Abstract
Inside individual cells, expression of genes is stochastic across organisms ranging from bacterial to human cells. A ubiquitous feature of stochastic expression is burst-like synthesis of gene products, which drives considerable intercellular variability in protein levels across an isogenic cell population. One common mechanism by which cells control such stochasticity is negative feedback regulation, where a protein inhibits its own synthesis. For a single gene that is expressed in bursts, negative feedback can affect the burst frequency or the burst size. In order to compare these feedback types, we study a piecewise deterministic model for gene expression of a self-regulating gene. Mathematically tractable steady-state protein distributions are derived and used to compare the noise suppression abilities of the two feedbacks. Results show that in the low noise regime, both feedbacks are similar in term of their noise buffering abilities. Intriguingly, feedback in burst size outperforms the feedback in burst frequency in the high noise regime. Finally, we discuss various regulatory strategies by which cells implement feedback to control burst sizes of expressed proteins at the level of single cells.






Similar content being viewed by others
References
Abramowitz M, Stegun IA (1972) Handbook of mathematical functions with formulas, graphs, and mathematical tables. National Bureau of Standards, Washington, D.C
Bandiera L, Pasini A, Pasotti L, Zucca S, Mazzini G, Magni P, Giordano E, Furini S (2016) Experimental measurements and mathematical modeling of biological noise arising from transcriptional and translational regulation of basic synthetic gene circuits. J Theor Biol 395:153–160
Bar-Even A, Paulsson J, Maheshri N, Carmi M, O’Shea E, Pilpel Y, Barkai N (2006) Noise in protein expression scales with natural protein abundance. Nat Genet 38:636–643
Becskei A, Serrano L (2000) Engineering stability in gene networks by autoregulation. Nature 405:590–593
Blake WJ, Kaern M, Cantor CR, Collins JJ (2003) Noise in eukaryotic gene expression. Nature 422:633–637
Bokes P, Singh A (2015) Protein copy number distributions for a self-regulating gene in the presence of decoy binding sites. PloS One 10(e0120):555
Bokes P, King JR, Wood ATA, Loose M (2012) Multiscale stochastic modelling of gene expression. J Math Biol 65:493–520
Bokes P, King JR, Wood ATA, Loose M (2013) Transcriptional bursting diversifies the behaviour of a toggle switch: hybrid simulation of stochastic gene expression. B Math Biol 75:351–371
Bothma J, Garcia H, Esposito E, Schlissel G, Gregor T, Levine M (2014) Dynamic regulation of eve stripe 2 expression reveals transcriptional bursts in living drosophila embryos. P Natl Acad Sci USA 111:10,598–10,603
Bressloff PC (2014) Stochastic processes in cell biology, Ch. 10. Springer, New York
Bundschuh R, Hayot F, Jayaprakash C (2003) The role of dimerization in noise reduction of simple genetic networks. J Theor Biol 220:261–269
Buratti E, Baralle F (2011) TDP-43: new aspects of autoregulation mechanisms in RNA binding proteins and their connection with human disease. FEBS J 278:3530–3538
Cai L, Friedman N, Xie XS (2006) Stochastic protein expression in individual cells at the single molecule level. Nature 440:358–62
Crudu A, Debussche A, Radulescu O (2009) Hybrid stochastic simplifications for multiscale gene networks. BMC Syst Biol 3(1):1
Crudu A, Debussche A, Muller A, Radulescu O et al (2012) Convergence of stochastic gene networks to hybrid piecewise deterministic processes. Ann Appl Probab 22(5):1822–1859
Dar RD, Razooky BS, Singh A, Trimeloni TV, McCollum JM, Cox CD, Simpson ML, Weinberger LS (2012) Transcriptional burst frequency and burst size are equally modulated across the human genome. Proc Natl Acad Sci USA 109:17,454–17,459
El-Samad H, Khammash M (2006) Regulated degradation is a mechanism for suppressing stochastic fluctuations in gene regulatory networks. Biophys J 90:3749–3761
Eldar A, Elowitz M (2010) Functional roles for noise in genetic circuits. Nature 467:167–173
