Abstract
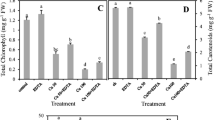
The involvement of auxin, abscisic acid (ABA), polyamines (PAs), and proline in adaptation to long-term exposure of woody plants to high levels of heavy metals in soil has received scant attention, even in poplar which is a good candidate for phytoremediation of metal-polluted soils and is regarded as a model for basic research in tree species. Three poplar clones (M1, PE19/66, and B229) were comparatively analyzed in a pot experiment for their responses to 300 mg kg−1 Cu(NO3)2 at morphological, physiological, and biochemical levels. After 4 months, despite the prevalent accumulation of Cu in roots, where the metal reached potentially toxic concentrations, the three clones showed distinct Cu accumulation and translocation capacities, whereas they did not display evident toxicity symptoms or growth inhibition. Several protective mechanisms, namely decreased photosynthetic functionality, enhanced guaiacol peroxidase (GPOD) activity, and accumulation of proline and PAs, were differentially activated in Cu-treated plants in an organ- and clone-specific manner. Overall, a positive relationship between root Cu concentration with GPOD, proline, and PAs was observed. In M1, higher Cu accumulation in roots and leaves compared with other clones was reflected in stimulation of GPOD activity in both organs and in enhanced proline, and PA levels. In PE19/66, these responses were observed only in roots concomitant with high Cu accumulation. Clone B229 accumulated very low amounts of Cu, therefore, these defense responses were attenuated compared with other clones. Enhanced ABA concentrations in response to Cu were observed in PE19/66 and B229; this was likely responsible for stomatal limitation of photosynthesis in PE19/66, whereas in B229 this effect may have been counteracted by increased IAA. Essentially unchanged leaf auxin levels under Cu stress may account for the lack of shoot growth inhibition observed in all three clones; B229 was the only clone that displayed Cu-induced IAA accumulation in roots. Results are discussed in terms of clone-specific adaptive mechanisms to Cu stress in tolerant poplars.






Similar content being viewed by others
References
Ali MB, Hahn E-J, Paeket K-Y (2006) Copper-induced changes in the growth, oxidative metabolism, and saponin production in suspension culture roots of Panax ginseng in bioreactors. Plant Cell Rep 25:1122–1132. doi:10.1007/s00299-006-0174-x
Alloway BJ (1995) Heavy metals in soils. Blackie and Son Ltd., London. ISBN 0751401986
Ashger M, Khan MIR, Anjum KNA, Khan NA (2015) Minimising toxicity of cadmium in plants-role of plant growth regulators. Protoplasma 252:399–413. doi:10.1007/s00709-014-0710-4
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216. doi:10.1016/j.envexpbot.2005.12.006
Baker AJM (1981) Accumulators and excluders: strategies in the response of plants to heavy metals. J Plant Nutr 3:643–654. doi:10.1080/01904168109362867
Baker DF, Senef JP (1995) Copper. In: Alloway BJ (ed) Heavy metals in soils. Blackie and Son Ltd., London, pp 179–205
Bankaji I, Sleimi N, López-Climent MF, Perez-Clemente RM, Gomez-Cadenas A (2014) Effects of combined abiotic stresses on growth, trace element accumulation, and phytohormone regulation in two halophytic species. J Plant Growth Regul 33:632–643. doi:10.1007/s00344-014-9413-5
