Abstract
Introduction
After an endoscopic third ventriculostomy (ETV) fails, it is unclear how well subsequent treatment fares, especially in comparison to shunts inserted as primary treatment. In this study, we present a further analysis of the infants enrolled a prospective multicentre study who failed ETV and describe the outcome of their subsequent treatment, comparing this to those who received shunt as their primary treatment.
Methods
This was a post hoc analysis of data from the International Infant Hydrocephalus Study (IIHS)—a prospective, multicentre study of infants with hydrocephalus from aqueductal stenosis who received either an ETV or shunt. In the current analysis, we compared the results of the 38 infants who failed ETV and the 43 infants who received primary shunt. Patients were followed prospectively for time to treatment failure, defined as the need for repeat CSF diversion procedure (shunt or ETV) or death due to hydrocephalus.
Results
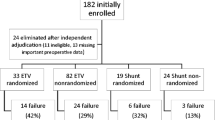
There were a total of 81 patients: 43 primary shunts, 34 shunt post-ETV, and 4 repeat ETV. The median time between the primary ETV and the second intervention was 29 days (IQR 14–69), with no significant difference between repeat ETV and shunt post-ETV. Median length of available follow-up was 800 days (IQR 266–1651), during which time, failure was noted in 3 (75.0%) repeat ETV patients, 10 (29.4%) shunt post-ETV patients, and 9 (20.9%) primary shunt patients. In an adjusted Cox regression model, the risk of failure was higher for repeat ETV compared to primary shunt, but there was no significant difference between primary shunt and shunt post-ETV. No other variable showed statistical significance.
Conclusions
In our prospective study of infants with aqueductal stenosis, there was no significant difference in failure outcome of shunts inserted after a failed ETV and primary shunts. Therefore, our data do not support the notion that previous ETV confers either a protective or negative effect on subsequently-placed shunts. Larger studies, in a wider ranging population, are required to establish how widely these data apply.
Trial registration
NCT00652470

Similar content being viewed by others
References
Kulkarni AV, Sgouros S, Constantini S (2016) International infant hydrocephalus study: initial results of a prospective, multicenter comparison of endoscopic third ventriculostomy (ETV) and shunt for infant hydrocephalus. Childs Nerv Syst 32:1039–1048. doi:10.1007/s00381-016-3095-1
Moreira I, Pereira J, Oliveira J et al (2016) Endoscopic re-opening of third ventriculostomy: case series and review of literature. Clin Neurol Neurosurg 145:58–63. doi:10.1016/j.clineuro.2016.04.007
Siomin V, Weiner H, Wisoff J et al (2001) Repeat endoscopic third ventriculostomy: is it worth trying? Childs Nerv Syst 17:551–555. doi:10.1007/s003810100475
Surash S, Chumas P, Bhargava D et al (2010) A retrospective analysis of revision endoscopic third ventriculostomy. Childs Nerv Syst 26:1693–1698. doi:10.1007/s00381-010-1176-0
Marano PJ, Stone SSD, Mugamba J et al (2015) Reopening of an obstructed third ventriculostomy: long-term success and factors affecting outcome in 215 infants. J Neurosurg Pediatr 15:399–405. doi:10.3171/2014.10.PEDS14250
Warf BC, Bhai S, Kulkarni AV, Mugamba J (2012) Shunt survival after failed endoscopic treatment of hydrocephalus. J Neurosurg Pediatr 10:463–470. doi:10.3171/2012.9.PEDS1236
Sgouros S, Kulkharni AV, Constantini S (2006) The international infant hydrocephalus study: concept and rational. Childs Nerv Syst 22:338–345. doi:10.1007/s00381-005-1253-y
Olschewski M, Schumacher M, Davis KB (1992) Analysis of randomized and nonrandomized patients in clinical trials using the comprehensive cohort follow-up study design. Control Clin Trials 13:226–239
Shim K-W, Kim D-S, Choi J-U (2008) Simultaneous endoscopic third ventriculostomy and ventriculoperitoneal shunt for infantile hydrocephalus. Childs Nerv Syst 24:443–451. doi:10.1007/s00381-007-0526-z
Drake J, Chumas P, Kestle J et al (2006) Late rapid deterioration after endoscopic third ventriculostomy: additional cases and review of the literature. J Neurosurg 105:118–126. doi:10.3171/ped.2006.105.2.118
Kahle KT, Kulkarni AV, Limbrick DD et al (2016) Hydrocephalus in children. Lancet 387:788–799. doi:10.1016/S0140-6736(15)60694-8
Acknowledgements
The authors would like to extend a special thanks to Adina Sherer, who ran the organizational logistics of this study and without whom the IIHS would not have been possible.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest with respect to this work.
