Abstract
Purpose
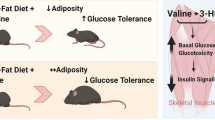
l-alanine (Ala) and l-arginine (Arg) have been reported to regulate pancreatic β-cell physiology and to prevent body fat accumulation in diet-induced obesity. Here, we assessed growth and adiposity parameters, glucose tolerance, insulin secretion and the expression of insulin and nutrient-regulated proteins in monosodium glutamate (MSG)-obese mice supplemented with either Ala or Arg.
Methods
Male newborn C57Bl/6 mice received a daily subcutaneous injection of MSG or saline solution (CTL group), during the first 6 days of life. From 30 to 90 days of age, MSG and CTL mice received or not 2.55 % Ala (CAla or MArg groups) or 1.51 % Arg-HCl (CArg or MArg groups) in their drinking water.
Results
Adult MSG mice displayed higher adiposity associated with lower phosphorylation of the adipogenic enzyme, ACC, in adipose tissue. Glucose intolerance in MSG mice was linked to lower insulin secretion and to lower expression of IRβ in adipose tissue, as well as AS160 phosphorylation in skeletal muscle. Perigonadal fat depots were smaller in Ala and Arg mice, while retroperitoneal fat pads were decreased by Ala supplementation only. Both Ala and Arg improved fed-state glycemia as well as IRβ and pAS160 content, but only Ala led to improved glucose tolerance and insulin secretion. Adipostatic signals were increased in MAla mice, as indicated by enhanced AMPK phosphorylation and pACC content in fat depots.
Conclusions
Ala supplementation led to more pronounced metabolic improvements compared to Arg, possibly due to suppression of lipogenesis through activation of the AMPK/ACC pathway.






Similar content being viewed by others
References
Joo JK, Lee KS (2014) Pharmacotherapy for obesity. J Menopausal Med 20(3):90–96. doi:10.6118/jmm.2014.20.3.90
Cerf ME (2013) Beta cell dysfunction and insulin resistance. Front Endocrinol 4:37. doi:10.3389/fendo.2013.00037
Wu G, Morris SM Jr (1998) Arginine metabolism: nitric oxide and beyond. Biochem J 336(Pt 1):1–17
Popov D, Costache G, Georgescu A, Enache M (2002) Beneficial effects of l-arginine supplementation in experimental hyperlipemia-hyperglycemia in the hamster. Cell Tissue Res 308(1):109–120. doi:10.1007/s00441-001-0509-4
Fu WJ, Haynes TE, Kohli R, Hu J, Shi W, Spencer TE, Carroll RJ, Meininger CJ, Wu G (2005) Dietary l-arginine supplementation reduces fat mass in Zucker diabetic fatty rats. J Nutr 135(4):714–721
Jobgen W, Meininger CJ, Jobgen SC, Li P, Lee MJ, Smith SB, Spencer TE, Fried SK, Wu G (2009) Dietary l-arginine supplementation reduces white fat gain and enhances skeletal muscle and brown fat masses in diet-induced obese rats. J Nutr 139(2):230–237. doi:10.3945/jn.108.096362
Monti LD, Setola E, Lucotti PC, Marrocco-Trischitta MM, Comola M, Galluccio E, Poggi A, Mammi S, Catapano AL, Comi G, Chiesa R, Bosi E, Piatti PM (2012) Effect of a long-term oral l-arginine supplementation on glucose metabolism: a randomized, double-blind, placebo-controlled trial. Diabetes Obes Metab 14(10):893–900. doi:10.1111/j.1463-1326.2012.01615.x
Jobgen WS, Fried SK, Fu WJ, Meininger CJ, Wu G (2006) Regulatory role for the arginine-nitric oxide pathway in metabolism of energy substrates. J Nutr Biochem 17(9):571–588. doi:10.1016/j.jnutbio.2005.12.001
