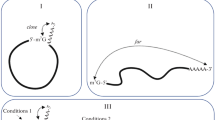
Abstract
Most eukaryotic mRNAs are recruited to the ribosome by recognition of a 5ʹ m7GpppN cap. 30 years of genetic and biochemical evidence point to a role for interaction between the 5ʹ cap-interacting factors and the 3ʹ poly(A)-binding protein in bringing the ends of the mRNA into close proximity and promoting both translation and stability of the mRNA, in a form known as the “closed loop”. However, the results of recent RNA–protein interaction studies suggest that not all mRNAs have equal access to the closed loop factors. Furthermore, association with closed loop factors appears to be highly biased towards mRNAs with short open reading frames, echoing the trend for higher translation of short mRNAs that has been observed in many eukaryotes. We recently reported that the ribosomal signaling scaffold protein RACK1 promotes the efficient translation of short mRNAs that strongly associate with the closed loop factors. Here, we discuss the implications of these observations with respect to translational control and suggest avenues through which the universality of the closed loop in eukaryotic translation could be revisited.


Similar content being viewed by others
Notes
The literature is inconsistent regarding the magnitude of stimulation required for synergy. In many cases, stimulation by the combined action of the cap and poly(A) tail that is greater than the sum of their independent effects is described as “synergy”. We favor the null hypothesis that the combination of independent effects will produce stimulation equal to the product of their individual effects. Thus, only enhancement greater than the product constitutes synergy.
References
Adamson SD, Howard GA, Herbert E (1969) The ribosome cycle in a reconstituted cell-free system from reticulocytes. Cold Spring Harb Symp Quant Biol 34:547–554
Albuquerque CP, Smolka MB, Payne SH et al (2008) A multidimensional chromatography technology for in-depth phosphoproteome analysis. Mol Cell Proteom 7:1389–1396. doi:10.1074/mcp.M700468-MCP200
Amrani N, Ghosh S, Mangus DA, Jacobson A (2008) Translation factors promote the formation of two states of the closed-loop mRNP. Nature 453:1276–1280. doi:10.1038/nature06974
Arava Y, Wang Y, Storey JD et al (2003) Genome-wide analysis of mRNA translation profiles in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 100:3889–3894. doi:10.1073/pnas.0635171100
Asano K, Shalev A, Phan L et al (2001) Multiple roles for the C-terminal domain of eIF5 in translation initiation complex assembly and GTPase activation. EMBO J 20:2326–2337. doi:10.1093/emboj/20.9.2326
Baglioni C, Vesco C, Jacobs-Lorena M (1969) The role of ribosomal subunits in mammalian cells. Cold Spring Harb Symp Quant Biol 34:555–565
Bergamini G, Preiss T, Hentze MW (2000) Picornavirus IRESes and the poly(A) tail jointly promote cap-independent translation in a mammalian cell-free system. RNA 6:1781–1790. doi:10.1017/S1355838200001679
Borman AM, Michel YM, Kean KM (2000) Biochemical characterisation of cap-poly(A) synergy in rabbit reticulocyte lysates: the eIF4G-PABP interaction increases the functional affinity of eIF4E for the capped mRNA 5ʹ-end. Nucleic Acids Res 28:4068–4075
Borman AM, Michel YM, Malnou CE, Kean KM (2002) Free poly(A) stimulates capped mRNA translation in vitro through the eIF4G-poly(A)-binding protein interaction. J Biol Chem 277:36818–36824. doi:10.1074/jbc.M205065200
Costello J, Castelli LM, Rowe W et al (2015) Global mRNA selection mechanisms for translation initiation. Genome Biol 16:10. doi:10.1186/s13059-014-0559-z
De Silva D, Tu YT, Amunts A et al (2015) Mitochondrial ribosome assembly in health and disease. Cell Cycle 14:2226–2250
Dephoure N, Zhou C, Villén J et al (2008) A quantitative atlas of mitotic phosphorylation. Proc Natl Acad Sci USA 105:10762–10767. doi:10.1073/pnas.0805139105