Additional information
See Appendix for full list of study investigators
Appendix: IIHS Personnel
Appendix: IIHS Personnel
Steering Committee: Shlomi Constantini (Principal Investigator), Spyros Sgouros, Abhaya V. Kulkarni
Consultant Neurologist: Yael Leitner
Data Safety Monitoring Committee: John RW Kestle (Chair), Douglas D Cochrane, Maurice Choux, Fleming Gjerris
Coordinating Administrator: Adina Sherer
Participating Investigators (in parentheses are the number of eligible patients contributed to the study by each investigator):
Medical Center | IIHS Participants | # of Patients |
Ankara, Turkey Hacettepe University Hospital | Nejat Akalan, Burçak Bilginer | (12) |
Barcelona, Spain Hospital Sant Joan de Deu | Ramon Navarro (currently at Cleveland Clinic Abu Dhabi, UAE) | (7) |
Belgrade, Serbia | Ljiljana Vujotic | (8) |
Clinical Center of Serbia, Belgrade, Neurosurgery Division | ||
Berlin, Germany Charité - Universitätsmedizin Berlin | Hannes Haberl, Ulrich-Wilhelm Thomale | (4) |
Birmingham, UK Birminghan Children’s Hospital | Spyros Sgouros (currently at "Mitera" Childrens Hospital) | (1) |
Buenos Aires, Argentina | Graciela Zúccaro, Roberto Jaimovitch | (21) |
Hospital De Pediatria Prof. Dr. J.P. Garrahan | ||
Chicago, USA | David Frim, Lori Loftis | (3) |
The University of Chicago Comer Children’s Hospital | ||
Dallas, USA Children's Medical Center of Dallas | Dale M. Swift, Brian Robertson, Lynn Gargan | (6) |
Debrecen, Hungary | László Bognár, László Novák, Georgina Cseke | (5) |
University of Debrecen, Clinical Center, Department of Neurosurgery | ||
Genova, Italy | Armando Cama, Giuseppe Marcello Ravegnani | (3) |
Giannina Gaslini Hospital, Gaslini Children Institute | ||
Giessen/Leipzig University Hospital Gießen and Marburg | Matthias Preuß Currently at University Hospital Leipzig | (4) |
Greifswald, Germany | Henry W. Schroeder, Michael Fritsch, Joerg Baldauf | (2) |
Ernst-Moritz-Arndt-Universität Klinik für Neurochirurgie | ||
Katowice, Poland Medical University of Silesia | Marek Mandera, Jerzy Luszawski, Patrycja Skorupka | (9) |
Liverpool, UK Alder Hey Children’s Hospital | Conor Mallucci, Dawn Williams | (4) |
Lodz, Poland | Krzysztof Zakrzewski, Emilia Nowoslawska | (2) |
Polish Mother’s Memorial Hospital, Research Institute | ||
Lucknow (KGMC), India CSM Medical University (KGMC) | Chhitij Srivastava | (4) |
Lucknow (SGPGI), India | Ashok K. Mahapatra, Raj Kumar, Rabi Narayan Sahu | (8) |
Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGI) | ||
Moscow, Russia Burdenko Neurosurgical Institute | Armen G. Melikian (Армен Меликян), Anton Korshunov (Антон Евгеньевич Коршунов), Anna Galstyan (Анна Галстян) | (11) |
New Delhi, India All India Institute of Medical Sciences | Ashish Suri, Deepak Gupta | (12) |
Nijmegen, The Netherlands Radboud University Medical Center | J. André Grotenhuis, Erik J. van Lindert | (9) |
Nova Lima, Brazil | José Aloysio da Costa Val | (5) |
Neurocirurgia Infantil, Biocor Instituto | ||
Rome, Italy | Concezio Di Rocco, Gianpiero Tamburrini | (4) |
Pediatric Neurosurgery, Policlinico Universitario “A. Gemelli” | ||
São Paulo, Brazil Escola Paulista de Medicina, UNIFESP | Samuel Tau Zymberg, Sergio Cavalheiro | (3) |
Shanghai, China | Ma Jie, Jiang Feng | (3) |
Xinhua Hospital, Shanghai JiaoTong University School of Medicine | ||
Tel Aviv, Israel | Shlomi Constantini, Orna Friedman | (20) |
Dana Children’s Hospital, Tel Aviv Medical Center | ||
Toronto, Canada Hospital for Sick Children | Abhaya V. Kulkarni | (5) |
Warsaw, Poland | Marcin Roszkowski, Slawomir Barszcz | (7) |
Children’s Memorial Health Institute | ||
The following centres (and investigators) participated in the IIHS, but did not enroll any patients: Baltimore, Maryland, USA (George Jallo); Gainesville, Florida, USA (David W. Pincus, Bridget Richter); Kiel, Germany (HM Mehdorn, Susan Schultka); London, Ontario, Canada (Sandrine de Ribaupierre); London, UK (Dominic Thompson, Silvia Gatscher); Mainz, Germany (Wolfgang Wagner, Dorothee Koch); Reggio Calabria, Italy (Saverio Cipri, Claudio Zaccone); Winnipeg, Manitoba, Canada (Patrick McDonald).
Rights and permissions
About this article
Cite this article
Kulkarni, A.V., Sgouros, S., Constantini, S. et al. Outcome of treatment after failed endoscopic third ventriculostomy (ETV) in infants with aqueductal stenosis: results from the International Infant Hydrocephalus Study (IIHS). Childs Nerv Syst 33, 747–752 (2017). https://doi.org/10.1007/s00381-017-3382-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-017-3382-5