Elowitz MB, Levine AJ, Siggia ED, Swain PS (2002) Stochastic gene expression in a single cell. Science 297:1183–6
Fournier T, Gabriel JP, Mazza C, Pasquier J, Galbete JL, Mermod N (2007) Steady-state expression of self-regulated genes. Bioinformatics 23:3185–3192
Friedman N, Cai L, Xie XS (2006) Linking stochastic dynamics to population distribution: an analytical framework of gene expression. Phys Rev Lett 97(168):302
Gillespie DT (1977) Exact stochastic simulation of coupled chemical reactions. J Phys Chem 81:2340–61
Grima R, Schmidt DR, Newman TJ (2012) Steady-state fluctuations of a genetic feedback loop: an exact solution. J Chem Phys 137(035):104
Grönlund A, Lötstedt P, Elf J (2013) Transcription factor binding kinetics constrain noise suppression via negative feedback. Nat Commun 4:1864
Hinch EJ (1991) Perturbation methods. Cambridge University Press, Cambridge
Hornos JEM, Schultz D, Innocentini GCP, Wang JAMW, Walczak AM, Onuchic JN, Wolynes PG (2005) Self-regulating gene: an exact solution. Phys Rev E 72(051):907
Hufton PG, Lin YT, Galla T, McKane AJ (2016) Intrinsic noise in systems with switching environments. Phys Rev E 93(5):052,119
Jangi M, Boutz P, Paul P, Sharp P (2014) Rbfox2 controls autoregulation in RNA-binding protein networks. Gene Dev 28:637–651
Kærn M, Elston T, Blake W, Collins J (2005) Stochasticity in gene expression: from theories to phenotypes. Nat Rev Genet 6:451–464
Kepler TB, Elston TC (2001) Stochasticity in transcriptional regulation: Origins, consequences, and mathematical representations. Biophys J 81:3116–3136
Kolesnikova O, Back R, Graille M, Sraphin B (2013) Identification of the Rps28 binding motif from yeast Edc3 involved in the autoregulatory feedback loop controlling RPS28b mRNA decay. Nucl Acids Res 41:9514–9523
Kuehn C (2015) Multiple time scale dynamics, vol 191. Springer, New York
Kumar N, Platini T, Kulkarni RV (2014) Exact distributions for stochastic gene expression models with bursting and feedback. Phys Rev Lett 113(26):268,105
Kumar N, Singh A, Kulkarni R (2015) Transcriptional bursting in gene expression: analytical results for general stochastic models. PLOS Comput Biol 11(e1004):292
Kumar S, Lopez AJ (2005) Negative feedback regulation among sr splicing factors encoded by rbp1 and rbp1-like in drosophila. EMBO J 24:2646–2655
Lehner B (2008) Selection to minimise noise in living systems and its implications for the evolution of gene expression. Mol Syst Biol 4:170
Lestas I, Vinnicombegv G, Paulsson J (2010) Fundamental limits on the suppression of molecular fluctuations. Nature 467:174–178
Libby E, Perkins TJ, Swain PS (2007) Noisy information processing through transcriptional regulation. Proc Natl Acad Sci USA 104:7151–7156
Lin YT, Doering CR (2016) Gene expression dynamics with stochastic bursts: construction and exact results for a coarse-grained model. Phys Rev E 93(022):409
Lin YT, Galla T (2016) Bursting noise in gene expression dynamics: linking microscopic and mesoscopic models. J R Soc Interf 13(20150):772
Maarleveld TR, Olivier BG, Bruggeman FJ (2013) Stochpy: a comprehensive, user-friendly tool for simulating stochastic biological processes. PloS One 8(e79):345
Mackey MC, Tyran-Kaminska M (2015) The limiting dynamics of a bistable molecular switch with and without noise. J Math Biol 73:367–395
Mackey MC, Tyran-Kaminska M, Yvinec R (2011) Molecular distributions in gene regulatory dynamics. J Theor Biol 274:84–96
Mackey MC, Tyran-Kaminska M, Yvinec R (2013) Dynamic behavior of stochastic gene expression models in the presence of bursting. SIAM J Appl Math 73:1830–1852
Matelska D, Purta E, Panek S, Boniecki M, Bujnicki J, Dunin-Horkawicz S (2013) S6:S18 ribosomal protein complex interacts with a structural motif present in its own mRNA. RNA 19:1341–1348
Munsky B, Neuert G, van Oudenaarden A (2012) Using gene expression noise to understand gene regulation. Science 336:183–187
Nayfeh AH (2008) Perturbation methods. Wiley, USA