Baraldi R, Bertazza G, Bregoli A, Fasolo F, Rotondi A, Predieri S, Serafini Fracassini D, Slovin JP, Cohen JD (1995) Auxins and polyamines in relation to differential in vitro root induction on microcuttings of two pear cultivars. J Plant Growth Regul 14:49–59
Bates LS, Waldren RP, Teare IK (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–208. doi:10.1007/BF00018060
Bhardwaj R, Sharma I, Handa N, Kapoor D, Kaur H, Gautam V, Kohli S (2014) Role of polyamines in stress management. In: Anjum NA, Gill SS, Gill R (eds) Plant adaptation to environmental changes: significance of amino acids and their derivatives. CABI Publishers, Wallingford, pp 245–265
Borghi M, Tognetti R, Monteforti G, Sebastiani L (2007) Responses of Populus×euramericana (P. deltoides×P. nigra) clone Adda to increasing copper concentrations. Environ Exp Bot 61:66–73. doi:10.1016/j.envexpbot.2007.03.001
Borghi M, Tognetti R, Monteforti G, Sebastiani L (2008) Responses of two poplar species (Populus alba and Populus x canadensis) to high copper concentrations. Environ Exp Bot 62:290–299. doi:10.1016/j.envexpbot.2007.10.001
Castiglione S, Todeschini V, Franchin C, Torrigiani P, Gastaldi D, Cicatelli A, Rinaudo C, Berta G, Biondi S, Lingua G (2009) Clonal differences in survival capacity, copper and zinc accumulation, and correlation with leaf polyamine levels in poplar: a large-scale field trial on heavily polluted soil. Environ Pollut 157:2108–2117. doi:10.1016/j.envpol.2009.02.011
Chaoui A, El Ferjani E (2005) Effects of cadmium and copper on antioxidant capacities, lignification and auxin degradation in leaves of pea (Pisum sativum L.) seedlings. C.R. Biol 328:23–31. doi:10.1016/j.crvi.2004.10.001
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560. doi:10.1093/aob/mcn125
Chen KH, Miller AN, Patterson GW, Cohen JD (1988) A rapid and simple procedure for purification of indole-3-acetic acid prior to GC-SIM-MS analysis. Plant Physiol 86:822–825. doi:10.1104/pp.86.3.822
Chen L, Wang L, Chen F, Korpelainen H, Li C (2013) The effects of exogenous putrescine on sex-specific responses of Populus cathayana to copper stress. Ecotoxicol Environ Safe 97:94–102. doi:10.1016/j.ecoenv.2013.07.009
Choudhary SP, Bhardwaj R, Gupta BD, Dutt P, Gupta RK, Biondi S, Kanwar M (2010) Epibrassinolide induces changes in indole-3-acetic acid, abscisic acid and polyamine concentrations and enhances antioxidant potential of radish seedlings under copper stress. Physiol Plant 140:280–296. doi:10.1371/journal.pone.0033210
Choudhary SP, Bhardwaj R, Gupta BD, Dutt P, Gupta RK, Kanwar M, Biondi S (2011) Enhancing effects of 24-epibrassinolide and putrescine on the antioxidant capacity and free radical scavenging activity of Raphanus sativus seedlings under Cu ion stress. Acta Physiol Plant 33:1319–1333. doi:10.1016/j.chemosphere.2011.03.056
Choudhary SP, Oral HV, Bhardwaj R, Yu J-Q, Phan Tran L-S (2012) Interaction of brassinosteroids and polyamines enhances copper stress tolerance in Raphanus sativus. J Exp Bot 63:5659–5675. doi:10.1093/jxb/err313
Cicatelli A, Lingua G, Todeschini V, Biondi S, Torrigiani P, Castiglione S (2010) Arbuscular mycorrhizal fungi restore normal growth in a white poplar clone grown on heavy metal-contaminated soil, and this is associated with upregulation of foliar metallothionein and polyamine biosynthetic gene expression. Ann Bot 106:791–802. doi:10.3832/ifor1045-007
