Abstract
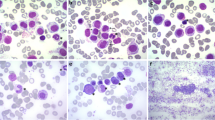
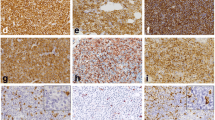
Mast cell leukemia (MCL) is a rare, life-threatening malignancy defined by a substantial increase in neoplastic mast cells (MCs) in bone marrow (BM) smears, drug-resistance, and a poor prognosis. In most patients, the survival time is less than 1 year. However, exceptional cases may present with a less malignant course. We report on a 49-year-old female patient with MCL diagnosed in 2013. In February 2013, first symptoms, including flushing, headache, and diarrhea, were recorded. In addition, mild anemia was detected. The disease was characterized by a massive increase in well-granulated, mature, and often spindle-shaped MCs (80 %) in BM smears. The serum tryptase level amounted to 332 ng/mL. Like in most other MCL patients, no skin lesions were detected. However, unlike in other patients, tryptase levels remained stable, and no other signs or symptoms of MCL-induced organ damage were found. Sequencing studies revealed an isolated S476I point mutation in KIT but no mutation in codon 816. The patient received histamine receptor blockers but refused cytoreductive therapy. After 9 months, still no progression or organ damage was detected. However, progression with transformation to acute MCL occurred after 12 months. We propose that the chronic type of MCL with stable conditions, absence of organ damage, and a mature MC morphology is recognized as a distinct entity that should be distinguished from the acute variant of MCL.



Similar content being viewed by others
References
Valent P, Akin C, Sperr WR et al (2003) Diagnosis and treatment of systemic mastocytosis: state of the art. Br J Haematol 122(5):695–717
Horny HP, Sotlar K, Valent P (2007) Mastocytosis: state of the art. Pathobiology 74(2):121–132
Metcalfe DD (2008) Mast cells and mastocytosis. Blood 112(4):946–956
Arock M, Valent P (2010) Pathogenesis, classification and treatment of mastocytosis: state of the art in 2010 and future perspectives. Expert Rev Hematol 3(4):497–516
Valent P, Horny HP, Escribano L et al (2001) Diagnostic criteria and classification of mastocytosis: a consensus proposal. Leuk Res 25(7):603–625
Valent P, Horny H-P, Li CY et al (2001) Mastocytosis (mast cell disease). In: Jaffe ES, Harris NL, Stein H, Vardiman JW (eds) World Health Organization (WHO) Classification of Tumours. Pathology & Genetics. Tumours of Haematopoietic and Lymphoid Tissues. IARC Press Lyon, France, pp 291–302
Horny HP, Akin C, Metcalfe DD et al (2008) Mastocytosis (mast cell disease). In: World Health Organization (WHO) Classification of Tumours. Pathology & Genetics. In: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW (eds) Tumours of haematopoietic and lymphoid tissues. IARC Press, Lyon, France, pp 54–63
Travis WD, Li CY, Hoagland HC et al (1986) Mast cell leukemia: report of a case and review of the literature. Mayo Clin Proc 61(12):957–966
Valent P, Akin C, Sperr WR et al (2003) Aggressive systemic mastocytosis and related mast cell disorders: current treatment options and proposed response criteria. Leuk Res 27(7):635–641
Georgin-Lavialle S, Lhermitte L, Dubreuil P et al (2013) Mast cell leukemia. Blood 121(8):1285–1295
Gotlib J, Berubé C, Growney JD et al (2005) Activity of the tyrosine kinase inhibitor PKC412 in a patient with mast cell leukemia with the D816V KIT mutation. Blood 106(8):2865–2870
Lim KH, Pardanani A, Butterfield JH et al (2009) Cytoreductive therapy in 108 adults with systemic mastocytosis: outcome analysis and response prediction during treatment with interferon-alpha, hydroxyurea, imatinib mesylate or 2-chlorodeoxyadenosine. Am J Hematol 84(12):790–794
Pardanani A, Lim KH, Lasho TL et al (2009) Prognostically relevant breakdown of 123 patients with systemic mastocytosis associated with other myeloid malignancies. Blood 114(18):3769–3772