Mohan S, Patel H, Bolinaga J, Soekamto N (2013) AMP-activated protein kinase regulates l-arginine mediated cellular responses. Nutr Metab 10(1):40. doi:10.1186/1743-7075-10-40
Kahn BB, Alquier T, Carling D, Hardie DG (2005) AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab 1(1):15–25. doi:10.1016/j.cmet.2004.12.003
Smith PA, Sakura H, Coles B, Gummerson N, Proks P, Ashcroft FM (1997) Electrogenic arginine transport mediates stimulus-secretion coupling in mouse pancreatic beta-cells. J Physiol 499(3):625–635
Krause MS, McClenaghan NH, Flatt PR, de Bittencourt PI, Murphy C, Newsholme P (2011) l-arginine is essential for pancreatic beta-cell functional integrity, metabolism and defense from inflammatory challenge. J Endocrinol 211(1):87–97. doi:10.1530/JOE-11-0236
Leiss V, Flockerzie K, Novakovic A, Rath M, Schonsiegel A, Birnbaumer L, Schurmann A, Harteneck C, Nurnberg B (2014) Insulin secretion stimulated by l-arginine and its metabolite l-ornithine depends on Galphai2. Am J Physiol Endocrinol Metab 307(9):E800–E812. doi:10.1152/ajpendo.00337.2014
Mullooly N, Vernon W, Smith DM, Newsholme P (2014) Elevated levels of branched-chain amino acids have little effect on pancreatic islet cells, but l-arginine impairs function through activation of the endoplasmic reticulum stress response. Exp Physiol 99(3):538–551. doi:10.1113/expphysiol.2013.077495
Prada PO, Hirabara SM, de Souza CT, Schenka AA, Zecchin HG, Vassallo J, Velloso LA, Carneiro E, Carvalheira JB, Curi R, Saad MJ (2007) L-glutamine supplementation induces insulin resistance in adipose tissue and improves insulin signalling in liver and muscle of rats with diet-induced obesity. Diabetologia 50(9):1949–1959. doi:10.1007/s00125-007-0723-z
Freudenberg A, Petzke KJ, Klaus S (2013) Dietary l-leucine and l-alanine supplementation have similar acute effects in the prevention of high-fat diet-induced obesity. Amino Acids 44(2):519–528. doi:10.1007/s00726-012-1363-2
McClenaghan NH, Barnett CR, Flatt PR (1998) Na+ cotransport by metabolizable and nonmetabolizable amino acids stimulates a glucose-regulated insulin-secretory response. Biochem Biophys Res Commun 249(2):299–303. doi:10.1006/bbrc.1998.9136
Sener A, Malaisse WJ (2002) The stimulus-secretion coupling of amino acid-induced insulin release. Insulinotropic action of l-alanine. Biochim Biophys Acta 1573(1):100–104
Brennan L, Shine A, Hewage C, Malthouse JP, Brindle KM, McClenaghan N, Flatt PR, Newsholme P (2002) A nuclear magnetic resonance-based demonstration of substantial oxidative l-alanine metabolism and l-alanine-enhanced glucose metabolism in a clonal pancreatic beta-cell line: metabolism of l-alanine is important to the regulation of insulin secretion. Diabetes 51(6):1714–1721
Cunningham GA, McClenaghan NH, Flatt PR, Newsholme P (2005) l-Alanine induces changes in metabolic and signal transduction gene expression in a clonal rat pancreatic beta-cell line and protects from pro-inflammatory cytokine-induced apoptosis. Clin Sci 109(5):447–455. doi:10.1042/CS20050149
Olney JW, Sharpe LG (1969) Brain lesions in an infant rhesus monkey treated with monsodium glutamate. Science 166(3903):386–388
