Abstract
Background
It has been shown that pulmonary retinol level is decreased during lung morphogenesis in the nitrofen-induced PH in congenital diaphragmatic hernia (CDH). Placenta has a major role in the retinol homeostasis in fetal life. Since there is no fetal retinol synthesis, maternal retinol has to cross the placenta. Placenta is the main fetal retinol store where retinol is stored in retinyl-ester formation. Trophoblasts have to produce its own retinol-binding protein (RBP) for retinol transport from placenta to fetus. Recently, we demonstrated that trophoblastic RBP expression is decreased in the nitrofen model of CDH. The aim of this study was to investigate the retinol transfer from mother to the placenta in nitrofen model of CDH.
Methods
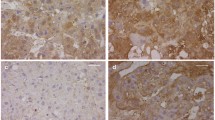
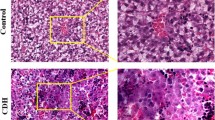
Pregnant rats were exposed to either olive oil or nitrofen on day 9 of gestation (D9). Fetal placenta harvested on D21 and divided into two groups: control (n = 11) and nitrofen with CDH (n = 11). Retinoid levels in placenta were measured using HPLC. Immunohistochemistry was performed to evaluate trophoblastic expression of main RSP genes.
Results
Total retinol levels in the placenta were significantly increased in CDH placenta compared to control placenta. The retinyl-ester levels were significantly increased in CDH placenta compared to control placenta. Markedly, decreased immunoreactivity of retinoid signaling pathway was observed in trophoblast cells in CDH compared to control placenta.
Conclusions
Increased placental retinol levels show that retinol is transferred from mother to placenta and stored in the placenta in nitrofen model of CDH during lung morphogenesis. Nitrofen may disturb the mobilization of retinol from placenta to fetal circulation causing PH in CDH.

Similar content being viewed by others
References
Colvin J, Bower C, Dickinson JE, Sokol J (2005) Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics 116(3):e356–e363
Stege G, Fenton A, Jaffray B (2003) Nihilism in the 1990s: the true mortality of congenital diaphragmatic hernia. Pediatrics 112(3 Pt 1):532–535
Gosche JR, Islam S, Boulanger SC (2005) Congenital diaphragmatic hernia: searching for answers. Am J Surg 190(2):324–332
Robinson PD, Fitzgerald DA (2007) Congenital diaphragmatic hernia. Paediatr Respir Rev 8(4):323–334 (quiz 334–325)
Montedonico S, Nakazawa N, Puri P (2008) Congenital diaphragmatic hernia and retinoids: searching for an etiology. Pediatr Surg Int 24(7):755–761
Noble BR, Babiuk RP, Clugston RD, Underhill TM, Sun H, Kawaguchi R, Walfish PG, Blomhoff R, Gundersen TE, Greer JJ (2007) Mechanisms of action of the congenital diaphragmatic hernia-inducing teratogen nitrofen. Am J Physiol Lung Cell Mol Physiol 293(4):L1079–L1087
Clagett-Dame M, DeLuca HF (2002) The role of vitamin A in mammalian reproduction and embryonic development. Annu Rev Nutr 22:347–381
Wassef L, Quadro L (2011) Uptake of dietary retinoids at the maternal-fetal barrier: in vivo evidence for the role of lipoprotein lipase and alternative pathways. J Biol Chem 286(37):32198–32207
Spiegler E, Kim YK, Wassef L, Shete V, Quadro L (2012) Maternal-fetal transfer and metabolism of vitamin A and its precursor beta-carotene in the developing tissues. Biochim Biophys Acta 1821(1):88–98