Cuypers A, Vangronsveld J, Clijsters H (2002) Peroxidases in roots and primary leaves of Phaseolus vulgaris copper and zinc phytotoxicity: a comparison. J Plant Physiol 159:869–887. doi:10.1078/0176-1617-00676
Cuypers A, Smeets K, Vangronsveld J (2009) Heavy metal stress in plants. In: Hirt H (ed) Plant stress biology: from genomics to systems biology. Wiley, Weinheim, pp 161–178
Cuypers A, Smeets K, Ruytinx J, Opdenakker K, Keunen E, Remans T, Horemans N, Vanhoudt N, Van Sanden S, Van Belleghem F, Guisez Y, Colpaert J, Vangronsvel J (2011) The cellular redox state as a modulator in cadmium and copper responses in Arabidopsis thaliana seedlings. J Plant Physiol 168:309–316. doi:10.1016/j.jplph.2010.07.010
Dörffling K (2015) the discovery of abscisic acid: a retrospect. J Plant Growth Regul 34:795–808. doi:10.1007/s00344-015-9525-6
Dos Santos Utmazian MN, Wieshammer G, Vega R, Wenzel WW (2007) Hydroponic screening for metal resistance and accumulation of cadmium and zinc in twenty clones of willows and poplars. Environ Pollut 148:155–165. doi:10.1016/j.envpol.2006.10.045
Elobeid M, Polle A (2012) Interference of heavy metal toxicity with auxin physiology. In: Metal toxicity in plants: perception, signaling and remediation. In: Gupta DK, Sandalio LM (eds) Metal toxicity in plants: perception, signaling and remediation. Springer, Heidelberg, pp 249–259
Elobeid M, Göbel C, Feussner I, Polle A (2012) Cadmium interferes with auxin physiology and lignification in poplar. J Exp Bot 63:1413–1421. doi:10.1093/jxb/err384
Franchin C, Fossati T, Pasquini E, Lingua G, Castiglione S, Torrigiani P, Biondi S (2007) High concentrations of zinc and copper induce differential polyamine responses in micropropagated white poplar (Populus alba). Physiol Plant 130:77–90. doi:10.1111/j.1399-3054.2007.00886.x
Gangwar S, Singh VP (2011) Indole acetic acid differently changes growth and nitrogen metabolism in Pisum sativum L. seedlings under chromium (VI) phytotoxicity: implication of oxidative stress. Sci Hortic 129:321–328. doi:10.1016/j.scienta.2011.03.026
Gangwar S, Singh VP, Tripathi DK, Chauhan DK, Prasad SM, Maurya JN (2014) Plant responses to metal stress: the emerging role of plant growth hormones in toxicity alleviation. In: Ahmad P, Rasool S (eds) Emerging technologies and management of crop stress tolerance: volume II—a sustainable approach. Academic Press, Elsevier, San Diego, pp 69–95. doi:10.1016/B978-0-12-800875-1.00010-7
Gaudet M, Pietrini F, Beritognolo I, Iori V, Zacchini M, Massacci A, Scarascia Mugnozza G, Sabatti M (2011) Intraspecific variation of physiological and molecular response to cadmium stress in Populus nigra L. Tree Physiol 31:1309–1318. doi:10.1093/treephys/tpr088
Guerra F, Duplessis S, Kohler A, Martin F, Tapia J, Lebed P, Zamudio F, González E (2009) Gene expression analysis of Populus deltoides roots subjected to copper stress. Environ Exp Bot 67:335–344. doi:10.1016/j.envexpbot.2009.08.004
He J, Ma C, Ma Y, Li H, Kang J, Liu T, Polle A, Peng C, Luo Z-B (2013) Cadmium tolerance in six poplar species. Environ Sci Pollut Res 20:163–174. doi:10.1007/s11356-012-1008-8
Kavi Kishore PB, Sangam S, Amrutha RN, Laxmi PS, Naidu KR, Rao KRSS, Rao S, Reddy KJ, Theriappan P, Sreenivasulu N (2005) Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88:424–438
Kim Y-H, Lee H-S, Kwak S-S (2010) Differential responses of sweetpotato peroxidases to heavy metals. Chemosphere 81:79–85. doi:10.1016/j.chemosphere.2010.06.063
