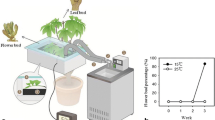
Abstract
Key message
Comparative transcriptome analysis of litchi ( Litchi chinensis Sonn.) buds at two developmental stages revealed multiple processes involving various phytohormones regulating floral initiation, and expression of numerous flowering-related genes.
Abstract
Floral initiation is a critical and complicated plant developmental process involving interactions of numerous endogenous and environmental factors, but little is known about the complex network regulating floral initiation in litchi (Litchi chinensis Sonn.). Illumina second-generation sequencing is an efficient method for obtaining massive transcriptional information resulting from phase changes in plant development. In this study, comparative transcriptomic analysis was performed with resting and emerging panicle stage buds, to gain further understanding of the molecular mechanisms involved in floral initiation in litchi. Abundance analysis identified 5,928 unigenes exhibiting at least twofold differences in expression between the two bud stages. Of these, 4,622 unigenes were up-regulated and 1,306 were down-regulated in panicle-emerging buds compared with resting buds. KEGG pathway enrichment analysis revealed that unigenes exhibiting differential expression were involved in the metabolism and signal transduction of various phytohormones. The expression levels of unigenes annotated as auxin, cytokinin, jasmonic acid, and salicylic acid biosynthesis were up-regulated, whereas those unigenes annotated as abscisic acid biosynthesis were down-regulated during floral initiation. In addition, 188 unigenes exhibiting sequence similarities to known flowering-related genes from other plants were differentially expressed during floral initiation. Thirteen genes were selected for confirmation of expression levels using quantitative-PCR. Our results provide abundant sequence resources for studying mechanisms underlying floral initiation in litchi and establish a platform for further studies of litchi and other evergreen fruit trees.








Similar content being viewed by others
References
Alabadi D, Blazquez MA, Carbonell J, Ferrandiz C, Perez-Amador MA (2009) Instructive roles for hormones in plant development. Int J Dev Biol 53:1597–1608
Amasino R (2010) Seasonal and developmental timing of flowering. Plant J 61:1001–1013
Andres F, Coupland G (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13:627–639
Barrero JM, Piqueras P, Gonzalez-Guzman M, Serrano R, Rodriguez PL, Ponce MR, Micol JL (2005) A mutational analysis of the ABA1 gene of Arabidopsis thaliana highlights the involvement of ABA in vegetative development. J Exp Bot 56:2071–2083
Bartrina I, Otto E, Strnad M, Werner T, Schmulling T (2011) Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell 23:69–80
Becker A, Theissen G (2003) The major clades of MADS-box genes and their role in the development and evolution of flowering plants. Mol Phylogenet Evol 29:464–489
Benlloch R, Berbel A, Serrano-Mislata A, Madueno F (2007) Floral initiation and inflorescence architecture: a comparative view. Ann Bot 100:659–676
Blazquez MA, Ferrandiz C, Madueno F, Parcy F (2006) How floral meristems are built. Plant Mol Biol 60:855–870
Bonhomme F, Kurz B, Melzer S, Bernier G, Jacqmard A (2000) Cytokinin and gibberellin activate SaMADS A, a gene apparently involved in regulation of the floral transition in Sinapis alba. Plant J 24:103–111
Boss PK, Bastow RM, Mylne JS, Dean C (2004) Multiple pathways in the decision to flower: enabling, promoting, and resetting. Plant Cell 16(Suppl):S18–S31
Brocard-Gifford I, Lynch TJ, Garcia ME, Malhotra B, Finkelstein RR (2004) The Arabidopsis thaliana ABSCISIC ACID-INSENSITIVE8 encodes a novel protein mediating abscisic acid and sugar responses essential for growth. Plant Cell 16:406–421
Chen WS (1991) Changes in cytokinins before and during early flower bud differentiation in lychee (Litchi chinensis Sonn.). Plant Physiol 96:1203–1206
Chen H (2002) Studies on flowering induction and differentiation in Litchi chinensis Sonn. with emphasis on their relation to temperature. Ph.D. thesis, South China Agricultural University, Guangzhou
Chen HB, Su ZX, Zhang R, Zhang HN, Ding F, Zhou BY (2014) Progresses in research of litchi floral differentiation. Sci Agric Sin 47(9):1774–1783
