Abstract
Purpose
This phase I study was conducted primarily to determine the maximum tolerated dose (MTD) of tesetaxel, a novel, orally active, semisynthetic microtubule inhibitor of the taxane class, administered with oral capecitabine to patients with advanced solid tumors.
Methods
During each 21-day cycle, patients were to receive tesetaxel on Day 1 and capecitabine twice daily on Days 1 through 14. The starting dose was tesetaxel 18 mg/m2 and capecitabine 1,250 mg/m2/day. These doses were increased based on tolerability during the first cycle according to the protocol-specified dose-escalation scheme. Response was evaluated every other treatment cycle according to RECIST. Serial blood samples were collected during the first and second cycles to explore possible pharmacokinetic drug interactions.
Results
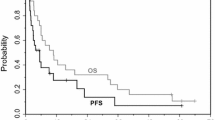
Twenty-seven patients were enrolled and treated. The most frequently reported dose-limiting toxicities were neutropenia and febrile neutropenia, with individual patients experiencing dose-limiting stomatitis and diarrhea. The MTD for the treatment regimen was defined as tesetaxel 27 mg/m2 and capecitabine 2,500 mg/m2/day. The most common ≥Grade 3 treatment-related adverse events included leukopenia (44% of patients) and neutropenia (41%). Of 22 evaluable patients, the best overall response was stable disease in 82% and progressive disease in 18%. No meaningful pharmacokinetic drug interactions were apparent.
Conclusions
The results of this study demonstrate that these two orally active agents can be combined at the individual MTD of each drug with acceptable toxicity. These data further support the continued clinical development of tesetaxel both as a single agent and in combination with other active cancer therapeutics.
Similar content being viewed by others
References
Shionoya M, Jimbo T, Kitagawa M, Soga T, Tohgo A (2003) DJ-927, a novel oral taxane, overcomes P-glycoprotein-mediated multidrug resistance in vitro and in vivo. Cancer Sci 94:459–466
Tohgo A, Shionoya M, Iwahana M, Uesugi Y, Jimbo T, Soga T (2002) DJ-927, a novel orally active taxane: 1. Preclinical antitumor activity and toxicity profile. Proceedings of the AACR 43:790 (Abstract 3916)
Chan S, Paridaens R, Awada A, Mukherjee A, Lawton P, Dumez H et al (2004) Efficacy and prediction of response to the new oral taxane DJ-927 in anthacycline <sic> pre-treated advanced breast cancer (ABC). Eur J Cancer Suppl 4 (No.12):193 (Abstract 641)
Evans T, Dobrila R, Berardi R, Sumpter KA, Wall LR, Oyama R et al (2006) A Phase II study of DJ-927 as second-line therapy in patients (pts) with advanced gastric cancer (GC) who have failed a 5-FU non taxane based regimen. J Clin Oncol, 2006 ASCO Annual Meeting Proceedings Part I. 24 (No. 18S) (Abstract 4081)
Moore MR, Jones C, Harker G, Lee F, Ardalan B, Saif MW et al (2006) Phase II trial of DJ-927, an oral tubulin depolymerization inhibitor, in the treatment of metastatic colorectal cancer. J Clin Oncol ASCO Annual Meeting Proceedings Part I. 24 (No. 18S) (Abstract 3591)
Ishikawa T, Utoh M, Sawada N, Nishida M, Fukase Y, Sekiguchi F et al (1998) Tumor selective delivery of 5-fluorouracil by capecitabine, a new oral fluoropyrimidine carbamate, in human cancer xenografts. Biochem Pharmacol 55:1091–1097
Maher JF, Villalona-Calero MA (2002) Taxanes and capecitabine in combination: rationale and clinical results. Clin Breast Cancer 2:287–293
Xeloda™ prescribing information. Genentech USA, Inc
Hoff PM, Ansari R, Batist G, Cox J, Kocha W, Kuperminc M et al (2001) Comparison of oral capecitabine versus intravenous fluorouracil plus leucovorin as first-line treatment in 605 patients with metastatic colorectal cancer: results of a randomized Phase III study. J Clin Oncol 19:2282–2292
Van Cutsem E, Twelves C, Cassidy J, Allman D, Bajetta E, Boyer M et al (2001) Oral capecitabine compared with intravenous fluorouracil plus leucovorin in patients with metastatic colorectal cancer: results of a large phase III study. J Clin Oncol 19:4097–4106
O’Shaughnessy J, Miles D, Vukelja S, Moiseyenko V, Ayoub JP, Cervantes G et al (2002) Superior survival with capecitabine plus docetaxel combination therapy in anthracycline-pretreated patients with advanced breast cancer: phase III trial results. J Clin Oncol 20:2812–2823
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L et al (2000) New guidelines to evaluate the response to treatment of solid tumors. J Natl Cancer Inst 92:205–216
Acknowledgment
This study was funded by Daiichi Sankyo, G. Schwartz, P. Cheverton, M. Kimura, and M. Danna.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saif, M.W., Sarantopoulos, J., Patnaik, A. et al. Tesetaxel, a new oral taxane, in combination with capecitabine: a phase I, dose-escalation study in patients with advanced solid tumors. Cancer Chemother Pharmacol 68, 1565–1573 (2011). https://doi.org/10.1007/s00280-011-1639-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-011-1639-3