Filiz V, Handge UA, Abetz V (2014) Analysis of glass transition and relaxation processes of low molecular weight polystyrene-b-polyisoprene diblock copolymers. Colloid Polym Sci 292:1877–1891
Jeon H, Rameshwaram J, Kim G, Weinkauf D (2003) Characterization of polyisoprene–clay nanocomposites prepared by solution blending. Polymer 44:5749–5758
Fu X, Huang G, Xie Z, Xing W (2015) New insights into reinforcement mechanism of nanoclay-filled isoprene rubber during uniaxial deformation by in situ synchrotron X-ray diffraction. RSC Adv 5:25171–25182
Tsujimoto T, Toshimitsu K, Uyama H, Takeno S, Nakazawa Y (2014) Maleated trans-1,4-polyisoprene from Eucommia ulmoides Oliver with dynamic network structure and its shape memory property. Polymer 55:6488–6493
Yu J, Tonpheng B, Gröbner G, Andersson O (2012) A MWCNT/polyisoprene composite reinforced by an effective load transfer reflected in the extent of polymer coating. Macromolecules 45:2841–2849
Liu C, Bonaccurso E, Sokuler M, Auernhammer GNK, Butt HJ (2009) Dynamic wetting of polyisoprene melts: influence of the end group. Langmuir 26:2544–2549
Zhang X, Tan D, Li J, Tan H, Fu Q (2011) Synthesis and hemocompatibity evaluation of segmented polyurethane end-capped with both a fluorine tail and phosphatidylcholine polar headgroups. Biofouling 27:919–930
Ojha U, Rajkhowa R, Agnihotra SR, Faust R (2008) A new general methodology for the syntheses of end-functional polyisobutylenes by nucleophilic substitution reactions. Macromolecules 41:3832–3841
Nakason C, Kaesaman A, Supasanthitikul P (2004) The grafting of maleic anhydride onto natural rubber. Polym Test 23:35–41
Ponce M, Ramirez R (1981) Mixing process of natural and synthetic polyisoprene rubbers. Rubber Chem Technol 54:211–226
Sun X-L, Biswas N, Kai T, Dai Z, Dluhy RA, Chaikof EL (2006) Membrane-mimetic films of asymmetric phosphatidylcholine lipid bolaamphiphiles. Langmuir 22:1201–1208
Pan Z, Hao H, Zhao Y, Li J, Tan H, Fu Q (2015) Preparation of hydrocarbon/fluorocarbon double-chain phospholipid polymer brusheson polyurethane films by ATRP. Colloids Surf B: Biointerfaces 128:36–43
Amnuaypornsri S, Sakdapipanich J, Tanaka Y (2009) Green strength of natural rubber: the origin of the stress–strain behavior of natural rubber. J Appl Polym Sci 111:2127–2133
Shrestha LK, Strzelczyk KM, Shrestha RG, Ichikawa K, Aramaki K, Hill JP, Ariga K (2015) Nonionic amphiphile nanoarchitectonics: self-assembly into micelles and lyotropic liquid crystals. Nanotechnology 26:204002
Dong R, Hao J (2010) Complex fluids of poly (oxyethylene) monoalkyl ether nonionic surfactants. Chem Rev 110:4978–5022
Acknowledgments
Financial supports from National Natural Science Foundation of China (51333003, 51273124) and National Science Fund for Distinguished Young Scholars (51425305) are acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chu, H., Song, Y., Li, J. et al. A novel phosphatidylcholine-modified polyisoprene: synthesis and characterization. Colloid Polym Sci 294, 433–439 (2016). https://doi.org/10.1007/s00396-015-3798-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3798-y