Pagano L, Valentini CG, Caira M et al (2008) Advanced mast cell disease: an Italian Hematological Multicenter experience. Int J Hematol 88(5):483–488
Valent P, Sotlar K, Sperr WR et al. (2014) Refined diagnostic criteria and classification of mast cell leukemia (MCL) and myelomastocytic leukemia (MML): a consensus proposal. Ann Oncol 25(9):1691–1700
Valent P, Akin C, Escribano L et al (2007) Standards and standardization in mastocytosis: consensus statements on diagnostics, treatment recommendations and response criteria. Eur J Clin Invest 37(6):435–453
Escribano L, Diaz-Agustin B, López A et al (2004) Spanish Network on Mastocytosis (REMA). Immunophenotypic analysis of mast cells in mastocytosis: When and how to do it. Proposals of the Spanish Network on Mastocytosis (REMA). Cytometry B Clin Cytom 58(1):1–8
Sotlar K, Escribano L, Landt O et al (2003) One-step detection of c-kit point mutations using peptide nucleic acid-mediated polymerase chain reaction clamping and hybridization probes. Am J Pathol 162(3):737–746
Sperr WR, Escribano L, Jordan JH et al (2001) Morphologic properties of neoplastic mast cells: delineation of stages of maturation and implication for cytological grading of mastocytosis. Leuk Res 25(7):529–536
Horny HP, Valent P (2001) Diagnosis of mastocytosis: general histopathological aspects, morphological criteria and immunohistochemical findings. Leuk Res 25(7):543–551
Horny HP, Sillaber C, Menke D et al (1998) Diagnostic value of immunostaining for tryptase in patients with mastocytosis. Am J Surg Pathol 22(9):1132–1140
Fritsche-Polanz R, Jordan JH, Feix A et al (2001) Mutation analysis of C-KIT in patients with myelodysplastic syndromes without mastocytosis and cases of systemic mastocytosis. Br J Haematol 113(2):357–364
Sotlar K (2013) c-kit mutational analysis in paraffin material. Methods Mol Biol 999:59–78
Gleixner KV, Mayerhofer M, Aichberger KJ et al (2006) PKC412 inhibits in vitro growth of neoplastic human mast cells expressing the D816V-mutated variant of KIT: comparison with AMN107, imatinib, and cladribine (2CdA) and evaluation of cooperative drug effects. Blood 107(2):752–759
Peter B, Gleixner KV, Cerny-Reiterer S et al (2011) Polo-like kinase-1 as a novel target in neoplastic mast cells: demonstration of growth-inhibitory effects of small interfering RNA and the Polo-like kinase-1 targeting drug BI 2536. Haematologica 96(5):672–680
Sotlar K, Cerny-Reiterer S, Petat-Dutter K et al (2011) Aberrant expression of CD30 in neoplastic mast cells in high-grade mastocytosis. Mod Pathol 24(4):585–595
Nagata H, Worobec AS, Oh CK et al (1995) Identification of a point mutation in the catalytic domain of the protooncogene c-kit in peripheral blood mononuclear cells of patients who have mastocytosis with an associated hematologic disorder. Proc Natl Acad Sci U S A 92(23):10560–10564
Longley BJ, Tyrrell L, Lu SZ et al (1996) Somatic c-KIT activating mutation in urticaria pigmentosa and aggressive mastocytosis: establishment of clonality in a human mast cell neoplasm. Nat Genet 12(3):312–314
Féger F, Ribadeau Dumas A et al (2002) Kit and c-kit mutations in mastocytosis: a short overview with special reference to novel molecular and diagnostic concepts. Int Arch Allergy Immunol 127(2):110–114
Bodemer C, Hermine O, Palmérini F et al (2010) Pediatric mastocytosis is a clonal disease associated with D816V and other activating c-KIT mutations. J Invest Dermatol 130(3):804–815
Valent P, Blatt K, Eisenwort G et al (2013) FLAG-induced remission in a patient with acute mast cell leukemia (MCL) exhibiting t(7;10)(q22;q26) and KIT D816H. Leuk Res Rep 3(1):8–13