Nevozhay D, Adams R, Murphy K, Josic K, Balazsi G (2009) Negative autoregulation linearizes the dose response and suppresses the heterogeneity of gene expression. Proc Natl Acad Sci USA 106:5123–5128
Newby JM (2015) Bistable switching asymptotics for the self regulating gene. J Phys A Math Theor 48(185):001
Ochab-Marcinek A, Tabaka M (2010) Bimodal gene expression in noncooperative regulatory systems. Proc Natl Acad Sci USA 107:22,096–22,101
Ochab-Marcinek A, Tabaka M (2015) Transcriptional leakage versus noise: a simple mechanism of conversion between binary and graded response in autoregulated genes. Phys Rev E 91(012):704
Paulsson J (2005) Model of stochastic gene expression. Phys Life Rev 2:157–175
Pedraza JM, Paulsson J (2008) Effects of molecular memory and bursting on fluctuations in gene expression. Science 319:339–343
Popovic N, Marr C, Swain PS (2016) A geometric analysis of fast-slow models for stochastic gene expression. J Math Biol 72:87–122
Raj A, Peskin CS, Tranchina D, Vargas D, Tyagi S (2006) Stochastic mRNA synthesis in mammalian cells. PLoS Biol 4:e309
Raser JM, O’Shea EK (2005) Noise in gene expression: origins, consequences, and control. Science 309:2010–2013
Rossbach O, Hung LH, Schreiner S, Grishina I, Heiner M, Hui J, Bindereif A (2009) Auto- and cross-regulation of the hnRNP L proteins by alternative splicing. Mol Cell Biol 29:1442–1451
Sandifer CE (2007) How Euler did it, vol 3. MAA spectrum. Mathematical Association of America
Schuss Z (2009) Theory and applications of stochastic processes: an analytical approach. Springer Science & Business Media, Germany
Singh A (2011a) Genetic negative feedback circuits for filtering stochasticity in gene expression. In: Decision and control and european control conference (CDC-ECC), 2011 50th IEEE conference on, IEEE, pp 4366–4370
Singh A (2011b) Negative feedback through mrna provides the best control of gene-expression noise. IEEE T NanoBiosci 10:194–200
Singh A (2014) Transient changes in intercellular protein variability identify sources of noise in gene expression. Biophys J 107:2214–2220
Singh A, Hespanha JP (2009a) Evolution of gene auto-regulation in the presence of noise. Syst Biol IET 3:368–378
Singh A, Hespanha JP (2009b) Optimal feedback strength for noise suppression in autoregulatory gene networks. Biophys J 96:4013–4023
Singh A, Hespanha JP (2009c) Reducing noise through translational control in an auto-regulatory gene network. In: American Control Conference, 2009. ACC’09., IEEE, pp 1712–1717
Soltani M, Bokes P, Fox Z, Singh A (2015) Nonspecific transcription factor binding can reduce noise in the expression of downstream proteins. Phys Biol 12(055):002
Strogatz SH (2014) Nonlinear dynamics and chaos: with applications to physics, biology, chemistry, and engineering. Westview press, USA
Suter DM, Molina N, Gatfield D, Schneider K, Schibler U, Naef F (2011) Mammalian genes are transcribed with widely different bursting kinetics. Science 332:472–474
Swain PS (2004) Efficient attenuation of stochasticity in gene expression through post-transcriptional control. J Mol Biol 344:965–976
Taniguchi Y, Choi PJ, Li GW, Chen H, Babu M, Hearn J, Emili A, Xie XS (2010) Quantifying E. coli proteome and transcriptome with single-molecule sensitivity in single cells. Science 329:533–538
Voliotis M, Bowsher C (2012) The magnitude and colour of noise in genetic negative feedback systems. Nucleic Acids Res p gks385
Wilkinson DJ (2006) Stochastic modelling for systems biology. CRC Press, USA
Yu J, Xiao J, Ren X, Lao K, Xie XS (2006) Probing gene expression in live cells, one protein molecule at a time. Science 311:1600–3
Zeiser S, Franz U, Müller J, Liebscher V (2009) Hybrid modeling of noise reduction by a negatively autoregulated system. B Math Biol 71:1006–1024
Acknowledgments
The authors thank Daniel Ševčovič and Branislav Novotný for discussion on some of the ideas contained herein.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
PB is supported by the Slovak Research and Development Agency grant APVV-14-0378 and also by the VEGA Grant 1/0319/15. AS is supported by the National Science Foundation Grant DMS-1312926.