Cheng Y, Dai X, Zhao Y (2006) Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Genes Dev 20:1790–1799
Corbesier L, Prinsen E, Jacqmard A, Lejeune P, Van Onckelen H, Perilleux C, Bernier G (2003) Cytokinin levels in leaves, leaf exudate and shoot apical meristem of Arabidopsis thaliana during floral transition. J Exp Bot 54:2511–2517
Corbesier L, Vincent C, Jang SH, Fornara F, Fan QZ, Searle I, Giakountis A, Farrona S, Gissot L, Turnbull C, Coupland G (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316:1030–1033
Davies PJ (2004) Plant hormones: biosynthesis, signal transduction, action!, 3rd edn. Springer, Dordrecht
Davis SJ (2009) Integrating hormones into the floral-transition pathway of Arabidopsis thaliana. Plant Cell Environ 32:1201–1210
Domagalska MA, Sarnowska E, Nagy F, Davis SJ (2010) Genetic analyses of interactions among gibberellin, abscisic acid, and brassinosteroids in the control of flowering time in Arabidopsis thaliana. PLoS One 5:e14012
Dubois A, Remay A, Raymond O, Balzergue S, Chauvet A, Maene M, Pecrix Y, Yang SH, Jeauffre J, Thouroude T, Boltz V, Martin-Magniette ML, Janczarski S, Legeai F, Renou JP, Vergne P, Le Bris M, Foucher F, Bendahmane M (2011) Genomic approach to study floral development genes in Rosa sp. PLoS One 6(12):e28455
Hecht V, Laurie RE, Vander SJ, Ridge S, Knowles CL, Liew LC, Sussmilch FC, Murfet IC, Macknight RC, Weller JL (2011) The pea GIGAS gene is a FLOWERING LOCUS T homolog necessary for graft-transmissible specification of flowering but not for responsiveness to photoperiod. Plant Cell 23:147–161
Heisler MG, Ohno C, Das P, Sieber P, Reddy GV, Long JA, Meyerowitz EM (2005) Patterns of auxin transport and gene expression during primordium development revealed by live imaging of the Arabidopsis inflorescence meristem. Curr Biol 15:1899–1911
Helliwell CA, Wood CC, Robertson M, James PW, Dennis ES (2006) The Arabidopsis FLC protein interacts directly in vivo with SOC1 and FT chromatin and is part of a high-molecular-weight protein complex. Plant J 46:183–192
Hennig L, Gruissem W, Grossniklaus U, Kohler C (2004) Transcriptional programs of early reproductive stages in Arabidopsis. Plant Physiol 135:1765–1775
Honma T, Goto K (2001) Complexes of MADS-box proteins are sufficient to convert leaves into floral organs. Nature 409:525–529
Huang HB, Chen HB (2003) A phasic approach towards the floral formation in Litchi chinensis Sonn. J Fruit Sci 20:487–492
Jack T (2004) Molecular and genetic mechanisms of floral control. Plant Cell 16S:S1–S17
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484
Krajncic B, Kristl J, Janzekovic I (2006) Possible role of jasmonic acid in the regulation of floral induction, evocation and floral differentiation in Lemna minor L. Plant Physiol Biochem 44:752–758
Krizek BA, Fletcher JC (2005) Molecular mechanisms of flower development: an armchair guide. Nat Rev Genet 6:688–698
Kushiro T, Okamoto M, Nakabayashi K, Yamagishi K, Kitamura S, Asami T, Hirai N, Koshiba T, Kamiya Y, Nambara E (2004) The Arabidopsis cytochrome P450CYP707A encodes ABA 8′-hydroxylases: key enzymes in ABA catabolism. EMBO J 23:1647–1656
Li C, Wang Y, Huang X, Li J, Wang H, Li J (2013a) De novo assembly and characterization of fruit transcriptome in Litchi chinensis Sonn. and analysis of differentially regulated genes in fruit in response to shading. BMC Genomics 14:552
Li N, Chen H, Zhang Z, Hu Z, Zhou B (2013b) Cloning and expression analysis of LEAFY homologue gene in litchi. J South China Agric Univ 57–61
Liljegren SJ, Gustafson-Brown C, Pinyopich A, Ditta GS, Yanofsky MF (1999) Interactions among APETALA1, LEAFY, and TERMINAL FLOWER1 specify meristem fate. Plant Cell 11:1007–1018
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT method. Methods 25:402–408
Mandel MA, Yanofsky MF (1995) A gene triggering flower formation in Arabidopsis. Nature 377:522–524
Martinez C, Pons E, Prats G, Leon J (2004) Salicylic acid regulates flowering time and links defence responses and reproductive development. Plant J 37:209–217
Melzer R, Wang YQ, Theissen G (2010) The naked and the dead: the ABCs of gymnosperm reproduction and the origin of the angiosperm flower. Semin Cell Dev Biol 21:118–128
Moon J, Lee H, Kim M, Lee I (2005) Analysis of flowering pathway integrators in Arabidopsis. Plant Cell Physiol 46:292–299