Sapin V, Chaib S, Blanchon L, Alexandre-Gouabau MC, Lemery D, Charbonne F, Gallot D, Jacquetin B, Dastugue B, Azais-Braesco V (2000) Esterification of vitamin A by the human placenta involves villous mesenchymal fibroblasts. Pediatr Res 48(4):565–572
Monaco HL, Rizzi M, Coda A (1995) Structure of a complex of two plasma proteins: transthyretin and retinol-binding protein. Science 268(5213):1039–1041
Quadro L, Hamberger L, Colantuoni V, Gottesman ME, Blaner WS (2003) Understanding the physiological role of retinol-binding protein in vitamin A metabolism using transgenic and knockout mouse models. Mol Asp Med 24(6):421–430
Quadro L, Hamberger L, Gottesman ME, Colantuoni V, Ramakrishnan R, Blaner WS (2004) Transplacental delivery of retinoid: the role of retinol-binding protein and lipoprotein retinyl ester. Am J Physiol Endocrinol Metab 286(5):E844–E851
Morriss-Kay GM, Ward SJ (1999) Retinoids and mammalian development. Int Rev Cytol 188:73–131
Sapin V, Ward SJ, Bronner S, Chambon P, Dolle P (1997) Differential expression of transcripts encoding retinoid binding proteins and retinoic acid receptors during placentation of the mouse. Dev Dyn 208(2):199–210
Kam RK, Deng Y, Chen Y, Zhao H (2012) Retinoic acid synthesis and functions in early embryonic development. Cell Biosci 2(1):11
Nakazawa N, Montedonico S, Takayasu H, Paradisi F, Puri P (2007) Disturbance of retinol transportation causes nitrofen-induced hypoplastic lung. J Pediatr Surg 42(2):345–349
Paik J, Vogel S, Quadro L, Piantedosi R, Gottesman M, Lai K, Hamberger L, Vieira Mde M, Vieira Mde M, Blaner WS (2004) Vitamin A: overlapping delivery pathways to tissues from the circulation. J Nutr 134(1):276S–280S
Kato Y, Braunstein GD (1991) Retinoic acid stimulates placental hormone secretion by choriocarcinoma cell lines in vitro. Endocrinology 128(1):401–407
Nakazawa N, Takayasu H, Montedonico S, Puri P (2007) Altered regulation of retinoic acid synthesis in nitrofen-induced hypoplastic lung. Pediatr Surg Int 23(5):391–396
O’Byrne SM, Wongsiriroj N, Libien J, Vogel S, Goldberg IJ, Baehr W, Palczewski K, Blaner WS (2005) Retinoid absorption and storage is impaired in mice lacking lecithin:retinol acyltransferase (LRAT). J Biol Chem 280(42):35647–35657
Gallot D, Marceau G, Coste K, Hadden H, Robert-Gnansia E, Laurichesse H, Dechelotte PJ, Labbe A, Dastugue B, Lemery D et al (2005) Congenital diaphragmatic hernia: a retinoid-signaling pathway disruption during lung development? Birth Defects Res A Clin Mol Teratol 73(8):523–531
Beurskens LW, Tibboel D, Lindemans J, Duvekot JJ, Cohen-Overbeek TE, Veenma DC, de Klein A, Greer JJ, Steegers-Theunissen RP (2010) Retinol status of newborn infants is associated with congenital diaphragmatic hernia. Pediatrics 126(4):712–720
Kutasy B, Gosemann JH, Doi T, Fujiwara N, Friedmacher F, Puri P (2012) Nitrofen interferes with trophoblastic expression of retinol-binding protein and transthyretin during lung morphogenesis in the nitrofen-induced congenital diaphragmatic hernia model. Pediatr Surg Int 28(2):143–148
Hosotani K, Kitagawa M (2003) Improved simultaneous determination method of beta-carotene and retinol with saponification in human serum and rat liver. J Chromatogr B Anal Technol Biomed Life Sci 791(1–2):305–313
Moulas ANZI, Taitzoglou IA, Tsantarliotou MP, Botsoglou NA (2003) Simultaneous determination of retinoic acid, retinol, and retinyl palmitate in ram plasma by liquid chromatography. J Liq Chromatogr 26:559–572
