Abstract
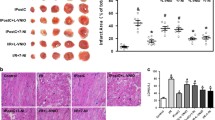
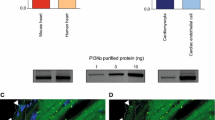
Protein kinase G type I (PKGI) plays a critical role in survival signaling of pre- and postconditioning downstream of cardiac cGMP. However, it is unclear whether PKGI exerts its protective effects in the cardiomyocyte or if other cardiac cell types are involved, and whether nitric oxide (NO) metabolism can target cardiomyocyte mitochondria independently of cGMP/PKGI. We tested whether protection against reperfusion injury by ischemic postconditioning (IPost), soluble guanylyl cyclase (sGC) activation and inhibition, adenosine A2B receptor (A2BAR) agonist, phosphodiesterase type-5 (PDE-5) inhibitor, or mitochondria-targeted S-nitrosothiol (MitoSNO) was affected by a cardiomyocyte-specific ablation of the PKGI gene in the mouse (CMG-KO). In situ hearts underwent 30 min of regional ischemia followed by 2 h of reperfusion. As expected, in CMG-CTRs all interventions at early reperfusion lead to profound infarct size reduction: IPost (six cycles of 10-s reperfusion and 10-s coronary occlusion) with or without treatment with the sGC inhibitor ODQ, treatment with the specific sGC activator BAY58-2667 (BAY58), the selective A2BAR agonist BAY60-6583 (BAY60), PDE-5 inhibitor sildenafil, and MitoSNO. MitoSNO accumulates within mitochondria, driven by the membrane potential, where it generates NO· and S-nitrosates thiol proteins. In contrast, the hearts of CMG-KO animals were not protected by BAY58 and sildenafil, whereas the protective effects of IPost, IPost with ODQ, BAY60, and MitoSNO were unaffected by the lack of PKGI. Taken together, PKGI is important for the protection against ischemia reperfusion injury afforded by sGC activation or PDE-5 inhibition. However, the beneficial effects of IPost, activation of the A2BAR, as well as the direct effects via mitochondrial S-nitrosation do not depend on PKGI in cardiomyocytes.





Similar content being viewed by others
References
Boengler K, Hilfiker-Kleiner D, Heusch G, Schulz R (2010) Inhibition of permeability transition pore opening by mitochondrial STAT3 and its role in myocardial ischemia/reperfusion. Basic Res Cardiol 105:771–785. doi:10.1007/s00395-010-0124-1
Burley DS, Ferdinandy P, Baxter GF (2007) Cyclic GMP and protein kinase-G in myocardial ischaemia-reperfusion: opportunities and obstacles for survival signaling. Br J Pharmacol 152:855–869. doi:10.1038/sj.bjp.0707409
Costa AD, Garlid KD, West IC, Lincoln TM, Downey JM, Cohen MV, Critz SD (2005) Protein kinase G transmits the cardioprotective signal from cytosol to mitochondria. Circ Res 97:329–336. doi:10.1161/01.RES.0000178451.08719.5b
Francis SH, Busch JL, Corbin JD, Sibley D (2010) cGMP-dependent protein kinases and cGMP phosphodiesterases in nitric oxide and cGMP action. Pharmacol Rev 62:525–563. doi:10.1124/pr.110.002907
Garcia-Dorado D, Agulló L, Sartorio CL, Ruiz-Meana M (2009) Myocardial protection against reperfusion injury: the cGMP pathway. Thromb Haemost 101:635–642. doi:10.1160/TH08-11-0764
Gorbe A, Giricz Z, Szunyog A, Csont T, Burley DS, Baxter GF, Ferdinandy P (2010) Role of cGMP-PKG signaling in the protection of neonatal rat cardiac myocytes subjected to simulated ischemia/reoxygenation. Basic Res Cardiol 105:643–650. doi:10.1007/s00395-010-0097-0
Grube K, Rüdebusch J, Xu Z, Böckenholt T, Methner C, Müller T, Cuello F, Zimmermann K, Yang X, Felix SB, Cohen MV, Downey JM, Krieg T (2011) Evidence for an intracellular localization of the adenosine A2B receptor in rat cardiomyocytes. Basic Res Cardiol 106:385–396. doi:10.1007/s00395-011-0151-6
Heusch G, Boengler K, Schulz R (2008) Cardioprotection: nitric oxide, protein kinases, and mitochondria. Circulation 118:1915–1919. doi:10.1161/CIRCULATIONAHA.108.805242
Heusch G, Boengler K, Schulz R (2010) Inhibition of mitochondrial permeability transition pore opening: the Holy Grail of cardioprotection. Basic Res Cardiol 105:151–154. doi:10.1007/s00395-009-0080-9
Heusch G, Musiolik J, Gedik N, Skyschally A (2011) Mitochondrial STAT3 activation and cardioprotection by ischemic postconditioning in pigs with regional myocardial ischemia/reperfusion. Circ Res 109:1302–1308. doi:10.1161/CIRCRESAHA.111.255604
Hofmann F, Bernhard D, Lukowski R, Weinmeister P (2009) cGMP regulated protein kinases (cGK). Handb Exp Pharmacol 191:137–162. doi:10.1007/978-3-540-68964-5_8
James AM, Sharpley MS, Manas AR, Frerman FE, Hirst J, Smith RA, Murphy MP (2007) Interaction of the mitochondria-targeted antioxidant MitoQ with phospholipid bilayers and ubiquinone oxidoreductases. J Biol Chem 282:14708–14718. doi:10.1074/jbc.M611463200
