Abstract
Purpose
Oxidative DNA damage is one of the mechanisms associated to initial colorectal carcinogenesis, but how it interacts with β-catenin, an adherence protein related to cancer evolution, is not clear. This study investigates the relationship between oxidative DNA damage and β-catenin expression in normal mucosa and colon tumor tissue (adenoma and adenocarcinoma) in colorectal adenocarcinoma evolution.
Method
One hundred and 13 samples were studied. Hematoxylin–eosin determined histological grade. β-Catenin expression was analyzed by immunohistochemistry. The oxidative DNA damage was evaluated using comet assay technique. The coefficient for rejection of the nullity hypothesis was taken to 5 %. Kruskal–Wallis, Spearman test, and partial correlation were used to analyze the data.
Results
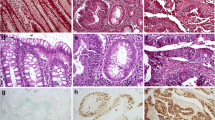
There was oxidative DNA damage increase in colorectal cancer evolution (p < 0.01). Histological grade was correlated with oxidative DNA damage (p < 0.01). There were differences in β-catenin expression among normal, adenoma, and adenocarcinoma tissue with progressive increase of β-catenin expression (p < 0.00). Histological grade was correlated to β-catenin expression (p < 0.00). There was a relationship (p < 0.00) between β-catenin and histological grade while controlling for the effect of oxidative DNA damage.
Conclusion
The findings of this study make it possible to establish a relationship between oxidative DNA damage and β-catenin expression in normal mucosa and colorectal tumor tissue. Additionally, they show a causal relationship between variations of β-catenin in different tissues analyzed while controlling for the effect of oxidative DNA damage.





Similar content being viewed by others
References
Muto T, Bussey HJ, Morson BC (1975) The evolution of cancer of the colon and rectum. Cancer 36(6):2251–2270
Fearon ER, Vogelstein B (1990) A genetic model for colorectal tumorigenesis. Cell 61(5):759–767
Martinez NP, Kanno DT, Aires J, Cardinalli IA, Priolli DG (2010–2011) β-Catenin and e-cadherin tissue content as a prognostic marker in left-side colorectal cancer. Cancer Biomark 8(3):129–135. doi:10.3233/DMA-2011-0843
Elzagheid A, Buhmeida A, Korkeila E, Collan Y, Syrjänen K, Pyrhönen S (2008) Nuclear β-catenin expression as a prognostic factor in advanced colorectal carcinoma. World J Gastroenterol 14(24):3866–3871. doi:10.3748/wjg.14.3866
Wanitsuwan W, Kanngurn S, Boonpipattanapong T, Sangthong R, Sangkhathat S (2008) Overall expression of betacatenin outperforms its nuclear accumulation in predicting outcomes of colorectal cancers. World J Gastroenterol 14(39):6052–6059. doi:10.3748/wjg.14.6052
Essers MA, de Vries-Smits LM, Barker N, Polderman PE, Burgering BM, Korswagen HC (2005) Functional interaction between β-catenin and FOXO in oxidative stress signaling. Science 308(5725):1181–1184. doi:10.1126/science.1109083
Bright-Thomas RM, Hargest R (2003) APC, β-catenin and hTCF-4; an unholy trinity in the genesis of colorectal cancer. Eur J Surg Oncol 29(2):107–117
Garcea G, Sharma RA, Dennison A, Steward WP, Gescher A, Berry DP (2003) Molecular biomarkers of colorectal carcinogenesis and their role in surveillance and early intervention. Eur J Cancer 39(8):1041–1052. doi:10.1016/S0959-8049(03)00027-3