Kutasy B, Gosemann JH, Duess JW, Puri P (2013) Increased trophoblastic apoptosis mediated by neutrophil gelatinase-associated lipocalin (NGAL) activation in the nitrofen model of congenital diaphragmatic hernia. Pediatr Surg Int 29(1):25–31
Furukawa S, Hayashi S, Usuda K, Abe M, Hagio S, Ogawa I (2011) Toxicological pathology in the rat placenta. J Toxicol Pathol 24(2):95–111
Quadro L, Hamberger L, Gottesman ME, Wang F, Colantuoni V, Blaner WS, Mendelsohn CL (2005) Pathways of vitamin A delivery to the embryo: insights from a new tunable model of embryonic vitamin A deficiency. Endocrinology 146(10):4479–4490
Ismadi SD, Olson JA (1982) Dynamics of the fetal distribution and transfer of vitamin A between rat fetuses and their mother. Int J Vitam Nutr Res 52(2):112–119
Kim YK, Wassef L, Hamberger L, Piantedosi R, Palczewski K, Blaner WS, Quadro L (2008) Retinyl ester formation by lecithin: retinol acyltransferase is a key regulator of retinoid homeostasis in mouse embryogenesis. J Biol Chem 283(9):5611–5621
Wendling O, Chambon P, Mark M (1999) Retinoid X receptors are essential for early mouse development and placentogenesis. Proc Natl Acad Sci USA 96(2):547–551
Guilbert TW, Gebb SA, Shannon JM (2000) Lung hypoplasia in the nitrofen model of congenital diaphragmatic hernia occurs early in development. Am J Physiol Lung Cell Mol Physiol 279(6):L1159–L1171
Jay PY, Bielinska M, Erlich JM, Mannisto S, Pu WT, Heikinheimo M, Wilson DB (2007) Impaired mesenchymal cell function in Gata4 mutant mice leads to diaphragmatic hernias and primary lung defects. Dev Biol 301(2):602–614
Doi T, Hajduk P, Puri P (2009) Upregulation of Slit-2 and Slit-3 gene expressions in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44(11):2092–2095
Doi T, Puri P (2009) Up-regulation of Wnt5a gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44(12):2302–2306
Ruttenstock E, Doi T, Dingemann J, Puri P (2011) Prenatal administration of retinoic acid upregulates insulin-like growth factor receptors in the nitrofen-induced hypoplastic lung. Birth Defects Res B Dev Reprod Toxicol 92(2):148–151
Kling DE, Cavicchio AJ, Sollinger CA, Schnitzer JJ, Kinane TB, Newburg DS (2010) Nitrofen induces apoptosis independently of retinaldehyde dehydrogenase (RALDH) inhibition. Birth Defects Res B Dev Reprod Toxicol 89(3):223–232
Manson JM (1986) Mechanism of nitrofen teratogenesis. Environ Health Perspect 70:137–147
Brown TJ, Manson JM (1986) Further characterization of the distribution and metabolism of nitrofen in the pregnant rat. Teratology 34(2):129–139
Clugston RD, Zhang W, Greer JJ (2010) Early development of the primordial mammalian diaphragm and cellular mechanisms of nitrofen-induced congenital diaphragmatic hernia. Birth Defects Res A Clin Mol Teratol 88(1):15–24
Kutasy B, Friedmacher F, Duess JW, Puri P (2014) Prenatal administration of retinoic acid increases the trophoblastic insulin-like growth factor 2 protein expression in the nitrofen model of congenital diaphragmatic hernia. Pediatr Surg Int 30(2):137–142
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kutasy, B., Pes, L., Friedmacher, F. et al. Nitrofen increases total retinol levels in placenta during lung morphogenesis in the nitrofen model of congenital diaphragmatic hernia. Pediatr Surg Int 30, 1017–1022 (2014). https://doi.org/10.1007/s00383-014-3525-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-014-3525-7