Okada K, Ueda J, Komaki MK, Bell CJ, Shimura Y (1991) Requirement of the auxin polar transport system in early stages of Arabidopsis floral bud formation. Plant Cell 3:677–684
Pelaz S, Ditta GS, Baumann E, Wisman E, Yanofsky MF (2000) B and C floral organ identity functions require SEPALLATA MADS-box genes. Nature 405:200–203
Putterill J, Laurie R, Macknight R (2004) It’s time to flower: the genetic control of flowering time. Bioessays 26:363–373
Razem FA, El-Kereamy A, Abrams SR, Hill RD (2006) The RNA-binding protein FCA is an abscisic acid receptor. Nature 439:290–294
Rounsley SD, Ditta GS, Yanofsky MF (1995) Diverse roles for MADS box genes in Arabidopsis development. Plant Cell 7:1259–1269
Shannon S, Meeks-Wagner DR (1991) A mutation in the Arabidopsis TFL1 gene affects inflorescence meristem development. Plant Cell 3:877–892
Singh VK, Garg R, Jain M (2013) A global view of transcriptome dynamics during flower development in chickpea by deep sequencing. Plant Biotechnol J 11:691–701
Sung ZR, Belachew A, Shunong B, Bertrand-Garcia R (1992) EMF, an Arabidopsis gene required for vegetative shoot development. Science 258:1645–1647
Vanneste S, Friml J (2009) Auxin: a trigger for change in plant development. Cell 136:1005–1016
Wasternack C, Forner S, Strnad M, Hause B (2013) Jasmonates in flower and seed development. Biochimie 95:79–85
Wei YZ, Hu FC, Hu GB, Li XJ, Huang XM, Wang HC (2011) Differential expression of anthocyanin biosynthetic genes in relation to anthocyanin accumulation in the pericarp of Litchi chinensis Sonn. PLoS One 6:e19455
Wei YZ, Zhang HN, Li WC, Xie JH, Wang YC, Liu LQ, Shi SY (2013) Phenological growth stages of lychee (Litchi chinensis Sonn.) using the extended BBCH-scale. Sci Hortic Amst 161:273–277
Weigel D (1995) The genetics of flower development: from floral induction to ovule morphogenesis. Annu Rev Genet 29:19–39
Wellmer F, Alves-Ferreira M, Dubois A, Riechmann JL, Meyerowitz EM (2006) Genome-wide analysis of gene expression during early Arabidopsis flower development. PLoS Genet 2:e117
Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, Schmulling T (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15:2532–2550
Wong CE, Singh MB, Bhalla PL (2009) Molecular processes underlying the floral transition in the soybean shoot apical meristem. Plant J 57:832–845
Wong CE, Singh MB, Bhalla PL (2013) The dynamics of soybean leaf and shoot apical meristem transcriptome undergoing floral initiation process. PLoS One 8:e65319
Yang CH, Chen LJ, Sung ZR (1995) Genetic regulation of shoot development in Arabidopsis: role of the EMF genes. Dev Biol 169:421–435
Zhang JZ, Li ZM, Liu L, Mei L, Yao JL, Hu CG (2008) Identification of early-flower-related ESTs in an early-flowering mutant of trifoliate orange (Poncirus trifoliata) by suppression subtractive hybridization and macroarray analysis. Tree Physiol 28:1449–1457
Zhang XM, Zhao L, Larson-Rabin Z, Li DZ, Guo ZH (2012) De novo sequencing and characterization of the floral transcriptome of Dendrocalamus latiflorus (Poaceae: Bambusoideae). PLoS One 7:e42082
Zhang D, Ren L, Yue JH, Wang L, Zhuo LH, Shen XH (2013) A comprehensive analysis of flowering transition in Agapanthus praecox ssp. orientalis (Leighton) Leighton by using transcriptomic and proteomic techniques. J Proteomics 80:1–25
Zhong HY, Chen JW, Li CQ, Chen L, Wu JY, Chen JY, Lu WJ, Li JG (2011) Selection of reliable reference genes for expression studies by reverse transcription quantitative real-time PCR in litchi under different experimental conditions. Plant Cell Rep 30:641–653
Acknowledgments
The authors would like to thank Prof. Gui-Bing Hu, Hui-Cong Wang and Bi-Yan Zhou for their help and advice, and Don Grierson for his suggestions concerning the English. This work was funded by the China Agricultural Research Service (CARS-33), the National Natural Science Foundation of China (No. 31201581), the Natural Science Foundation of Hainan Province (No. 312033).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jim Register.
H.-N. Zhang and Y.-Z. Wei contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, HN., Wei, YZ., Shen, JY. et al. Transcriptomic analysis of floral initiation in litchi (Litchi chinensis Sonn.) based on de novo RNA sequencing. Plant Cell Rep 33, 1723–1735 (2014). https://doi.org/10.1007/s00299-014-1650-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-014-1650-3