Glei M, Latunde-Dada GO, Klinder A, Becker TW, Hermann U, Voigt K, Pool-Zobel BL (2002) Iron-overload induces oxidative DNA damage in the human colon carcinoma cell line HT29 clone 19A. Mutat Res 519:151–161. doi:10.1016/S1383-5718(02)00135-3
Ladeira MSP, Rodríguez MAM, Salvadori DMF, Queiroz DMM, Freire-Maia DV (2004) DNA damage in patients infected by Helicobacter pylori. Cancer Epidemiol Biomarkers Prev 13:631–637
ItzKowitz SH, Yio X (2004) Inflammation and cancer. IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol 287:G7–G17. doi:ajpgi.00079. 2004/ajpgi.00079.2004
Ribeiro ML, Priolli DG, Miranda DD, Arçari DP, Pedrazzoli J Jr, Martinez CA (2008) Analysis of oxidative DNA damage in patients with colorectal cancer. Clin Colorectal Cancer 7(4):267–272. doi:10.3816/CCC.2008.n.034
Balasubramanyam M, Adaikalakoteswari A, Sameermahmood Z, Mohan V (2010) Biomarkers of oxidative stress: methods and measures of oxidative DNA damage (COMET assay) and telomere shortening. Methods Mol Biol 610:245–261. doi:10.1007/978-1-60327-029-8_15
Oliveira R, Johansson B (2012) Quantitative DNA damage and repair measurement with the yeast comet assay. Methods Mol Biol 920:101–109. doi:10.1007/978-1-61779-998-3_8
Schlemper RJ, Riddell RH, Kato Y, Borchard F, Cooper HS, Dawsey SM, Dixon MF, Fenoglio-Preiser CM, Fléjou JF, Geboes K, Hattori T, Hirota T, Itabashi M, Iwafuchi M, Iwashita A, Kim YI, Kirchner T, Klimpfinger M, Koike M, Lauwers GY, Lewin KJ, Oberhuber G, Offner F, Price AB, Rubio CA, Shimizu M, Shimoda T, Sipponen P, Solcia E, Stolte M, Watanabe H, Yamabe H (2000) The Vienna Classification of gastrointestinal epithelial neoplasia. Gut 47(2):251–255. doi:10.1136/gut.47.2.251
Willis J, Riddell RH (2003) Biology versus terminology: east meets west in surgical pathology. Gastrointest Endosc 57(3):369–376. doi:10.1067/mge.2003.106
Priolli DG, Martinez CA, Piovesan H, Cardinalli IA, Margarido NF, Waisberg J (2010) Morphofunctional malignancy grading is a valuable prognostic factor for colorectal cancer. Arq Gastroenterol 47(3):225–232. doi:10.1590/S0004-28032010000300003
Redon CE, Dickey JS, Nakamura AJ, Kareva IG, Naf D, Nowsheen S, Kryston TB, Bonner WM, Georgakilas AG, Sedelnikova OA (2010) Tumors induce complex DNA damage in distant proliferative tissues in vivo. Proc Natl Acad Sci USA 107(42):17992–17997. doi:10.1073/pnas.1008260107
Halliwell B, Whiteman M (2004) Measuring reactive species and oxidative damage in vivo and in cell culture: how should you do it and what do the results mean? Br J Pharmacol 142(2):231–255. doi:10.1038/sj.bjp.0705776
Pool-Zobel B, Leucht U (1997) Induction of DNA damage in human colon cells derived from biopsies by suggested risk factors of colon cancer. Mutat Res 375:105–116. doi:10.1016/S0027-5107(97)00006-7
Gedik CM, Boyle SP, Wood SG, Vaughan NJ, Collins AR (2002) Oxidative stress in humans: validation of biomarkers of DNA damage. Carcinogenesis 23(9):1441–1446. doi:10.1093/carcin/23.9.1441
Ames BN, Shigenaga MK, Hagen TM (1993) Oxidants, antioxidants, and degenerative diseases of aging. Proc Natl Acad Sci USA 90:7915–7922
Goodman M, Bostick RM, Dash C, Terry P, Flanders WD, Mandel J (2008) A summary measure of pro- and anti-oxidant exposures and risk of incident, sporadic, colorectal adenomas. Cancer Causes Control 19:1051–1064. doi:10.1007/s10552-008-9169-y
