Abstract
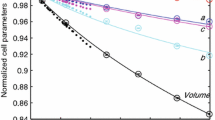
The comparative compressibility and high-pressure stability of a natural epidote (0.79 Fe-total per formula unit, Fetot pfu) and clinozoisite (0.40 Fetot pfu) were investigated by single-crystal X-ray diffraction and Raman spectroscopy. The lattice parameters of both phases exhibit continuous compression behavior up to 30 GPa without evidence of phase transformation. Pressure–volume data for both phases were fitted to a third-order Birch–Murnaghan equation of state with V 0 = 461.1(1) Å3, K 0 = 115(2) GPa, and \(K_{0}^{'}\) = 3.7(2) for epidote and V 0 = 457.8(1) Å3, K 0 = 142(3) GPa, and \(K_{0}^{'}\) = 5.2(4) for clinozoisite. In both epidote and clinozoisite, the b-axis is the stiffest direction, and the ratios of axial compressibility are 1.19:1.00:1.15 for epidote and 1.82:1.00:1.19 for clinozoisite. Whereas the compressibility of the a-axis is nearly the same for both phases, the b- and c-axes of the epidote are about 1.5 times more compressible than in clinozoisite, consistent with epidote having a lower bulk modulus. Raman spectra collected up to 40.4 GPa also show no indication of phase transformation and were used to obtain mode Grüneisen parameters (γ i) for Si–O vibrations, which were found to be 0.5–0.8, typical for hydrous silicate minerals. The average pressure coefficient of Raman frequency shifts for M–O modes in epidote, 2.61(6) cm−1/GPa, is larger than found for clinozoisite, 2.40(6) cm−1/GPa, mainly due to the different compressibility of FeO6 and AlO6 octahedra in M3 sites. Epidote and clinozoisite contain about 2 wt% H2O are thus potentially important carriers of water in subducted slabs.






Similar content being viewed by others
References
Alvaro M, Angel RJ, Camara F (2012) High-pressure behavior of zoisite. Am Mineral 97:1165–1176
Angel RJ, Alvaro M, Gonzales-Platas J (2014) EosFit7c and a Fortran module (library) for equation of state calculations. Z Kristallogr 229:405–419
Aroyo MI, Perez-Mato JM, Capillas C, Kroumova E, Ivantchev S, Madariaga G, Kirov A, Wondratschek H (2006a) Bilbao crystallographic server: I. Databases and crystallographic computing programs. Z Kristallogr 221:15–27
Aroyo MI, Kirov A, Capillas C, Perez-MatoaJM WondratschekH (2006b) Bilbao crystallographic server. II. Representations of crystallographic point groups and space groups. Acta Cryst A62:115–128
Bevington PR, Robinson DK (2003) Data reduction and error analysis for the physical sciences, 3rd edn. McGraw-Hill, New York
Bird DK, Spieler AR (2004) Epidote in geothermal systems. Rev Miner Geochem 56:235–300
Bonazzi P, Menchetti S (1995) Monoclinic members of the epidote group: effects of the Al↔Fe3+↔Fe2+ substitution and of the entry of REE3+. Miner Mag 53:133–153
Bonazzi P, Menchetti S (2004) Manganese in monoclinic members of the epidote group: piemontite and related minerals. Rev Miner Geochem 56:495–552
Born M, Huang K (1954) Dynamical theory of crystal lattices. Oxford University Press, UK
Bradbury SE, Williams Q (2003) Contrasting bonding behavior of two hydroxyl-bearing metamorphic minerals under pressure: Clinozoisite and topaz. Am Miner 88:1460–1470
Comodi P, Zanazzi PF (1997) The pressure behavior of clinozoisite and zoisite: an X-ray diffraction study. Am Miner 82:61–68
Dera P (2007) GSE-ADA data analysis program for monochromatic single crystal diffraction with area detector. GeoSoilEnviroCARS, Argonne, Illinois
Enami M, Mizukami T, Yokoyama K (2004) Metamorphic evolution of garnet-bearing ultramafic rocks from the Gongen area, Sanbagawa belt, Japan. J Metamorph Geol 22:1–15
Fan DW, Ma MN, Yang JJ, Wei SY, Chen ZQ, Xie HS (2011) In situ high-pressure synchrotron X-ray diffraction study of clinozoisite. Chin Phys Lett 28:126103