Kopponen P, Utriainen M, Lukkari K, Suntioinen S, Kärenlampi L, Kärenlampi S (2001) Clonal differences in copper and zinc tolerance of birch in metal-supplemented soils. Environ Pollut 112:89–97. doi:10.1016/S0269-7491(00)00096-8
Kučera T, Horáková H, Šonská A (2008) Toxic metal ions in photoautotrophic organisms. Photosynthetica 46:481–489. doi:10.1007/s11099-008-0083-z
Lagriffoul A, Mocquot B, Mench M, Vangronsveld J (1998) Cadmium toxicity effect on growth, mineral and chlorophyll contents and activities of stress related enzymes in young maize plants (Zea mays L.). Plant Soil 200:241–250
Laureysens I, Blust R, Temmerman L, Lemmens C, Ceulemans R (2004) Clonal variation in heavy metal accumulation and biomass production in a poplar coppice culture: I. Seasonal variation in leaf, wood and bark concentrations. Environ Pollut 131:485–494. doi:10.1016/j.envpol.2004.02.009
Lequeux H, Hermans C, Lutts S, Verbruggen N (2010) Response to copper excess in Arabidopsis thaliana: impact on the root system architecture, hormone distribution, lignin accumulation and mineral profile. Plant Physiol Biochem 48:673–682. doi:10.1016/j.plaphy.2010.05.005
Li SW, Leng Y, Feng L, Zeng XY (2014) Involvement of abscisic acid in regulating antioxidative defense systems and IAA-oxidase activity and improving adventitious rooting in mung bean [Vigna radiata (L.) Wilczek] seedlings under cadmium stress. Environ Sci Pollut Res 21:525–537. doi:10.1007/s11356-013-1942-0
Lichtenthaler HK, Wellburn AR (1983) Determination of total carotenoids and chlorophyll a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592. doi:10.1042/bst0110591
Lidon FC, Henriques FS (1991) Limiting step on photosynthesis of rice plants treated with varying copper levels. J Plant Physiol 138:115–118. doi:10.1016/S0176-1617(11)80741-8
Lingua G, Franchin C, Todeschini V, Castiglione S, Biondi S, Burlando B, Parravicini V, Torrigiani P, Berta G (2008) Arbuscular mycorrhizal fungi differentially affect the response to high zinc concentrations of two registered poplar clones. Environ Pollut 153:137–147. doi:10.1016/j.envpol.2007.07.012
Lohse G, Hedrich R (1992) Characterization of the plasma-membrane H+-ATPase from Vicia faba guard cells. Planta 188:206–214. doi:10.1007/BF00216815
Maestri E, Marmiroli M, Visioli G, Marmiroli N (2010) Metal tolerance and hyperaccumulation: costs and trade-offs between traits and environment. Environ Exp Bot 68:1–13. doi:10.1016/j.envexpbot.2009.10.011
Maksymiec W (2007) Signaling response in plants to heavy metal stress. Acta Physiol Plant 29:177–187. doi:10.1007/s11738-007-0036-3
Mandal C, Ghosh N, Maiti S, Das K, Gupta S, Dey N, Adak MK (2013) Antioxidative responses of Salvinia (Salvinia natans Linn.) to aluminium stress and it’s modulation by polyamine. Physiol Mol Biol Plants 19:91–103. doi:10.1007/s12298-012-0144-4
Marmiroli M, Imperiale D, Maestri E, Marmiroli N (2013) The response of Populus spp. to cadmium stress: chemical, morphological and proteomics study. Chemosphere 93:1333–1344. doi:10.1016/j.chemosphere.2013.07.065
Marschner H (1995) Mineral nutrition of higher plants. Academic Press, London, pp 344–346
Minocha R, Majumdar R, Minocha SC (2014) Polyamines and abiotic stress in plants: a complex relationship. Front Plant Sci 5:1–17. doi:10.3389/fpls.2014.00175
