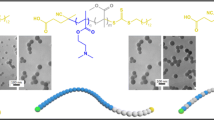
Abstract
In order to understand the effect of molecular structures of emulsifiers on the cationic emulsion copolymerization of styrene (St) and butyl acrylate (BA) in details, hexadecyl trimethyl ammonium bromide (CTAB), methacryloxy ethyl hexadecyl dimethyl ammonium bromide (DMHB), ethanediyl bis(hexadecyl dimethyl ammonium bromide) (G16-2-16), and maleic acid diethyl bis(hexadecyl dimethyl ammonium bromide) (P16-8-16) were used as emulsifiers. TEM photos, instantaneous conversions, and colloidal features, such as the particle size, surface charge density, and glass transition temperature (Tg), were measured. Compared with polymerizable emulsifiers (DMHB and P16-8-16), non-polymerizable emulsifiers (CTAB and G16-2-16) produced more uniform nanospheres. In the reactions with polymerizable emulsifiers, higher reaction rates, lower surface charge densities, and lower Tg were observed. Compared with the single-chain emulsifiers, the double-chain emulsifiers resulted in lower reaction rates, bigger particle sizes, better monodispersities, and higher surface charge densities.






Similar content being viewed by others
References
Teixeira H, Dubernet C, Puisieux F, Benita S, Couvreur P (1999) Submicron cationic emulsions as a new delivery system for oligonucleotides. Pharm Res 16:30–36
Tae W, Hesson C, Ick K, Ha S, Seo J (2000) In vivo gene transfer to the mouse nasal cavity mucosa using a stable cationic lipid emulsion. Mol Cells 10:142–147
Danny G, Taher N, Gregory L, Jean K, Simon B (2005) The design and evaluation of a novel targeted drug delivery system using cationic emulsion-antibody conjugates. J Controlled Release 108:418–432
Gleason PA, Piccoline MA (1986) U.S. Patent No. 4,626,363. Washington, DC: U.S. Patent and Trademark Office
Dillon KL, Lamb JA (1991) U.S. Patent No. 5,049,377. Washington, DC: U.S. Patent and Trademark Office
Baumgen H, Bomer B, Konig J, Mottweiler R, Probst J (1989). U.S. Patent No. 4,806,591. Washington, DC: U.S. Patent and Trademark Office
Hou HC, Hsin YH (1997) Stabilizing effect of the cationic surfactant (CPB) in emulsion polymerization. Polym Bull 38:295–302
Jose R, Jacqueline F (2003) Semi-continuous seeded cationic emulsion polymerization of styrene: the effect of the concentration and type of cationic surfactant. J Polym Sci Part A Polym Chem 41:2322–2334
Capek I (2001) Microemulsion polymerization of styrene in the presence of a cationic emulsifier. Adv Colloid Interfac 92:195–233
Hu XF, Zhang JY, Yang WT (2009) Preparation of transparent polystyrene nano-latexes by an UV-induced routine emulsion polymerization. Polymer 50:141–147
Kong XZ, Zhu XL, Jiang XB, Li XF (2009) Preparation and full characterization of cationic latex of styrene-butyl acrylate. Polymer 50:4220–4227
Zou D, Li XF, Zhu XL, Kong XZ (2012) Preparation of cationic latexes of poly (styrene-co-butyl acrylate) and their properties evolution in latex dilution. Chinese J Polym Sci 30:278–286
Zhang JZ, Jia C, Chai SG, Yu H, Chen H, Xue YD, Chen Z, Zou QC (2010) Effect of Gemini surfactant on St/MMA nano cationic emulsion. Chinese J Colloid Polym 28:106–108
Chen LF, Xie HQ, Yang L, Yu W (2008) Application of cationic Gemini surfactant in preparing multi-walled carbon nanotube contained nanofluids. Colloids Surf A 330:176–179
Bernd T (2005) Polymerization of styrene in microemulsion with cationic surfactant mixtures. Colloid Polym Sci 283:421–430
Xu F, Zhang Q, Gao ZN (2012) Simple one-step synthesis of gold nanoparticles with controlled size using cationic Gemini surfactants as ligands: effect of the variations in concentrations and tail lengths. Colloids Surf A 417:201–210
Chen H, Zou QC, Yu H, Zhang JZ, Chai SG (2012) Synthesis and characterization of size controllable cationic polystyrene nanoparticles. Chin Int J Appl Chem 29:29–36
Michael D, Bernd T (1998) Polymerization of styrene in ternary microemulsion using cationic Gemini surfactants. Langmuir 14:800–807
Dipti S, Tyagi VK (2006) Cationic Gemini surfactants: a review. J Oleo Sci 55:381–390