Matyskova R, Maletinska L, Maixnerova J, Pirnik Z, Kiss A, Zelezna B (2008) Comparison of the obesity phenotypes related to monosodium glutamate effect on arcuate nucleus and/or the high fat diet feeding in C57BL/6 and NMRI mice. Physiol Res/Acad Sci Bohemoslov 57(5):727–734
Hirata AE, Alvarez-Rojas F, Carvalheira JB, Carvalho CR, Dolnikoff MS, Abdalla Saad MJ (2003) Modulation of IR/PTP1B interaction and downstream signaling in insulin sensitive tissues of MSG-rats. Life Sci 73(11):1369–1381
Nardelli TR, Ribeiro RA, Balbo SL, Vanzela EC, Carneiro EM, Boschero AC, Bonfleur ML (2011) Taurine prevents fat deposition and ameliorates plasma lipid profile in monosodium glutamate-obese rats. Amino Acids 41(4):901–908. doi:10.1007/s00726-010-0789-7
Lubaczeuski C, Balbo SL, Ribeiro RA, Vettorazzi JF, Santos-Silva JC, Carneiro EM, Bonfleur ML (2015) Vagotomy ameliorates islet morphofunction and body metabolic homeostasis in MSG-obese rats. Braz J Med Biol Res Revista brasileira de pesquisas medicas e biologicas/Sociedade Brasileira de Biofisica [et al] 0:0. doi:10.1590/1414-431X20144340
Lucinei Balbo S, Gravena C, Bonfleur ML, de Freitas Mathias PC (2000) Insulin secretion and acetylcholinesterase activity in monosodium l-glutamate-induced obese mice. Horm Res 54(4):186–191. doi:10.1159/000053257
Balbo SL, Bonfleur ML, Carneiro EM, Amaral ME, Filiputti E, Mathias PC (2002) Parasympathetic activity changes insulin response to glucose and neurotransmitters. Diabetes Metab 28(6.2):3S13–3S17 (discussion 13S108-112)
Nagata M, Suzuki W, Iizuka S, Tabuchi M, Maruyama H, Takeda S, Aburada M, Miyamoto K (2006) Type 2 diabetes mellitus in obese mouse model induced by monosodium glutamate. Exp Anim/Jpn Assoc Lab Anim Sci 55(2):109–115
Ribeiro RA, Balbo SL, Roma LP, Camargo RL, Barella LF, Vanzela EC, de Freitas Mathias PC, Carneiro EM, Boschero AC, Bonfleur ML (2013) Impaired muscarinic type 3 (M3) receptor/PKC and PKA pathways in islets from MSG-obese rats. Mol Biol Rep 40(7):4521–4528. doi:10.1007/s11033-013-2546-y
Duivenvoorden I, Teusink B, Rensen PC, Romijn JA, Havekes LM, Voshol PJ (2005) Apolipoprotein C3 deficiency results in diet-induced obesity and aggravated insulin resistance in mice. Diabetes 54(3):664–671
Bernardis LL, Patterson BD (1968) Correlation between ‘Lee index’ and carcass fat content in weanling and adult female rats with hypothalamic lesions. J Endocrinol 40(4):527–528
Carneiro EM, Latorraca MQ, Araujo E, Beltra M, Oliveras MJ, Navarro M, Berna G, Bedoya FJ, Velloso LA, Soria B, Martin F (2009) Taurine supplementation modulates glucose homeostasis and islet function. J Nutr Biochem 20(7):503–511. doi:10.1016/j.jnutbio.2008.05.008
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ribeiro RA, Santos-Silva JC, Vettorazzi JF, Cotrim BB, Mobiolli DD, Boschero AC, Carneiro EM (2012) Taurine supplementation prevents morpho-physiological alterations in high-fat diet mice pancreatic beta-cells. Amino Acids 43(4):1791–1801. doi:10.1007/s00726-012-1263-5
Henquin JC (2011) The dual control of insulin secretion by glucose involves triggering and amplifying pathways in beta-cells. Diabetes Res Clin Pract 93(Suppl 1):S27–S31. doi:10.1016/S0168-8227(11)70010-9
Iwase M, Ichikawa K, Tashiro K, Iino K, Shinohara N, Ibayashi S, Yoshinari M, Fujishima M (2000) Effects of monosodium glutamate-induced obesity in spontaneously hypertensive rats vs. Wistar Kyoto rats: serum leptin and blood flow to brown adipose tissue. Hypertens Res 23(5):503–510