Mocquot B, Vangronsveld J, Clijsters H, Mench M (1996) Copper toxicity in young maize (Zea mays L.) plants: effects on growth, mineral and chlorophyll contents, and enzyme activities. Plant Soil 182:287–300
Mourato MP, Martins LL, Cuypers A (2009) Effect of copper on antioxidant enzyme activities and mineral nutrition of white lupin plants grown in nutrient solution. J Plant Nutr 32:1882–1900. doi:10.1080/01904160903242375
Murphy A, Eisinger W, Shaff J, Kochian L, Taiz L (1999) Early copper-induced leakage of K+ from arabidopsis seedlings is mediated by ion channels and coupled to citrate efflux. Plant Physiol 121:1375–1382. doi:10.1104/pp.121.4.1375
Nezames CD, Ochoa V, Larsen PB (2013) Mutational loss of Arabidopsis SLOW WALKER2 results in reduced endogenous spermine concomitant with increased aluminum sensitivity. Funct Plant Biol 40:67–78. doi:10.1071/FP12234
Official Bulletin of the Republic of Serbia No. 52/2002
Ouzounidou G, Ilias I (2005) Hormone-induced protection of sunflower photosynthetic apparatus against copper toxicity. Biol Plant 49:223–228. doi:10.1007/s10535-005-3228-yID
Pal M, Szalai G, Janda T (2015) Speculation: polyamines are important in abiotic stress signaling. Plant Sci 237:16–23. doi:10.1016/j.plantsci.2015.05.003
Pasternak T, Rudas V, Potters G, Jansen MAK (2005) Morphogenic effects of abiotic stress: reorientation of growth in Arabidopsis thaliana seedlings. Environ Exp Bot 53:299–314. doi:10.1016/j.envexpbot.2004.04.009
Pető A, Lehotai N, Lozano-Juste J, León J, Tari I, Erdei L, Kolbert Z (2011) Involvement of nitric oxide and auxin in signal transduction of copper-induced morphological responses in Arabidopsis seedlings. Ann Bot 108:449–457. doi:10.1093/aob/mcr176
Popko J, Hänsch R, Mendel R-R, Polle A, Teichmann T (2010) The role of abscisic acid and auxin in the response of poplar to abiotic stress. Plant Biol 12:242–258. doi:10.1111/j.1438-8677.2009.00305.x
Pottosin I, Velarde-Buendía AM, Bose J, Zepeda-Jazo I, Shabala S, Dobrovinskaya O (2014) Cross-talk between reactive oxygen species and polyamines in regulation of ion transport across the plasma membrane: implications for plant adaptive responses. J Exp Bot 65:1271–1283. doi:10.1093/jxb/ert423
Pulford ID, Watson C (2003) Phytoremediation of heavy metal-contaminated land by trees—a review. Environ Int 29:529–540. doi:10.1016/S0160-4120(02)00152-6
Pulliainen TK, Wallin HC (1994) Determination of total phosphorus in foods by colorimetric measurement of phosphorus as molybdenum blue after dry-ashing: NMKL interlaboratory study. J AOAC Int 77:1557–1561
Rademacher W (2015) Plant growth regulators: backgrounds and uses in plant production. J Plant Growth Regul 34:845–872. doi:10.1007/s00344-015-9541-6
Saha J, Brauer EK, Sengupta A, Popescu SC, Gupta K, Gupta B (2015) Polyamines as redox homeostasis regulators during salt stress in plants. Front Environ Sci 3:21. doi:10.3389/fenvs.2015.00021
Scaramagli S, Franceschetti M, Michael AJ, Torrigiani P, Bagni N (1999) Polyamines and flowering: spermidine biosynthesis in the different whorls of developing flowers of Nicotiana tabacum L. Plant Biosyst 133:229–237. doi:10.1080/11263509909381554
Shabala S, White RG, Djordjevic MA, Ruan Y-L, Mathesius U (2016) Root-to-shoot signalling: integration of diverse molecules, pathways and functions. Funct Plant Biol 43:87–104. doi:10.1071/FP15252
Shevyakova NI, Cheremisina AI, Kuznetsov VV (2011) Phytoremediation potential of Amaranthus hybrids: antagonism between nickel and iron and chelating role of polyamines. Russ J Plant Physl 58:634–642. doi:10.1134/S1021443711040145