Su X, Wang BQ, Lu ZY, Wei LM, Feng YJ (2011) A new cationic surfmer: synthesis and surface activities. J Surfactants Deterg 14:73–76
Liu JJ, Ding YS, Yu Y, Wu SY, Feng WG (2010) Effect of two kinds of imidazolium ionic liquids on the microemulsion polymerization of methyl methylacrylate. Macromol Symp 298:167–173
Yang YN, Zhang LB, Zhu XL, Kong XZ (2012) Preparation of cationic functional polymer latexes and measurement of involatile monomer conversion. J Appl Polym Sci 124:3662–3668
Denise J, Sherrington DC (1997) Novel polymerizable mono and divalent quaternary ammonium cationic surfactant: 2. Surface active properties and use in emulsion polymerization. Polymer 38:1427–1438
Michael D, Wim PH, Bernd T (1998) Copolymerization behavior and structure of styrene and polymerizable surfactants in three component cationic microemulison. Macromolecules 31:272–280
Jin LQ, Liu ZL, Xu QH, Li YC (2006) Preparation of soap-free cationic emulsion using polymerizable surfactant. J Appl Polym Sci 99:1111–1116
Chern CS (2006) Emulsion polymerization mechanisms and kinetics. Prog Polym Sci 31:443–486
Montoya GA, Sherrington DC, Schoonbrood H, Asua JM (1999) Reactive surfactants in heterophase polymerization, XXIV. Emulsion polymerization of styrene with maleate and succinate containing cationic surfactants. Polymer 40:1359–1366
Paul A, Gregory G (2012) Structure of polymerizable surfactant micelles: insights from neutron scattering. Adv Colloid Interface Sci 179:14–21
Masahiko A, Kazuyuki T, Takaaki K, Koji T, Takahiro O, Hideki S (2006) Polymerizable cationic Gemini surfactant. Langmuir 22:8293–8297
Li R, Yan F, Zhang J, Xu C, Wang J (2014) The self-assembly properties of a series of polymerizable cationic Gemini surfactants: effect of the acryloxyl group. Colloids Surf A 444:276–282
You Y, Zheng O, Qiu Y, Zheng YH, Zhao JX (2001) Synthesis of alkanediyl-α, w-bis (dimethyl dodecylammonium bromide) and their micelle formation in aqueous solutions. Acta Phys-Chim Sin 17:74–78
Zheng DB, Fang KJ, Zhang X, Fu SH (2008) Synthesis and application of a polymerizable cationic surfactant DMDB for pigment dispersions. Fine Chemicals 25:143–146
Jose R, Jacqueline F (2010) The role of cationic monomers in emulsion polymerization. Eur Polym J 46:1106–1110
Jose R, Alvaro C, Jacqueline F (2006) Kinetics of the batch cationic emulsion polymerization of styrene: a comparative study with the anionic case. J Polym Sci Part A Polym Chem 44:4461–4478
Gong YK, Kenichi N, Xu RL (2001) A novel method to determine effective charge of polystyrene latex particles in aqueous dispersion. Langmuir 17:2889–2892
Smith WV (1948) The kinetics of styrene emulsion polymerization. J Am Chem Soc 70:3695–3702
Smith WV (1949) Chain initiation in styrene emulsion polymerization. J Am Chem Soc 71:4077–4082
Gardon JL (1968) Emulsion polymerization. I. Recalculation and extension of the Smith-Ewart theory. J Polym Sci Part A Polym Chem 6:623–641
Gardon JL (1968) Emulsion polymerization. II. Review of experimental data in the context of the revised Smith-Ewart theory. J Polym Sci Part A Polym Chem 6:643–664
Urquiola MB, Dimonie VL, Sudol ED, Elaasser MS (1992) Emulsion polymerization of vinyl acetate using a polymerizable surfactant. J Polym Sci Part A Polym Chem 30:2631–2644
Yan C, Cheng S, Feng L (1992) Kinetics and mechanism of emulsifier-free emulsion copolymerization: styrene-methyl methacrylate-acrylic acid system. J Polym Sci Part A Polym Chem 37:2649–2656
Acknowledgments
This work has been supported by (1) National Natural Science Foundation of China, Project No. 51173086; (2) National Key Technology R&D Program, Grant No. 2014BAC13B02 and 2014BAE01B01; and (3) Industrialization Projects of Major Independent Innovation Achievements of Shandong Province, Grant No. 2012ZHZX1A0914.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, F., Fang, K. Effect of molecular structures of Gemini and polymerizable emulsifiers on cationic emulsion copolymerization of styrene and butyl acrylate. Colloid Polym Sci 292, 1449–1455 (2014). https://doi.org/10.1007/s00396-014-3212-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-014-3212-1