Abstract
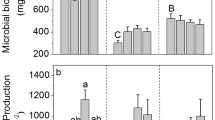
Quantifying soil P fractions is essential for understanding soil P cycling because these fractions are potential sources of bioavailable P. Here, we investigated whether soil P fractions (i.e., labile inorganic P [Pi], intermediately available Pi, organic P, occluded P, and apatite P) differed among three tropical forests at different stages of succession (early, middle, and late) in the Dinghushan Biosphere Reserve (DBR), Southern China. We also determined which soil P fractions was closely related to soil microbial and chemical properties in these forests. Soil microbial biomass and chemical properties (except pH, exchangeable Ca, and Mn) were higher in the late successional forest than those in the other two forests. We found that soil organic and occluded P were the dominant fraction in all three forests and together accounted for 78.9, 84.2, and 78.6 % of total P in the early, middle, and late successional forests, respectively. Soil P fractions and acid phosphomonoesterase activity differed significantly among forest successional stages; intermediately available Pi and organic P (potential sources of labile Pi) were highest in the late successional forest, occluded P was highest in the middle successional forest, and acid phosphomonoesterase activity significantly increased in middle and late successional forest. Soil labile Pi fraction and its potential sources (i.e., intermediately available Pi and soil organic P) were both positively correlated with soil microbial and chemical properties, suggesting that these properties may play critical roles in maintaining high levels of available or potentially available P fractions during this forest succession. Overall, our study indicates that succession in tropical forest ecosystems may not always lead to lower soil P availability.




Similar content being viewed by others
References
Acosta-Martínez V, Tabatabai MA (2011) Phosphorus cycle enzymes. In: Dick RP (ed) Methods of soil enzymology. Soil Science Society of America, Madison, WI, USA, pp 161–184
Bossio DA, Scow KM (1998) Impacts of carbon and flooding on soil microbial communities: phospholipid fatty acid profiles and substrate utilization patterns. Microb Ecol 35:265–278. doi:10.1007/s002489900082
Brown S, Lenart MT, Mo JM, Kong GH (1995) Structure and organic matter dynamics of a human-impacted pine forest in a MAB reserve of subtropical China. Biotropica 27:276–289
Bunemann EK, Prusisz B, Ehlers K (2011) Characterization of phosphorus forms in soil microorganisms. In: Bünemann EK, Frossard E, Oberson A (eds) Phosphorus in action: biological processes in soil phosphorus cycling, soil biology 26. Springer, Berlin, Germany, pp 37–58
Celi L, Cerli C, Turner BL, Santoni S, Bonifacio E (2013) Biogeochemical cycling of soil phosphorus during natural revegetation of Pinus sylvestris on disused sand quarries in Northwestern Russia. Plant Soil 367:121–134. doi:10.1007/s11104-013-1627-y
Chapin FS III, Barsdate RJ, Barèl D (1978) Phosphorus cycling in Alaskan coastal tundra: a hypothesis for the regulation of nutrient cycling. Oikos 31:189–199
Chen CR, Condron LM, Sinaj S, Davis MR, Sherlock RR, Frossard E (2003) Effects of plant species on phosphorus availability in a range of grassland soils. Plant Soil 256:115–130. doi:10.1023/A:1026273529177
Chen CR, Condron LM, Davis MR, Sherlock RR (2004) Effects of plant species on microbial biomass phosphorus and phosphatase activity in a range of grassland soils. Biol Fertil Soils 40:313–322. doi:10.1007/s00374-004-0781-z
Crews T, Kitayama K, Fownes J, Riley R, Herbert D, Mueller-Dombois D, Vitousek PM (1995) Changes in soil phosphorus fractions and ecosystem dynamics across a long chronosequence in Hawaii. Ecology 76:1407–1424. doi:10.2307/1938144
Cross AF, Schlesinger WH (1995) A literature review and evaluation of the Hedley fractionation: applications to the biogeochemical cycle of soil phosphorus in natural ecosystems. Geoderma 64:197–214. doi:10.1016/0016-7061(94)00023-4