Eisenberg E, Levanon EY (2003) Human housekeeping genes are compact. Trends Genet 19:362–365. doi:10.1016/S0168-9525(03)00140-9
Furic L, Rong L, Larsson O et al (2010) eIF4E phosphorylation promotes tumorigenesis and is associated with prostate cancer progression. Proc Natl Acad Sci USA 107:14134–14139. doi:10.1073/pnas.1005320107
Gallie DR (1991) The cap and poly(A) tail function synergistically to regulate mRNA translational efficiency. Genes Dev 5:2108–2116. doi:10.1101/gad.5.11.2108
Gallie DR (1998) A tale of two termini. Gene 216:1–11. doi:10.1016/S0378-1119(98)00318-7
Gavin A-C, Bösche M, Krause R et al (2002) Functional organization of the yeast proteome by systematic analysis of protein complexes. Nature 415:141–147. doi:10.1038/415141a
Gavin A-C, Aloy P, Grandi P et al (2006) Proteome survey reveals modularity of the yeast cell machinery. Nature 440:631–636. doi:10.1038/nature04532
Gierer A (1963) Function of aggregated reticulocyte ribosomes in protein synthesis. J Mol Biol 6:148–157. doi:10.1016/S0022-2836(63)80131-X
Guo J, Lian X, Zhong J et al (2015) Length-dependent translation initiation benefits the functional proteome of human cells. Mol BioSyst 11:370–378. doi:10.1039/c4mb00462k
Holt LJ, Tuch BB, Villén J et al (2009) Global analysis of Cdk1 substrate phosphorylation sites provides insights into evolution. Science 325:1682–1686. doi:10.1126/science.1172867
Iizuka N, Najita L, Franzusoff A, Sarnow P (1994) Cap-dependent and cap-independent translation by internal initiation of mRNAs in cell extracts prepared from Saccharomyces cerevisiae. Mol Cell Biol 14:7322–7330. doi:10.1128/MCB.14.11.7322
Jackson RJ, Hellen CUT, Pestova TV (2010) The mechanism of eukaryotic translation initiation and principles of its regulation. Nat Rev Mol Cell Biol 11:113–127. doi:10.1038/nrm2838
Jacobson A (1996) Poly(A) metabolism and translation: the closed-loop model. In: Hershey JWB, Mathews MB, Sonenberg N (eds) Translational control. Cold Spring Harbor Laboratory Press, New York, pp 451–480
Jacobson A, Favreau M (1983) Possible involvement of poly(A) in protein synthesis. Nucleic Acids Res 11:6353–6368
Jivotovskaya AV, Valásek L, Hinnebusch AG, Nielsen KH (2006) Eukaryotic translation initiation factor 3 (eIF3) and eIF2 can promote mRNA binding to 40S subunits independently of eIF4G in yeast. Mol Cell Biol 26:1355–1372. doi:10.1128/MCB.26.4.1355-1372.2006
Joshi B, Cai AL, Keiper BD et al (1995) Phosphorylation of eukaryotic protein synthesis initiation factor 4E at Ser-209. J Biol Chem 270:14597–14603
Kahvejian A, Svitkin YV, Sukarieh R et al (2005) Mammalian poly(A)-binding protein is a eukaryotic translation initiation factor, which acts via multiple mechanisms. Genes Dev 19(1):104–113. doi:10.1101/gad.1262905
Korneeva NL, First EA, Benoit CA, Rhoads RE (2005) Interaction between the NH2-terminal domain of eIF4A and the central domain of eIF4G modulates RNA-stimulated ATPase activity. J Biol Chem 280:1872–1881. doi:10.1074/jbc.M406168200
Kozak M (2004) How strong is the case for regulation of the initiation step of translation by elements at the 3′ end of eukaryotic mRNAs? Gene 343:41–54. doi:10.1016/j.gene.2004.08.011
Lachance PED, Miron M, Raught B et al (2002) Phosphorylation of eukaryotic translation initiation factor 4E is critical for growth. Mol Cell Biol 22:1656–1663. doi:10.1128/MCB.22.6.1656
Ladhoff AM, Uerlings I, Rosenthal S (1981) Electron microscopic evidence of circular molecules in 9-S globin mRNA from rabbit reticulocytes. Mol Biol Rep 7:101–106
Lamphear BJ, Kirchweger R, Skern T, Rhoads RE (1995) Mapping of functional domains in eukaryotic protein synthesis initiation factor 4G (eIF4G) with picornaviral proteases. Implications for cap-dependent and cap-independent translational initiation. J Biol Chem 270:21975–21983