Korkmaz S, Radovits T, Barnucz E, Hirschberg K, Neugebauer P, Loganathan S, Veres G, Páli S, Seidel B, Zöllner S, Karck M, Szabó G (2009) Pharmacological activation of soluble guanylate cyclase protects the heart against ischemic injury. Circulation 120:677–686. doi:10.1161/CIRCULATIONAHA.109.870774
Krieg T, Liu Y, Rütz T, Methner C, Yang XM, Dost T, Felix SB, Stasch JP, Cohen MV, Downey JM (2009) BAY 58–2667, a nitric oxide-independent guanylyl cyclase activator, pharmacologically post-conditions rabbit and rat hearts. Eur Heart J 30:1607–1613. doi:10.1093/eurheartj/ehp143
Kuno A, Solenkova NV, Solodushko V, Dost T, Liu Y, Yang XM, Cohen MV, Downey JM (2008) Infarct limitation by a protein kinase G activator at reperfusion in rabbit hearts is dependent on sensitizing the heart to A2b agonists by protein kinase C. Am J Physiol Heart Circ Physiol 295:H1288–H1295. doi:10.1152/ajpheart.00209.2008
Li Q, Guo Y, Wu W-J, Ou Q, Zhu X, Tan W, Yuan F, Chen N, Dawn B, Luo L, O’Brian E, Bolli R (2011) Gene transfer as a strategy to achieve permanent cardioprotection I: rAAV-mediated gene therapy with inducible nitric oxide synthase limits infarct size 1 year later without adverse functional consequences. Basic Res Cardiol 106:1355–1366. doi:10.1007/s00395-011-0207-7
Lukowski R, Rybalkin SD, Loga F, Leiss V, Beavo JA, Hofmann F (2010) Cardiac hypertrophy is not amplified by deletion of cGMP-dependent protein kinase I in cardiomyocytes. Proc Natl Acad Sci USA 107:5646–5651. doi:10.1073/pnas.1001360107
Methner C, Schmidt K, Cohen MV, Downey JM, Krieg T (2010) Both A2a and A2b adenosine receptors at reperfusion are necessary to reduce infarct size in mouse hearts. Am J Physiol Heart Circ Physiol 299:H1262–H1264. doi:10.1152/ajpheart.00181.2010
Penna C, Cappello S, Mancardi D, Raimondo S, Rastaldo R, Gattullo D, Losano G, Pagliaro P (2006) Post-conditioning reduces infarct size in the isolated rat heart: role of coronary flow and pressure and the nitric oxide/cGMP pathway. Basic Res Cardiol 101:168–179. doi:10.1007/s00395-005-0543-6
Prime TA, Blaikie FH, Evans C, Nadtochiy SM, James AM, Dahm CC, Vitturi DA, Patel RP, Hiley CR, Abakumova I, Requejo R, Chouchani ET, Hurd TR, Garvey JF, Taylor CT, Brookes PS, Smith RA, Murphy MP (2009) A mitochondria-targeted S-nitrosothiol modulates respiration, nitrosates thiols, and protects against ischemia-reperfusion injury. Proc Natl Acad Sci USA 106:10764–10769. doi:10.1073/pnas.0903250106
Salloum FN, Abbate A, Das A, Houser JE, Mudrick CA, Qureshi IZ, Hoke NN, Roy SK, Brown WR, Prabhakar S, Kukreja RC (2008) Sildenafil (Viagra) attenuates ischemic cardiomyopathy and improves left ventricular function in mice. Am J Physiol Heart Circ Physiol 294:H1398–H1406. doi:10.1152/ajpheart.91438.2007
Sips PY, Brouckaert P, Ichinose F (2011) The alpha1 isoform of soluble guanylate cyclase regulates cardiac contractility but is not required for ischemic preconditioning. Basic Res Cardiol 106:635–643. doi:10.1007/s00395-011-0167-y
Sun J, Aponte AM, Kohr MJ, Tong G, Steenbergen C, Murphy E (2012) Essential role of nitric oxide in acute ischemic preconditioning: S-Nitros(yl)ation versus sGC/cGMP/PKG signaling? Free Radic Biol Med 54:105–112. doi:10.1016/j.freeradbiomed.2012.09.005
Sun J, Murphy E (2010) Protein S-nitrosylation and cardioprotection. Circ Res 106:285–296. doi:10.1161/CIRCRESAHA.109.209452
Wegener JW, Nawrath H, Wolfsgruber W, Kühbandner S, Werner C, Hofmann F, Feil R (2002) cGMP-dependent protein kinase I mediates the negative inotropic effect of cGMP in the murine myocardium. Circ Res 90:18–20. doi:10.1161/hh0102.103222
Wollert KC, Fiedler B, Gambaryan S, Smolenski A, Heineke J, Butt E, Trautwein C, Lohmann SM, Drexler H (2002) Gene transfer of cGMP-dependent protein kinase I enhances the antihypertrophic effects of nitric oxide in cardiomyocytes. Hypertension 39:87–92. doi:10.1161/hy1201.097292
Acknowledgments
We thank Ines Suhr, Institute of Clinical Chemistry, University of Greifswald, for part of the troponin analysis. BAY60 and BAY58 were kindly provided by Bayer Healthcare, Wuppertal, Germany. The study was supported by grants from the Deutsche Forschungsgemeinschaft (T.K., R.L., and F.H), the Academic Research Collaboration Program of the British Council and the German Academic Exchange Service (T.K. and K.G.), the Isaac Newton Trust, Trinity College, Cambridge (T.K. and C.M.), and the Eliteprogramm für Postdocs of the Baden-WürttembergStiftung (R.L.).
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Methner, C., Lukowski, R., Grube, K. et al. Protection through postconditioning or a mitochondria-targeted S-nitrosothiol is unaffected by cardiomyocyte-selective ablation of protein kinase G. Basic Res Cardiol 108, 337 (2013). https://doi.org/10.1007/s00395-013-0337-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-013-0337-1