Cross AF, Schlesinger WH (2001) Biological and geochemical controls on phosphorus fractions in semiarid soils. Biogeochemistry 52:155–172. doi:10.1023/A:1006437504494
Cusack DF, Silver WL, Torn MS, Burton SD, Firestone MK (2011) Changes in microbial community characteristics and soil organic matter with nitrogen additions in two tropical forests. Ecology 92:621–632. doi:10.1890/10-0459.1
Eger A, Almond PC, Condron LM (2011) Pedogenesis, soil mass balance, phosphorus dynamics and vegetation communities across a Holocene soil chronosequence in a super-humid climate, South Westland, New Zealand. Geoderma 163:185–196. doi:10.1016/j.geoderma.2011.04.007
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine, and terrestrial ecosystems. Ecol Lett 10:1135–1142. doi:10.1111/j.1461-0248.2007.01113.x
FAO (2010) Global forest resources assessment main report. Food and Agriculture Organisation of the United Nations, Rome
Fanin N, Fromin N, Buatois B, Hättenschwiler S (2013) An experimental test of the hypothesis of non-homeostatic consumer stoichiometry in a plant litter-microbe system. Ecol Lett 16:764–772. doi:10.1111/ele.12108
Fox TR, Miller BW, Rubilar R, Stape JL, Albaugh TJ (2011) Phosphorus nutrition of forest plantations: the role of inorganic and organic phosphorus. In: Bünemann EK, Frossard E, Oberson A (eds) Phosphorus in action: biological processes in soil phosphorus cycling, soil biology 26. Springer, Berlin, Germany, pp 317–338
Frizano J, Johnson AH, Vann DR, Scatena FN (2002) Soil phosphorus fractionation during forest development on landslide scars in the Luquillo mountains, Puerto Rico. Biotropica 34:17–26. doi:10.1111/j.1744-7429.2002.tb00238.x
Frossard E, Brossard M, Hedley MJ, Metherell A (1995) Reactions controlling the cycling of P in soils. In: Tiessen H (ed) Phosphorus in the global environment: transfers. Cycles and Management. Wiley, Chichester, UK, pp 107–137
Frostegård A, Bååth E (1996) The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol Fertil Soils 22:59–65. doi:10.1007/BF00384433
Garcia-Monteil DC, Neill C, Melillo J, Thomas S, Steudler PA, Cerri CC (2000) Soil phosphorus transformations following forest clearing for pasture in the Brazilian Amazon. Soil Sci Soc Am J 64:1792–1804. doi:10.2136/sssaj2000.6451792x
Giaveno C, Celi L, Aveiro Cessa RM, Prati M, Bonifacio E, Barberis E (2008) Interaction of organic phosphorus with clays extracted from Oxisols. Soil Sci 173:694–706. doi:10.1097/SS.0b013e3181893b59
Giesler R, Petersson T, Högberg P (2002) Phosphorus limitation in boreal forests: effects of aluminum and iron accumulation in the humus layer. Ecosystems 5:300–314. doi:10.1007/s10021-001-0073-5
Hedley MJ, Stewart JWB, Chauhan BS (1982) Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci Soc Am J 46:970–976. doi:10.2136/sssaj1982.03615995004600050017x
Hendershot WH, Lalande H, Duquette M (2007) Ion exchange and exchangeable cations. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. CRC Press, Boca Raton, FL, USA, pp 197–206
Hou EQ, Chen CC, McGroddy ME, Wen DZ (2012) Nutrient limitation on ecosystem productivity and processes of mature and old-growth subtropical forests in China. PLoS One 7:e52071. doi:10.1371/journal.pone.0052071
Huang WJ, Liu JX, Wang YP, Zhou GY, Han TF, Li Y (2013) Increasing phosphorus limitation along three successional forests in southern China. Plant Soil 364:181–191. doi:10.1007/s11104-012-1355-8
Izquierdo JE, Houlton BZ, van Huysen TL (2013) Evidence for progressive phosphorus limitation over long-term ecosystem development: examination of a biogeochemical paradigm. Plant Soil 367:135–147. doi:10.1007/s11104-013-1683-3
Johnson AH, Frizano J, Vann DR (2003) Biogeochemical implications of labile phosphorus in forest soils determined by the Hedley fractionation procedure. Oecologia 135:487–499. doi:10.1007/s00442-002-1164-5
Jones DL, Oburger E (2011) Solubilization of phosphorus by soil microorganisms. In: Bünemann EK, Frossard E, Oberson A (eds) Phosphorus in action: biological processes in soil phosphorus cycling, soil biology 26. Springer, Berlin, Germany, pp 169–198
