Abstract
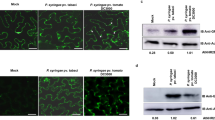
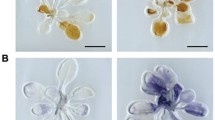
The voltage-dependent anion channel (VDAC) is a major outer mitochondrial membrane protein. It is well documented that VDAC plays an important role in apoptosis, a kind of programmed cell death, in mammalian systems. However, little is known about the role of the plant counterpart during the process of plant-specific cell death such as pathogen-induced hypersensitive response. To address this issue, we isolated three VDAC full-length cDNAs (NtVDAC1–3) from Nicotiana tabacum. The deduced products, NtVDACs, share 78–85% identity and retain the conserved eukaryotic mitochondrial porin signature distal to their C-terminal regions. Mitochondrial localization of three NtVDACs in plant cells was confirmed via a green fluorescent protein fusion method. Then, we addressed the main issue concerning pathogenesis relation. The N. benthamiana orthologues of NtVDACs were upregulated by challenge with the non-host pathogen Pseudomonas cichorii, but not after challenge with the virulent pathogen P. syringae pv. tabaci. Both the pharmaceutical inhibition of VDAC and silencing of NbVDACs genes compromised the non-host resistance against P. cichorii, suggesting the involvement of VDACs in defense against non-host pathogen. Involvement of NbVDACs in Bax-mediated cell death was also verified using a similar approach.





Similar content being viewed by others
Abbreviations
- ANT:
-
Adenine nucleotide translocator
- CsA:
-
Cyclosporine A
- DCFH-DA:
-
2′,7′-Dichlorodihydrofluorescein diacetate
- DEX:
-
Dexamethasone
- DIDS:
-
Dihydro-4,4′ diisothiocyanostilbene-2,2′-disulfonic acid
- GFP:
-
Green fluorescent protein
- HA:
-
Hemagglutinin
- HR:
-
Hypersensitive response
- MPS:
-
Mitochondrial porin signature
- MPT:
-
Mitochondrial permeability transition
- PCD:
-
Programmed cell death
- PCR:
-
Polymerase chain reaction
- PR:
-
Pathogenesis-related
- PTP:
-
Permeability transition pore
- ROS:
-
Reactive oxygen species
- TRV:
-
Tobacco rattle virus
- VDAC:
-
Voltage-dependent anion channel
- VIGS:
-
Virus-induced gene silencing
References
Adrain C, Martin SJ (2001) The mitochondrial apoptosome: a killer unleashed by the cytochrome seas. Trends Biochem Sci 26:390–397
Aoyama T, Chua NH (1997) A glucocorticoid-mediated transcriptional induction system in transgenic plants. Plant J 11:1233–1241
Arabidopsis Genome Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408:796–815
Aravind L, Dixit VM, Koonin EV (2001) Apoptotic molecular machinery: vastly increased complexity in vertebrates revealed by genome comparisons. Science 291:1279–1284
Arpagaus S, Rawyler A, Braendle R (2002) Occurrence and characteristics of the mitochondrial permeability transition in plants. J Biol Chem 277:1780–1787
Baek D, Nam J, Koo YD, Kim DH, Lee J, Jeong JC, Kwak SS, Chung WS, Lim CO, Bahk JD, Hong JC, Lee SY, Kawai-Yamada M, Uchimiya H, Yun DJ (2004) Bax-induced cell death of Arabidopsis is mediated through reactive oxygen-dependent and -independent processes. Plant Mol Biol 56:15–27
Baines CP, Kaiser RA, Sheiko T, Craigen WJ, Molkentin JD (2007) Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death. Nat Cell Biol 9:550–555
Berberich T, Sano H, Kusano T (1999) Involvement of a MAP kinase, ZmMPK5, in senescence and recovery from low-temperature stress in maize. Mol Gen Genet 262:534–542
Brenner C, Grimm S (2006) The permeability transition pore complex in cancer cell death. Oncogene 25:4744–4756
Clausen C, Ilkavets I, Thompson R, Philippar K, Vojta A, Möhlmann T, Neuhaus E, Fulgosi H, Soll J (2004) Intracellular localization of VDAC proteins in plants. Planta 220:30–37
Colombini M (1989) Voltage gating in the mitochondrial channel, VDAC. J Membr Biol 111:103–111
Curtis MJ, Wolpert TJ (2002) The oat mitochondrial permeability transition and its implication in victorin binding and induced cell death. Plant J 29:295–312
Desai MK, Mishra RN, Verma D, Nair S, Sopory SK, Reddy MK (2006) Structural and functional analysis of a salt stress inducible gene encoding voltage dependent anion channel (VDAC) from pearl millet (Pennisetum glaucum). Plant Physiol Biochem 44:483–493