Leung EY, Crozier JE, Talwar D, O’Reilly DS, McKee RF, Horgan PG, McMillan DC (2008) Vitamin antioxidants, lipid peroxidation, tumour stage, the systemic inflammatory response and survival in patients with colorectal cancer. Int J Cancer 123:2460–2464. doi:10.1002/ijc.23811
Pool-Zobel BL, Abrahamse SL, Collins AR, Kark W, Gugler R, Oberreuther D, Siegel EG, Treptow-van Lishaut S, Rechkemmer G (1999) Analysis of DNA strand breaks, oxidized bases, and glutathione S-tranferase P1 in human colon cells from biopsies. Cancer Epidemiol Biomarker Prev 8:609–614
Obtulowicz T, Swoboda M, Speina E, Gackowski D, Rozalski R, Siomek A, Janik J, Janowska B, Ciesla JM, Jawien A, Banaszkiewicz Z, Guz J, Dziaman T, Szpila A, Olinski R, Tudek B (2010) Oxidative stress and 8-oxoguanine repair are enhanced in colon adenoma and carcinoma patients. Mutagenesis 25(5):463–471. doi:10.1093/mutage/geq028
Oliva MR, Ripoll F, Muñiz P, Iradi A, Trullenque R, Valls V, Drehmer E, Sáez GT (1997) Genetic alterations and oxidative metabolism in sporadic colorectal tumors from a Spanish community. Mol Carcinog 18(4):232–243. doi:10.1002/(SICI)1098-2744(199704)18:4<232::AID-MC7>3.0.CO;2-F
Chen S, Liu J, Li G, Mo F, Xu X, Zhang T, Li X, Han X, Sun Y (2008) Altered distribution of betacatenin and prognostic roles in colorectal carcinogenesis. Scand J Gastroenterol 43(4):456–464. doi:10.1080/00365520701785194
Akiyama T (2000) Wnt/β-catenin signaling. Cytokine Growth Factor Rev 11:273–282. doi:10.1016/S1359-6101(00)00011-3
Smith JJ, Deane NG, Dhawan P, Beauchamp RD (2008) Regulation of metastasis in colorectal adenocarcinoma: a collision between development and tumor biology. Surgery 144(3):353–366. doi:10.1016/j.surg.2008.05.007
Marchenko ND, Marchenko GN, Weinreb RN, Lindsey JD, Kyshtoobayeva A, Crawford HC, Strongin AY (2004) β-Catenin regulates the gene of MMP-26, a novel matrix metalloproteinase expressed both in carcinomas and normal epithelial cells. Int J Biochem Cell Biol 36:942–956. doi:10.1016/j.biocel.2003.12.007
Oloumi A, McPhee T, Dedhar S (2004) Regulation of E-cadherin expression and β-catenin/Tcf transcriptional activity by the integrin-linked kinase. Biochim Biophys Acta 1691(1):1–15. doi:10.1016/j.bbamcr.2003.12.002
van der Wurff AA, Vermeulen SJ, van der Linden EP, Mareel MM, Bosman FT, Arends JW (1997) Patterns of alpha- and β-catenin and E-cadherin expression in colorectal adenomas and carcinomas. J Pathol 182(3):325–330. doi:10.1002/(SICI)1096-9896(199707)182:3<325::AID-PATH865>3.0.CO;2-Y
Debruyne P, Vermeulen S, Mareel M (1999) The role of the E-cadherin/catenin complex in gastrointestinal cancer. Acta Gastroenterol Belg 62(4):393–402
Acknowledgments
The authors acknowledge FAPESP—São Paulo Research Foundation, process no. 2007/01196-5.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was performed within the Postgraduate Program Strictu Senso in Health Science, Sao Francisco University Medical School, Sao Paulo, Brazil.
Rights and permissions
About this article
Cite this article
Priolli, D.G., Canelloi, T.P., Lopes, C.O. et al. Oxidative DNA damage and β-catenin expression in colorectal cancer evolution. Int J Colorectal Dis 28, 713–722 (2013). https://doi.org/10.1007/s00384-013-1688-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-013-1688-7