Lambers H, Raven JA, Shaver GR, Smith SE (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23:95–103. doi:10.1016/j.tree.2007.10.008
Lawrence D, Schlesinger WH (2001) Changes in soil phosphorus during 200 years of shifting cultivation in Indonesia. Ecology 82:2769–2780. doi:10.1890/0012-9658
Liebisch F, Keller F, Huguenin-Elie O, Frossard E, Oberson A, Bünemann EK (2014) Seasonal dynamics and turnover of microbial phosphorus in a permanent grassland. Biol Fertil Soils 50:465–475. doi:10.1007/s00374-013-0868-5
Marklein AR, Houlton BZ (2012) Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol 193:696–704. doi:10.1111/j.1469-8137.2011.03967.x
McGill WB, Cole CV (1981) Comparative aspects of cycling of organic C, N, S and P through soil organic matter. Geoderma 26:267–286. doi:10.1016/0016-7061(81)90024-0
McGroddy ME, Silver WL, de Oliveira RC, de Mello WZ, Keller M (2008) Retention of phosphorus in highly weathered soils under a lowland Amazonian forest ecosystem. J Geophys Res 113:G04012. doi:10.1029/2008JG000756
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. doi:10.1016/S0003-2670(00)88444-5
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil. In: Bünemann EK, Frossard E, Oberson A (eds) Phosphorus in action: biological processes in soil phosphorus cycling, soil biology 26. Springer, Berlin, Germany, pp 215–244
Nordby HE, Nemec S, Nagy S (1981) Fatty acids and sterols associated with citrus root mycorrhizae. J Agric Food Chem 29:396–401. doi:10.1021/jf00104a043
Oberson A, Joner EJ (2005) Microbial turnover of phosphorus in soil. In: Turner BL, Frossard E, Baldwin DS (eds) Organic phosphorus in the environment. CAB International, Wallingford, UK, pp 133–164
Olander LP, Vitousek PM (2000) Regulation of soil phosphatase and chitinase activity by N and P availability. Biogeochemistry 49:175–190. doi:10.1023/A:1006316117817
Olander LP, Vitousek PM (2004) Biological and geochemical sinks for phosphorus in soil from a wet tropical forest. Ecosystems 7:404–419. doi:10.1007/s10021-004-0264-y
Olsson PA (1999) Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soil. FEMS Microbiol Ecol 29:303–310. doi:10.1111/j.1574-6941.1999.tb00621.x
Reed SC, Townsend AR, Taylor PG, Cleveland CC (2011) Phosphorus cycling in tropical forests growing on highly weathered soils. In: Bünemann EK, Frossard E, Oberson A (eds) Phosphorus in action: biological processes in soil phosphorus cycling, soil biology 26. Springer, Berlin, German, pp 339–369
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Plant Physiol 156:989–996. doi:10.1104/pp.111.175448
Richter DD, Allen L, Li J, Markewitz D, Raikes J (2006) Bioavailability of slowly cycling soil phosphorus: major restructuring of soil P fractions over four decades in an aggrading forest. Oecologia 150:259–271. doi:10.1007/s00442-006-0510-4
Rutherford PM, McGill WB, Arocena JM, Figueiredo CT (2007) Total nitrogen. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. CRC Press, Boca Raton, FL, USA, pp 239–250
Sanchez PA (1976) Properties and management of soils in the tropics. John Wiley and Sons, New York, New York, USA
Sanyal SK, De Datta SK (1991) Chemistry of phosphorus transformations in soil. Adv Soil Sci 16:1–120. doi:10.1007/978-1-4612-3144-8_1
Selmants P, Hart S (2010) Phosphorus and soil development: does the Walker and Syers model apply to semiarid ecosystems? Ecology 91:474–484. doi:10.1890/09-0243.1
Sims JT, Pierzynski GM (2005) Chemistry of phosphorus in soils. In: Tabatabai MA, Sparks DL (eds) Chemical processes in soils. Soil Science Society of America, Madison, WI, USA, pp 151–192
Skjemstad JO, Baldock JA (2007) Total and organic carbon. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. CRC Press, Boca Raton, FL, USA, pp 225–238
Spohn M, Kuzyakov Y (2013) Phosphorus mineralization can be driven by microbial need for carbon. Soil Biol Biochem 61:69–75. doi:10.1016/j.soilbio.2013.02.013
Sullivan BW, Alvarez-Clare S, Castle SC, Porder S, Reed SC, Schreeg L, Townsend AR, Cleveland CC (2014) Assessing nutrient limitation in complex forested ecosystems: alternatives to large-scale fertilization experiments. Ecology 95:668–681. doi:10.1890/13-0825.1