Dickman MB, Oltersdorf T, Li W, Clements T, French R (2001) Abrogation of disease development in plants expressing animal antiapoptotic genes. Proc Natl Acad Sci USA 98:6957–6962
Ferri KS, Kroemer G (2001) Mitochondria—the suicide organelles. Bioessays 23:111–115
Godbole A, Varghese J, Sarin A, Mathew MK (2003) VDAC is conserved element of death pathways in plant and animal systems. Biochim Biophys Acta 1642:87–96
Hikichi Y, Suzuki K, Toyoda K, Horikoshi M, Hirooka T, Okuno T (1998) Successive observation of growth and movement of genetically lux-marked Pseudomonas cichorii and the response of host tissues in the same lettuce leaf. Ann Phytopathol Soc Jpn 64:519–525
Kawai-Yamada M, Jin L, Yoshinaga K, Hirata A, Uchimiya H (2001) Mammalian Bax-induced plant cell death can be down-regulated by overexpression of Arabidopsis Bax Inhibitor-1 (AtBI-1). Proc Natl Acad Sci USA 98:12295–12300
Kawai-Yamada M, Ohori Y, Uchimiya H (2004) Dissection of Arabidopsis Bax inhibitor-1 suppressing Bax, hydrogen peroxide and salicylic acid -induced cell death. Plant Cell 16:21–32
Krause M, Durner J (2004) Harpin inactivates mitochondria in Arabidopsis suspension cells. Mol Plant Microbe Interact 17:131–139
Kroemer G, Zamzami N, Susin SA (1997) Mitochondrial control of apoptosis. Immunol Today 18:44–51
Lacomme C, Santa Cruz S (1999) Bax-induced cell death in tobacco is similar to the hypersensitive response. Proc Natl Acad Sci USA 96:7956–7961
Liu Y, Schiff M, Marathe R, Dinesh-Kumar SP (2002) Tobacco Rar1, EDS1 and NPR1/NIM1 like genes are required for N-mediated resistance to tobacco mosaic virus. Plant J 30:415–429
Madesh M, Hajnóczky G (2001) VDAC-dependent permeabilization of the outer mitochondrial membrane by superoxide induces rapid and massive cytochrome c release. J Cell Biol 155:1003–1016
Matsumura H, Nirasawa S, Kiba A, Urasaki N, Saitoh H, Ito M, Kawai-Yamada M, Uchimiya H, Terauchi R (2003) Overexpression of Bax inhibitor suppresses the fungal elicitor-induced cell death in rice (Oryza sativa L) cells. Plant J 33:425–434
Mitsuhara I, Malik KA, Miura M, Ohashi Y (1999) Animal cell-death suppressors Bcl-xL and Ced-9 inhibit cell death in tobacco plants. Curr Biol 9:775–778
Nagy F, Kay SA, Chua N-H (1988) Analysis of gene expression in transgenic plants. In: Gelvin SB, Schilperoort RA (eds) Plant molecular biology manual. Kluwer, Dordrecht, pp 1–29
Niwa Y, Hirano T, Yoshimoto K, Shimizu M, Kobayashi H (1999) Non-invasive quantitative detection and application of non-toxic, S65T-type green fluorescent protein in living plants. Plant J 18:455–463
Page RD (1996) TreeView: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Ratcliff F, Martin-Hernandez AM, Baulcombe DC (2001) Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J 25:237–245
Roosens N, Al Bitar F, Jacobs M, Homble F (2000) Characterization of a cDNA encoding a rice mitochondrial voltage-dependent anion channel and its gene expression studied upon. Biochim Biophys Acta 1463:470–476
Sanchez FA, Santema JS, Hilhorst R, Visser AJ (1990) Fluorescence detection of enzymatically formed hydrogen peroxide in aqueous solution and in reversed micelles. Anal Biochem 187:129–132
Sharma PC, Ito A, Shimizu T, Terauchi R, Kamoun S, Saitoh H (2003) Virus-induced silencing of WIPK and SIPK genes reduces resistance to a bacterial pathogen, but has no effect on the INF1-induced hypersensitive response (HR) in Nicotiana benthamiana. Mol Genet Genomics 269:583–591
Shimizu S, Narita M, Tsujimoto Y (1999) BcL-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 399:483–487
Shimizu S, Ide T, Yanagida T, Tsujimoto Y (2000) Electrophysiological study of a novel large pore formed by Bax and the voltage-dependent anion channel that is permeable to cytochrome c. J Biol Chem 275:12321–12325
Sugiyama T, Shimizu S, Matsuoka Y, Yoneda Y, Tsujimoto Y (2002) Activation of mitochondrial -dependent anion channel by apro-apoptotic BH3-only protein Bim. Oncogene 21:4944–4956