Le H, Tanguay RL, Balasta ML et al (1997) Translation initiation factors eIF-iso4G and eIF-4B interact with the poly(A)-binding protein and increase its RNA binding activity. J Biol Chem 272:16247–16255. doi:10.1074/jbc.272.26.16247
Le H, Browning KS, Gallie DR (2000) The phosphorylation state of poly(A)-binding protein specifies its binding to poly(A) RNA and its interaction with eukaryotic initiation factor (eIF) 4F, eIFiso4F, and eIF4B. J Biol Chem 275:17452–17462. doi:10.1074/jbc.M001186200
Lempiäinen H, Shore D (2009) Growth control and ribosome biogenesis. Curr Opin Cell Biol 21:855–863
Marintchev A, Wagner G (2005) eIF4G and CBP80 share a common origin and similar domain organization: implications for the structure and function of eIF4G. Biochemistry 44:12265–12272. doi:10.1021/bi051271v
Mathias AP, Williamson R, Huxley HE, Page S (1964) Occurrence and function of polysomes in rabbit reticulocytes. J Mol Biol 9:154–167
Meyuhas O, Hornstein E (2000) Translational control of TOP mRNAs. In: Sonenberg N, Hershey JWB, Mathews MB (eds) Translational control of gene expression. Cold Spring Harbor Laboratory Press, New York, pp 671–693
Meyuhas O, Kahan T (2014) The race to decipher the top secrets of TOP mRNAs. Biochim Biophys Acta 1849:801–811. doi:10.1016/j.bbagrm.2014.08.015
Michel YM, Poncet D, Piron M et al (2000) Cap-poly(A) synergy in mammalian cell-free extracts. Investigation of the requirements for poly(A)-mediated stimulation of translation initiation. J Biol Chem 275:32268–32276. doi:10.1074/jbc.M004304200
Morino S, Imataka H, Svitkin YV et al (2000) Eukaryotic translation initiation factor 4E (eIF4E) binding site and the middle one-third of eIF4GI constitute the core domain for cap-dependent translation, and the C-terminal one-third functions as a modulatory region. Mol Cell Biol 20:468–477
Munroe D, Jacobson A (1990) mRNA poly(A) tail, a 3′ enhancer of translational initiation. Mol Cell Biol 10:3441–3455. doi:10.1128/MCB.10.7.3441
O’Leary SE, Petrov A, Chen J, Puglisi JD (2013) Dynamic recognition of the mRNA cap by Saccharomyces cerevisiae eIF4E. Structure 21:2197–2207. doi:10.1016/j.str.2013.09.016
Otero LJ, Ashe MP, Sachs AB (1999) The yeast poly(A)-binding protein Pab1p stimulates in vitro poly(A)-dependent and cap-dependent translation by distinct mechanisms. EMBO J 18:3153–3163. doi:10.1093/emboj/18.11.3153
Palatnik CM, Wilkins C, Jacobson A (1984) Translational control during early Dictyostelium development: possible involvement of poly(A) sequences. Cell 36:1017–1025
Park E-H, Walker SE, Lee JM et al (2011) Multiple elements in the eIF4G1 N-terminus promote assembly of eIF4G1 PABP mRNPs in vivo. EMBO J 30:302–316. doi:10.1038/emboj.2010.312
Philipps GR (1965) Haemoglobin synthesis and polysomes in intact reticulocytes. Nature 205:567–570
Polymenis M, Aramayo R (2015) Translate to divide: control of the cell cycle by protein synthesis. Microb Cell 2:94–104. doi:10.15698/mic2015.04.198
Preiss T, Hentze MW (1998) Dual function of the messenger RNA cap structure in poly(A)-tail-promoted translation in yeast. Nature 392:516–520. doi:10.1038/33192
Ptushkina M, von der Haar T, Vasilescu S et al (1998) Cooperative modulation by eIF4G of eIF4E-binding to the mRNA 5′ cap in yeast involves a site partially shared by p20. EMBO J 17:4798–4808. doi:10.1093/emboj/17.16.4798
Raught B, Gingras A-C (2007) Signaling to translation initiation. In: Mathews MB, Sonenberg N, Hershey JWB (eds) Translational control in biology and medicine. Cold Spring Harbor Laboratory Press, New York, pp 369–400
Raught B, Gingras A-C, Gygi SP et al (2000) Serum-stimulated, rapamycin-sensitive phosphorylation sites in the eukaryotic translation initiation factor 4GI. EMBO J 19:434–444. doi:10.1093/emboj/19.3.434
Richter JD, Sonenberg N (2005) Regulation of cap-dependent translation by eIF4E inhibitory proteins. Nature 433:477–480. doi:10.1038/nature03205