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307. doi:10.1016/0038-0717(69)90012-1
Tang XL, Liu S, Zhou GY, Zhang DQ, Zhou C (2006) Soil-atmospheric exchange of CO2, CH4, and N2O in three subtropical forest ecosystems in southern China. Glob Chang Biol 12:546–560. doi:10.1111/j.1365-2486.2006.01109.x
Tang XL, Wang YP, Zhou GY, Zhang DQ, Liu S, Liu SZ, Zhang QM, Liu JX, Yan JH (2011) Different patterns of ecosystem carbon accumulation between a young and an old-growth subtropical forest in Southern China. Plant Ecol 212:1385–1395. doi:10.1007/s11258-011-9914-2
Tiessen H, Moir JO (2007) Characterization of available P by sequential extraction. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. CRC Press, Boca Raton, FL, USA, pp 293–306
Tiessen H, Stewart JWB, Cole CV (1984) Pathways of phosphorus transformations in soils of differing pedogenesis. Soil Sci Soc Am J 48:853–858. doi:10.2136/sssaj1984.03615995004800040031x
Turner BL, Lambers H, Condron LM, Cramer MD, Leake JR, Richardson AE, Smith SE (2013) Soil microbial biomass and the fate of phosphorus during long-term ecosystem development. Plant Soil 367:225–234. doi:10.1007/s11104-012-1493-z
van Diepen LT, Lilleskov EA, Pregitzer KS, Miller RM (2010) Simulated nitrogen deposition causes a decline of intra- and extraradical abundance of arbuscular mycorrhizal fungi and changes in microbial community structure in northern hardwood forests. Ecosystems 13:683–695. doi:10.1007/s10021-010-9347-0
Vandecar KL, Lawrence D, Wood T, Oberbauer SF, Das R, Tully K, Schwendenmann L (2009) Biotic and abiotic controls on diurnal fluctuations in labile soil phosphorus of a wet tropical forest. Ecology 90:2547–2555. doi:10.1890/08-1516.1
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol Appl 20:5–15. doi:10.1890/08-0127.1
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19. doi:10.1016/0016-7061(76)90066-5
Wardle D, Walker L, Bardgett R (2004) Ecosystem properties and forest decline in contrasting long-term chronosequences. Science 305:509. doi:10.1126/science.1098778
Yang X, Post WM (2011) Phosphorus transformations as a function of pedogenesis: a synthesis of soil phosphorus data using Hedley fractionation method. Biogeosciences 8:2907–2916. doi:10.5194/bg-8-2907-2011
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129. doi:10.1007/s003740050533
Zhou GY, Liu SG, Li ZA, Zhang DQ, Tang XL, Zhou CY, Yan JH, Mo JM (2006) Old-growth forests can accumulate carbon in soils. Science 314:1417. doi:10.1126/science.1130168
Zhou GY, Guan LL, Wei XH, Zhang DQ, Zhang QM, Yan JH, Wen DZ, Liu JX, Liu SG, Huang ZL, Kong GH, Mo JM, Yu QF (2007) Litterfall production along successional and altitudinal gradients of subtropical monsoon evergreen broadleaved forests in Guangdong, China. Plant Ecol 188:77–89. doi:10.1007/s11258-006-9149-9
Zhou J, Wu YH, Prietzel J, Bing HJ, Yu D, Sun SQ, Luo J, Sun HY (2013) Changes of soil phosphorus speciation along a 120-year soil chronosequence in the Hailuogou Glacier retreat area (Gongga Mountain, SW China). Geoderma 195:251–259. doi:10.1016/j.geoderma.2012.12.010
Zogg GP, Zak DR, Ringelberg DB, MacDonald NW, Pregitzer KS, White DC (1997) Compositional and functional shifts in microbial communities due to soil warming. Soil Sci Soc Am J 61:475–481. doi:10.2136/sssaj1997.03615995006100020015x
Acknowledgments
This research was funded by the National Natural Science Foundation of China (no. 31070409; 31570483) and the Strategic Priority Research Program-Climate Change: Carbon Budget and Relevant Issues of the Chinese Academy of Sciences (no. XDA05050205). The authors are grateful to Jiong Li for his assistance in field work and laboratory analysis and Dr. Christiane W. Runyan for her helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Shi, L., Wen, D. et al. Soil potential labile but not occluded phosphorus forms increase with forest succession. Biol Fertil Soils 52, 41–51 (2016). https://doi.org/10.1007/s00374-015-1053-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-015-1053-9