Shevyakova NI, Musatenko LI, Stetsenko LA, Vedenicheva NP, Voitenko LP, Sytnik KM, Kuznetsov VV (2013) Effects of abscisic acid on the contents of polyamines and proline in common bean plants under salt stress. Russ J Plant Physiol 60:200–211. doi:10.1134/S102144371301007x
Siripornadulsil S, Traina S, Verma S, Sayre R (2002) Molecular mechanisms of proline-mediated tolerance to toxic heavy metals in transgenic microalgae. Plant Cell 14:2837–2847. doi:10.1105/tpc.004853
Smulders MJM, Cottrell JE, Lefevre F, van der Schoot J, Arens P, Vosman B, Tabbener HE, Grassi F, Fossati T, Castiglione S, Krystufek V, Fluch S, Burg K, Vornam B, Pohl A, Gebhardt K, Alba N, Agundez D, Maestro C, Notivol E, Volosyanchuk R, Pospiskova M, Bordacs S, Bovenschen J, van Dam BC, Koelewijn HP, Halfmaerten D, Ivens B, van Slycken J, Broeck AV, Storme V, Boerjan W (2008) Structure of the genetic diversity in black poplar (Populus nigra L.) populations across European river systems: consequences for conservation and restoration. For Ecol Manag 255:1388–1399. doi:10.1016/j.foreco.2007.10.063
Song Y, Ci D, Tian M, Zhang D (2014) Comparison of the physiological effects and transcriptome responses of Populus simonii under different abiotic stresses. Plant Mol Biol 86:139–156. doi:10.1007/s11103-014-0218-5
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97. doi:10.1016/j.tplants.2009.11.009
Tognetti VB, Mühlenbock P, Van Breusegem F (2012) Stress homeostasis—the redox and auxin perspective. Plant Cell Environ 35:321–333. doi:10.1111/j.1365-3040.2011.02324.x
Trudić B, Kebert M, Popović BM, Štajner D, Orlović S, Galović V (2012) The level of oxidative stress in poplars due to heavy metal pollution in soil. Balt For 18:214–227
Tsai C-J, Harding SA, Tschaplinski TJ, Lindroth RL, Yuan Y (2006) Genome-wide analysis of the structural genes regulating defense phenylpropanoid metabolism in Populus. New Phytol 172:47–62. doi:10.1186/1471-2164-11-150
Van Assche F, Clijsters C (1990) Effects of metals on enzyme activity in plants. Plant Cell Environ 13:195–206. doi:10.1111/j.1365-3040.1990.tb01304.x
Vandecasteele B, Meers E, Vervaeke P, De Vos B, Quataert P, Tack FMG (2005) Growth and trace metal accumulation of two Salix clones on sediment-derived soils with increasing contamination levels. Chemosphere 58:995–1002. doi:10.1016/j.chemosphere.2004.09.062
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Protective role of exogenous polyamines. Plant Sci 151:59–66. doi:10.1016/S0168-9452(99)00197-1
Velikova VB, Edreva AM, Tsonev TD, Jones HG (2007) Singlet oxygen quenching by phenylamides and their parent compounds. Z. Naturforsch C 62:833–838
Vitti A, Nuzzazi M, Scopa A, Tataranni G, Tamburrino I, Sofo A (2014) Hormonal response and roots architecture in Arabidopsis thaliana subjected to heavy metals. Int J Plant Biol 5226:16–21. doi:10.4081/pb.2014.5226
Von Caemmerer SV, Farquhar GD (1981) Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 153:376–387. doi:10.1007/BF00384257
Wang Y, WangY Kai W, Zhao B, Chen P, Sun L, Ji K, Li Q, Dai S, Sun Y, Wang Y, Pei Y, Leng P (2014) Transcriptional regulation of abscisic acid signal core components during cucumber seed germination and under Cu2þ, Zn2þ, NaCl and simulated acid rain stresses. Plant Physiol Biochem 76:67–76. doi:10.1016/j.plaphy.2014.01.003