Appendices
Appendix A: Reduction to the deterministic limit
In the main text we showed that, irrespective of whether the feedback acts on the burst frequency or burst size, the steady-state mean \(\langle x\rangle \) of the protein concentration tends in the small-noise limit \(\varepsilon \rightarrow 0\) to the fixed point \(x_\mathrm{s}\) of the ordinary differential equation (14). Here we provide a stronger result, showing that for both feedback types the master equation (3)–(4) reduces as \(\varepsilon \rightarrow 0\) to Liouville’s partial differential equation associated with the ordinary differential equation (14). Thus, we show that both regulation strategies yield the same deterministic model in the limit of small noise.
For feedback in burst frequency (8) the probability flux (4) is given by
If \(\varepsilon \) is small, a dominant contribution to the integral in (A.1) comes from a neighbourhood of the upper integration limit \(y=x\). Following Watson’s lemma (Hinch 1991), we extend the lower integration limit in (A.1) to \(-\infty \) and use the approximation
obtaining
at the leading order; higher-order terms, which are not required for our present purposes, can be determined by including in (A.2) additional terms of the Taylor series expansion in y around x. The right-hand side of (A.3), being the product of the protein pdf and the right-hand side of the ODE (14), gives the flux of probability induced by the drift of the deterministic model. Inserting (A.3) into the probability conservation law (3) yields a Liouville equation (Schuss 2009, p. 213)—a Chapman–Kolmogorov equation without diffusion or jumps—whose solutions are time-dependent pdfs for a variable which evolves deterministically according to (14). Thus, the stochastic model with feedback in burst frequency given by the conservation law (3) and (A.1) reduces as \(\varepsilon \) tends to zero to the deterministic model (14).
If feedback acts on burst size (19), the probability flux (4) simplifies to
Again, a neighbourhood of the upper limit \(y=x\) of integration dominates in its contribution to the integral in (A.4). Therefore, we extend the lower integration limit to \(-\infty \) without incurring appreciable error; we also use the approximations
which are valid for y that is close to x. Inserting (A.5) into (A.4) and integrating the simple exponential, we obtain the same leading-order approximation (A.3) for the probability flux (A.4) as we previously did for the flux (A.1). Thus, whether the bursting stochastic model operates a feedback in burst frequency (A.1) or in burst size (A.4), it reduces to the same deterministic model (14) in the small-noise limit of \(\varepsilon \rightarrow 0\).
Appendix B: Strong feedback asymptotics (burst size)
Inserting the second Lyapunov function (20) into the WKB form (9), we obtain
for the protein pdf in the case of feedback in burst size.
Similarly as in the Main Text, we express the protein moments as
where instead of (26) we have
Again, \(B_0^{-1}=C\) is the normalisation constant. Substituting \(x=Ky\) in the integral (B.3) yields
where
in which \(\lambda =K/\varepsilon \) is an auxiliary parameter.
In the case of strong feedback, we have \(\lambda \ll 1\), which implies \(y\gg 1\), and therefore the term \(y^{H+1}/(H+1)\) dominates the term y in the exponential of (B.5), so that
where \({\varGamma }(z)\) is the gamma function (Abramowitz and Stegun 1972). Unlike for feedback in burst frequency, here is no need to treat \(A_0\) differently from \(A_1\) or \(A_2\).
For the protein mean we have
where the prefactor \(D_\varepsilon \) is given by
Unlike for feedback in burst frequency, which yielded a slow logarithmic decrease of the mean with decreasing dissociation constant K, here we obtain a faster power-law decrease, which is consistent with the LNA prediction (44). Additionally, as \(\varepsilon \) tends to zero the prefactor \(D_\varepsilon \) converges to one, which is the prefactor of the LNA-predicted power law. The asymptotics of \(D_\varepsilon \) as \(\varepsilon \rightarrow 0\) follow from
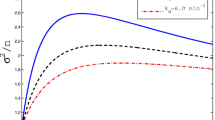
in which we take \(z=\varepsilon ^{-1}/(H+1)\) and \(a=1/(H+1)\); see Abramowitz and Stegun (1972) for this and other properties of the gamma function. These results suggest that, unlike for feedback in burst frequency, where the LNA approximation could only be used for intermediate ranges of K, here the LNA yields a uniform (i.e. valid for all K) approximation (Fig. 7).