Ruan Y, Sun L, Hao Y et al (2012) Ribosomal RACK1 promotes chemoresistance and growth in human hepatocellular carcinoma. J Clin Investig 122:2554–2566. doi:10.1172/JCI58488
Rush J, Moritz A, Lee KA et al (2005) Immunoaffinity profiling of tyrosine phosphorylation in cancer cells. Nat Biotechnol 23:94–101. doi:10.1038/nbt1046
Safaee N, Kozlov G, Noronha AM et al (2012) Interdomain allostery promotes assembly of the poly (A) mRNA complex with PABP and eIF4G. Mol Cell 48:375–386. doi:10.1016/j.molcel.2012.09.001
Scheper GC, Proud CG (2002) Does phosphorylation of the cap-binding protein eIF4E play a role in translation initiation? Eur J Biochem 269:5350–5359. doi:10.1046/j.1432-1033.2002.03291.x
Scheper GC, van Kollenburg B, Hu J et al (2002) Phosphorylation of eukaryotic initiation factor 4E markedly reduces its affinity for capped mRNA. J Biol Chem 277:3303–3309. doi:10.1074/jbc.M103607200
Svitkin YV, Imataka H, Khaleghpour K et al (2001) Poly(A)-binding protein interaction with elF4G stimulates picornavirus IRES-dependent translation. RNA 7:1743–1752
Swaney DL, Beltrao P, Starita L et al (2013) Global analysis of phosphorylation and ubiquitylation cross-talk in protein degradation. Nat Methods 10:676–682. doi:10.1038/nmeth.2519
Tarun SZ, Sachs AB (1995) A common function for mRNA 5′ and 3′ ends in translation initiation in yeast. Genes Dev 9:2997–3007. doi:10.1101/gad.9.23.2997
Tarun SZ, Sachs AB (1996) Association of the yeast poly(A) tail binding protein with translation initiation factor eIF-4G. EMBO J 15:7168–7177
Tarun SZ, Wells SE, Deardorff JA, Sachs AB (1997) Translation initiation factor eIF4G mediates in vitro poly(A) tail-dependent translation. Proc Natl Acad Sci USA 94:9046–9051. doi:10.1073/pnas.94.17.9046
Thomas G (2000) An encore for ribosome biogenesis in the control of cell proliferation. Nat Cell Biol 2:E71–E72. doi:10.1038/35010581
Thompson MK, Rojas-Duran MF, Gangaramani P, Gilbert WV (2016) The ribosomal protein Asc1/RACK1 is required for efficient translation of short mRNAs. Elife 5:1–22. doi:10.7554/eLife.11154
von Der Haar T, Ball PD, Mccarthy JEG (2000) Stabilization of eukaryotic initiation factor 4E binding to the mRNA 5′-cap by domains of eIF4G. J Biol Chem 275:30551–30555. doi:10.1074/jbc.M004565200
Warner JR (1999) The economics of ribosome biosynthesis in yeast. Trends Biochem Sci 24:437–440
Warner JR, Rich A, Hall CE (1962) Electron microscope studies of ribosomal clusters synthesizing hemoglobin. Science 138:1399–13403. doi:10.1126/science.138.3548.1399
Wei C-C, Balasta ML, Ren J, Goss DJ (1998) Wheat germ poly(A) binding protein enhances the binding affinity of eukaryotic initiation factor 4F and (iso)4F for cap analogues. Biochemistry 37:1910–1916. doi:10.1021/bi9724570
Wells SE, Hillner PE, Vale RD, Sachs AB (1998) Circularization of mRNA by eukaryotic translation initiation factors. Mol Cell 2:135–140. doi:10.1016/S1097-2765(00)80122-7
Wettstein F, Staehelin T, Noll H (1963) Ribosomal aggregate engaged in protein synthesis: characterization of the ergosome. Nature 197:430–435. doi:10.1038/197430a0
Wullschleger S, Loewith R, Hall MN (2006) TOR signaling in growth and metabolism. Cell 124:471–484
Acknowledgements
We thank Allan Jacobson, David Bartel, and members of the Gilbert lab for helpful discussions. This work was supported by the National Institutes of Health (GM094303) to WVG and in part by the NIH Pre-Doctoral Training Grant T32GM007287.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Rights and permissions
About this article
Cite this article
Thompson, M.K., Gilbert, W.V. mRNA length-sensing in eukaryotic translation: reconsidering the “closed loop” and its implications for translational control. Curr Genet 63, 613–620 (2017). https://doi.org/10.1007/s00294-016-0674-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-016-0674-3