Wang R, Wang J, Zhao L, Yang S, Song Y (2015) Impact of heavy metal stresses on the growth and auxin homeostasis of Arabidopsis seedlings. Biometals 28:123–132. doi:10.1007/s10534-014-9808-6
Wen X-P, Hiromichi I, Yusuke Ban Y, Matsuda N, Moriguchi T (2010) Spermidine levels are implicated in heavy metal tolerance in a spermidine synthase overexpressing transgenic European pear by exerting antioxidant activities. Transgenic Res 19:91–103. doi:10.1007/s11248-009-9296-6
Wen X-P, Ban Y, Hiromichi I, Matsuda N, Kita M, Moriguchi T (2011) Antisense inhibition of a spermidine synthase gene highlights the role of polyamines for stress alleviation in pear shoots subjected to salinity and cadmium. Environ Exp Bot 72:157–166. doi:10.1016/j.envexpbot.2011.03.001
Wullschleger SD, Weston DJ, Davis JM (2009) Populus responses to edaphic and climatic cues: emerging evidence from systems biology research. Crit Rev Plant Sci 28:368–374. doi:10.1080/07352680903241246
Xiong L, Ishitani M, Lee H, Zhu J-K (2001) The Arabidopsis LOS5/ABA3 locus encodes a molybdenum cofactor sulfurase and modulates cold stress and osmotic stress responsive gene expression. Plant Cell 13:2063–2083. doi:10.1105/TPC.010101
Xu J, Yang L, Wang Z, Dong G, Huang J, Wang Y (2006) Toxicity of copper on rice growth and accumulation of copper in rice grain in copper contaminated soil. Chemosphere 62:602–607. doi:10.1016/j.chemosphere.2005.05.050
Yamasaki H, Cohen MF (2006) NO signal at the crossroads: polyamine-induced nitric oxide synthesis in plants. Trends Plant Sci 11:522–524. doi:10.1016/j.tplants.2006.09.009
Yruela I (2009) Copper in plants: acquisition, transport and interactions. Funct Plant Biol 36:409–430. doi:10.1071/FP08288
Yuan H-M, Xu H-H, Liu W-C, Lu Y-T (2013) Copper regulates primary root elongation through PIN1-mediated auxin redistribution. Plant Cell Physiol 54:766–778. doi:10.1093/pcp/pct030
Yusuf M, Khan TA, Fariduddin Q (2016) Interaction of epibrassinolide and selenium ameliorates the excess copper in Brassica juncea through altered proline metabolism and antioxidants. Ecotoxicol Environ Safe 129:25–34. doi:10.1016/j.ecoenv.2016.03.00101
Zimmerlin A, Wojtaszek P, Bolwell GP (1994) Synthesis of dehydrogenation polymers of ferulic acid with high specificity by a purified cell-wall peroxidase from French bean (Phaseolus vulgaris L.). Biochem J 299:747–753. doi:10.1042/bj2990747
Acknowledgments
This study was supported by STSM Grants from COST Action FP0903 “Climate Change and Forest Mitigation and Adaptation in a Polluted environment (MAFor)” and by COST Action FP1106 ‘Studying Tree Responses to extreme Events: a SynthesiS (STReESS)’ to MK. Additional support was provided by funds (Ricerca Fondamentale Orientata, RFO 2012) from the University of Bologna to SB and by the Ministry of Education, Science and Technological Development of the Republic of Serbia, Project No. III 43002—Biosensing technology and the global systems for continuous research and integrated management of ecosystems to SO and MK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kebert, M., Rapparini, F., Neri, L. et al. Copper-Induced Responses in Poplar Clones are Associated with Genotype- and Organ-Specific Changes in Peroxidase Activity and Proline, Polyamine, ABA, and IAA Levels. J Plant Growth Regul 36, 131–147 (2017). https://doi.org/10.1007/s00344-016-9626-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-016-9626-x