For the protein CV\(^2\) we have
which, for a fixed \(\varepsilon \), is a constant independent of K. As \(\varepsilon \) tends to zero, we can again use (B.9) to show that the right-hand side of (B.10) is equal to \(\varepsilon /(H+1)\) at the leading order in \(\varepsilon \), which is the same value as that obtained by taking K very small in the LNA prediction (23). This suggests that the LNA approximation of the coefficient of variation, like that of the mean, can be used uniformly for all K.
Appendix C: Noncooperative feedback
Here we present variants of the figures from the Results in the Main Text obtained by taking \(H=1\) (noncooperative feedback) instead of \(H=4\). We shall not repeat the points made in the Main Text, focusing instead on the main differences that occur in the absence of cooperativity.
\(\mathbf{CV}^2\) monotonically increases with strengthening noncooperative feedback in burst frequency In contrast with the cooperative case, where a gradual increase in burst-frequency feedback strength led at first to a transient decrease in protein noise (Fig. 3), without cooperativity the CV\(^2\) is strictly increasing (Fig. 8).
Protein mean and \(\mathbf{CV}^2\) are less sensitive to feedback strength Wider ranges of dissociation constants are required to achieve similar changes in protein mean and noise as those reported previously. In order to appreciate this, one needs to compare the scales on the horizontal axes of Figs. 8, 9, 10, 11 with those of their counterparts in the Main Text.
Noncooperative performs worse than the cooperative in reducing noise Noncooperative feedback, even if acting through burst size, leads at best to a \(50~\%\) reduction in CV\(^2\) (Fig. 11), which is inferior to a \(80~\%\) reduction achievable in the cooperative case with \(H=4\) (Fig. 6).
The main conclusion of the Main Text holds also in noncooperative case. The regulation in burst size performs better in reducing noise, especially for noisy proteins subject to strong self-repression (Fig. 11).
Appendix D: Discrete simulations
The discrete model is a chemical system of two species (Wilkinson 2006), A and P, whereby \(A\in \{0,1\}\) is an indicator variable describing whether the gene is active (\(A=1\)) or inactive (\(A=0\)) and P gives the number of protein.
The two species are subject to four reactions, gene activation, gene inactivation, protein production, and protein decay. Each reaction is characterised by the change in copy numbers that a single occurrence of the reaction induces and by the stochastic rate with which the reaction occurs (Table 1).
The dependence of the rates of activation \(\tilde{k}_\mathrm{on}(P)\), inactivation \(\tilde{k}_\mathrm{off}(P)\), protein production \(\tilde{k}_\mathrm{p}(P)\) and protein decay \(\tilde{k}_\mathrm{d}(P)\) is as yet undefined in Table 1, but is specified below for feedbacks in burst frequency and burst size. We use tildes to distinguish the microscopic rates (expressed in terms of individual molecules) from the macroscopic ones (expressed in terms of concentrations) which were used throughout the Main Text.
For feedback in burst frequency, we choose
while for feedback in burst size, we use
In addition to the noise parameter \(\varepsilon \), dimensionless dissociation constant K and the cooperativity coefficient H, which have been introduced in the Main Text, cf. Eq. (8) and (19), we have in (D.1) and (D.2) two new parameters: \(\delta \) and \({\varOmega }\). The parameter \(\delta \) compares the time scale of gene activity to that of protein turnover, and \({\varOmega }\) is the system size parameter: the number of proteins corresponding to the unit of concentration.
Provided that \(\delta \ll 1\) and \({\varOmega }\gg 1\), the protein concentration defined as \(x=P/{\varOmega }\) can be compared to the predictions of the continuous bursting model (7). For mathematical analysis of the bursting asymptotics (\(\delta \ll 1\)) as well as system-size asymptotics (\({\varOmega }\gg 1\)), we refer the reader to Bokes et al. (2012).
In Fig. 2, we used \(\varepsilon =0.2\), \(H=4\), a range of values of K (detailed within the figure panels), \(\delta =0.01\) and \({\varOmega }=100\). Each distribution was estimated from a single large run (\(10^5\) iterations) of Gillespie’s direct method (Gillespie 1977) implemented in the StochPy stochastic modelling software package (Maarleveld et al. 2013).
Rights and permissions
About this article
Cite this article
Bokes, P., Singh, A. Gene expression noise is affected differentially by feedback in burst frequency and burst size. J. Math. Biol. 74, 1483–1509 (2017). https://doi.org/10.1007/s00285-016-1059-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-016-1059-4