Leguisamo NM, Lehnen AM, Machado UF, Okamoto MM, Markoski MM, Pinto GH, Schaan BD (2012) GLUT4 content decreases along with insulin resistance and high levels of inflammatory markers in rats with metabolic syndrome. Cardiovas Diabetol 11:100. doi:10.1186/1475-2840-11-100
Wang WF, Li SM, Ren GP, Zheng W, Lu YJ, Yu YH, Xu WJ, Li TH, Zhou LH, Liu Y, Li DS (2014) Recombinant murine fibroblast growth factor 21 ameliorates obesity-related inflammation in monosodium glutamate-induced obesity rats. Endocrine. doi:10.1007/s12020-014-0433-5
Andreazzi AE, Scomparin DX, Mesquita FP, Balbo SL, Gravena C, De Oliveira JC, Rinaldi W, Garcia RM, Grassiolli S, Mathias PC (2009) Swimming exercise at weaning improves glycemic control and inhibits the onset of monosodium L-glutamate-obesity in mice. J Endocrinol 201(3):351–359. doi:10.1677/JOE-08-0312
Roman-Ramos R, Almanza-Perez JC, Garcia-Macedo R, Blancas-Flores G, Fortis-Barrera A, Jasso EI, Garcia-Lorenzana M, Campos-Sepulveda AE, Cruz M, Alarcon-Aguilar FJ (2011) Monosodium glutamate neonatal intoxication associated with obesity in adult stage is characterized by chronic inflammation and increased mRNA expression of peroxisome proliferator-activated receptors in mice. Basic Clin Pharmacol Toxicol 108(6):406–413. doi:10.1111/j.1742-7843.2011.00671.x
Watanabe N, Kobayashi M, Maegawa H, Ishibashi O, Takata Y, Shigeta Y (1986) Long-term in vitro effects of insulin on insulin binding and glucose transport. Diabetes Res Clin Pract 2(1):1–8
Cigolini M, Zancanaro C, Benati D, Cavallo E, Bosello O, Smith U (1987) Metformin enhances insulin binding to “in vitro” down regulated human fat cells. Diabete Metab 13(1):20–22
Sullivan JE, Brocklehurst KJ, Marley AE, Carey F, Carling D, Beri RK (1994) Inhibition of lipolysis and lipogenesis in isolated rat adipocytes with AICAR, a cell-permeable activator of AMP-activated protein kinase. FEBS Lett 353(1):33–36
Treebak JT, Glund S, Deshmukh A, Klein DK, Long YC, Jensen TE, Jorgensen SB, Viollet B, Andersson L, Neumann D, Wallimann T, Richter EA, Chibalin AV, Zierath JR, Wojtaszewski JF (2006) AMPK-mediated AS160 phosphorylation in skeletal muscle is dependent on AMPK catalytic and regulatory subunits. Diabetes 55(7):2051–2058. doi:10.2337/db06-0175
Acknowledgments
This study was supported by Grants from Conselho Nacional para o Desenvolvimento Científico e Tecnológico (CNPq 442846/2014-2), Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (Faperj E-26/110.921/2013 and E-26/101.129/2014) and Fundação de Amparo à Pesquisa do Estado de São Paulo (Fapesp). We are grateful to Gilliana Neves and Josué Almeida from Biotério de Experimentação de Roedores for animal care and Nicola Conran for editing the English.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Rights and permissions
About this article
Cite this article
Araujo, T.R., Freitas, I.N., Vettorazzi, J.F. et al. Benefits of l-alanine or l-arginine supplementation against adiposity and glucose intolerance in monosodium glutamate-induced obesity. Eur J Nutr 56, 2069–2080 (2017). https://doi.org/10.1007/s00394-016-1245-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-016-1245-6