Taguchi F, Shimizu R, Nakajima R, Toyoda K, Shiraishi T, Ichinose Y (2003) Differential effects of flagellins from Pseudomonas syringae pv. tabaci, tomato and glycinea on plant defense response. Plant Physiol Biochem 41:165–174
Takabatake R, Ando Y, Seo S, Katou S, Tsuda S, Ohashi Y, Mitsuhara I (2007) MAP kinases function downstream of HSP90 and upstream of mitochondria in TMV resistance gene N-mediated hypersensitive cell death. Plant Cell Physiol 48:498–510
Takahashi Y, Berberich T, Miyazaki A, Seo S, Ohashi Y, Kusano T (2003) Spermine signaling in tobacco: activation of mitogen-activated protein kinases by spermine is mediated through mitochondrial dysfunction. Plant J 36:820–829
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tiwari BS, Belenghi B, Levine A (2002) Oxidative stress increased respiration and generation of reactive oxygen species, resulting in ATP depletion, opening of mitochondrial permeability transition, and programmed cell death. Plant Physiol 128:1271–1281
Tsujimoto Y, Shimizu S (2007) Role of the mitochondrial membrane permeability transition in cell death. Apoptosis 12:835–840
Wandrey M, Trevaskis B, Brewin N, Udvardi MK (2004) Molecular and cell biology of a family of voltage-dependent anion channel porins in Lotus japonicus. Plant Physiol 134:182–193
Xie Z, Chen Z (2000) Harpin-induced hypersensitive cell death is associated with altered mitochondrial functions in tobacco cells. Mol Plant Microbe Interact 13:183–190
Yao N, Tada Y, Sakamoto M, Nakayashiki H, Park P, Tosa Y, Mayama S (2002) Mitochondrial oxidative burst involved in apoptotic response in oats. Plant J 30:567–579
Yao N, Eisfelder BJ, Marvin J, Greenberg JT (2004) The mitochondrion an organelle commonly involved in programmed cell death in Arabidopsis thaliana. Plant J 40:596–610
Yoshinaga K, Arimura S, Hirata A, Niwa Y, Yun D-J, Tsutsumi N, Uchimiya H, Kawai-Yamada M (2005a) Mammalian Bax initiates plant cell death through organelle destruction. Plant Cell Rep 24:408–417
Yoshinaga K, Arimura S, Niwa Y, Tsutsumi N, Uchimiya H, Kawai-Yamada M (2005b) Mitochondrial behaviour in the early stages of ROS stress leading to cell death in Arabidopsis thaliana. Ann Bot 96:337–342
Young MJ, Bay D, Hauser G, Court DA (2007) The evolutionary history of mitochondrial porins. BMC Evol Biol 7:31–52
Yu XH, Perdue TD, Heimer YM, Jones AM (2002) Mitochondrial involvement in tracheary element programmed cell death. Cell Death Differ 9:189–198
Zamzami N, Marchetti P, Castedo M, Decaudin D, Macho A, Hirsch T, Petit PX, Mignotte B, Kroemer G (1995) Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med 182:367–377
Acknowledgments
We acknowledge Drs I. Mitsuhara, Y. Niwa, Y. Ichinose, T. Shirakawa, H. Takahashi, D. Baulcombe, N.-H. Chua, and S. P. Dinesh-Kumar for providing vectors and bacterial strains. Dr. T. Berberich is acknowledged for critically reading the manuscript. This work was supported in part by Grant-in-Aid for Scientific Research for Plant Graduate Student from the Nara Institute of Science and Technology (supported by the Ministry of Education, Culture, Sports, Science, and Technology of Japan) to C. Tateda and Grant-in-Aids from the Japan Society for the Promotion of Science to T. Kusano (19658039) and Y. Takahashi (19780002), the Saito Gratitude Foundation to C. Tateda and the Sumitomo Foundation to Y. Takahashi.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Reski.
The nucleotide sequence reported in this paper has been submitted to DDBJ under the following accession numbers: NtVDAC1 (AB286176), NtVDAC2 (AB286177), and NtVDAC3 (AB286178).
An erratum to this article can be found at http://dx.doi.org/10.1007/s00299-009-0727-x
Rights and permissions
About this article
Cite this article
Tateda, C., Yamashita, K., Takahashi, F. et al. Plant voltage-dependent anion channels are involved in host defense against Pseudomonas cichorii and in Bax-induced cell death. Plant Cell Rep 28, 41–51 (2009). https://doi.org/10.1007/s00299-008-0630-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-008-0630-x