Yang Y, Létard S, Borge L et al (2010) Pediatric mastocytosis-associated KIT extracellular domain mutations exhibit different functional and signaling properties compared with KIT-phosphotransferase domain mutations. Blood 116(7):1114–1123
Valent P, Arock M, Bonadonna P et al (2012) European Competence Network on Mastocytosis (ECNM): 10-year jubilee, update, and future perspectives. Wien Klin Wochenschr 124(23–24):807–814
Escribano L, Orfao A, Villarrubia J et al (1997) Sequential immunophenotypic analysis of mast cells in a case of systemic mast cell disease evolving to a mast cell leukemia. Cytometry 30(2):98–102
Escribano L, Orfao A, Díaz-Agustin B et al (1998) Indolent systemic mast cell disease in adults: immunophenotypic characterization of bone marrow mast cells and its diagnostic implications. Blood 91(8):2731–2736
Teodosio C, García-Montero AC, Jara-Acevedo M et al (2013) Gene expression profile of highly purified bone marrow mast cells in systemic mastocytosis. J Allergy Clin Immunol 131(4):1213–1224
Hoermann G, Blatt K, Greiner G et al. (2014) CD52 is a molecular target in advanced systemic mastocytosis. FASEB J 28(8):3540–3551
Schernthaner GH, Jordan JH, Ghannadan M et al (2001) Expression, epitope analysis, and functional role of the LFA-2 antigen detectable on neoplastic mast cells. Blood 98(13):3784–3792
Valent P, Sotlar K, Horny HP (2011) Aberrant expression of CD30 in aggressive systemic mastocytosis and mast cell leukemia: a differential diagnosis to consider in aggressive hematopoietic CD30-positive neoplasms. Leuk Lymphoma 52(5):740–744
Morgado JM, Perbellini O, Johnson RC et al (2013) CD30 expression by bone marrow mast cells from different diagnostic variants of systemic mastocytosis. Histopathology 63(6):780–787
Schwaab J, Schnittger S, Sotlar K et al (2013) Comprehensive mutational profiling in advanced systemic mastocytosis. Blood 122(14):2460–2466
Damaj G, Joris M, Chandesris O et al (2014) ASXL1 but not TET2 mutations adversely impact overall survival of patients suffering systemic mastocytosis with associated clonal hematologic non-mast-cell diseases. PLoS ONE 9(1):e85362
Acknowledgments
We would like to thank Daniela Berger, Gabriele Stefanzl, and Susanne Herndlhofer for their skillful technical assistance. This study was supported by a Mastocytosis Grant of the Medical University of Vienna and the Austrian National Science Fund (FWF) grant no. SFB-F4704.
Contributions
P.V. and M.A. designed the study and wrote the paper. S.C.R., L.M. and H.P.H. contributed histomorphological, histopathological, and immunohistochemical studies. G.H. and C.M. provided molecular studies and KIT sequencing analyses. P.V., J.B., M.S., and M.A. contributed patients and provided patient-derived material. B.P. performed in vitro proliferation studies using patient-derived cells and KIT-targeting tyrosine kinase inhibitors. G.E. and P.B. performed flow cytometry studies. P.V., P.B., and M.A. contributed cytomorphological studies. All authors approved the final version of the manuscript.
Conflict of interest
P.V. and H.P.H. are Novartis-Consultants in a global trial examining the effects of PKC412 (midostaurin) in advanced systemic mastocytosis. P.V. received a research grant from Novartis and a research grant from The Mastocytosis Society.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by the Austrian Science Fund (FWF) grant no. SFB-F4704
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 85 kb)
Rights and permissions
About this article
Cite this article
Valent, P., Berger, J., Cerny-Reiterer, S. et al. Chronic mast cell leukemia (MCL) with KIT S476I: a rare entity defined by leukemic expansion of mature mast cells and absence of organ damage. Ann Hematol 94, 223–231 (2015). https://doi.org/10.1007/s00277-014-2207-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-014-2207-9