Fan DW, Xu JG, Wei SY, Chen ZQ, Xie HS (2014) In situ high-pressure synchrotron X-ray diffraction of natural epidote. Chin J High Press Phys 28:257–261 (in Chinese)
Franz G, Liebscher A (2004) Physical and chemical properties of the epidote minerals: an introduction. Rev Miner Geochem 56:1–82
Gatta GD, Merlini M, Lee Y, Poli S (2011) Behavior of epidote at high pressure and high temperature: a powder diffraction study up to 10 GPa and 1200 K. Phys Chem Miner 38:419–428
Gottschalk M (2004) Thermodynamic properties of zoisite, clinozoisite and epidote. Rev Miner Geochem 56:83–124
Grevel KD, Nowlan EU, Fasshauer DW, Burchard M (2000) In situ X-ray diffraction investigation of lawsonite and zoisite at high pressures and temperatures. Am Miner 85:206–216
Hammersley AP, Svensson SO, Hanfland M, Fitch AN, Hausermann D (1996) Two-dimensional detector software: from real detector to idealised image or two-theta scan. High Pres Res 14:235–248
Hermann J (2002) Allanite: thorium and light rare earth element carrier in subducted crust. Chem Geol 192:289–306
Holdaway MJ (1972) Thermal stability of Al-Fe epidote as a function of fO2 and Fe content. Contrib Min Pet 37:307–340
Holland TJB, Redfern SAT (1997) Unit cell refinement from powder diffraction data: the use of regression diagnostics. Miner Mag 61:65–77
Holland TJB, Redfern SAT, Pawley AR (1996) Volume behavior of hydrous minerals at high pressure and temperature: II. Compressibilities of lawsonite, zoisite, clinozoisite, and epidote. Am Miner 81:341–348
Klemd R (2004) Fluid inclusions in epidote minerals and fluid development in epidote-bearing rocks. Rev Miner Geochem 56:197–234
Lafuente B, Downs RT, Yang H, Stone N (2015) The power of databases: the RRUFF project. In: Armbruster T, Danisi RM (eds) Highlights in mineralogical crystallography. W. De Gruyter, Berlin, pp 1–30
Langer K, Raith M (1974) Infrared spectra of Al-Fe(III)-epidotes and zoisites, Ca2(Al1-pFe3+ p)Al2O(OH)[(Si2O7][SiO4]. Am Miner 59:1249–1258
Liebscher A (2004) Spectroscopy of epidote minerals. Rev Miner Geochem 56:125–170
Makreski P, Jovanovski G, Kaitner B, Gajović A, Biljan T (2007) Minerals from Macedonia XVIII. Vibrational spectra of some sorosilicates. Vib Spectrosc 44:162–170
Mao HK, Xu J, Bell P (1986) Calibration of the ruby pressure gauge to 800 kbar under quasi-hydrostatic conditions. J Geophys Res 91:4673–4676
Mao Z, Jiang FM, Duffy TS (2007) Single-crystal elasticity of zoisite Ca2Al3Si3O12(OH) by Brillouin scattering. Am Miner 92:570–576
Nagashima M (2011) Pumpellyite-, sursassite-, and epidote-type structures: common principles-individual features. J Miner Pet Sci 106:211–222
Pawley AR, Redfern SAT, Holland TJB (1996) Volume behavior of hydrous minerals at high pressure and temperature: I. Thermal expansion of lawsonite, zoisite, clinozoisite, and diaspore. Am Miner 81:335–340
Pawley AR, Chinnery NJ, Clark SM (1998) Volume measurements of zoisite at simultaneously elevated pressure and temperature. Am Miner 83:1030–1036
Poli S, Schmidt MW (1998) The high-pressure stability of zoisite and phase relationships of zoisite-bearing assemblages. Contrib Miner Pet 130:162–175
Poli S, Schmidt MW (2004) Experimental subsolidus studies on epidote minerals. Rev Miner Geochem 56:171–195
Rivers M, Prakapenka VB, Kubo A, Pullins C, Holl CM, Jacobsen SD (2008) The COMPRES/GSECARS gas-loading system for diamond anvil cells at the Advanced Photon Source. High Pres Res 28:273–292
Shieh SR, Duffy TS, Liu ZX, Ohtani E (2009) High-pressure infrared spectroscopy of the dense hydrous magnesium silicates phase D and phase E. Phys Earth Planet Inter 175:106–114
Shikazono N (1984) Compositional variations in epidote from geothermal areas. Geotherm J 18:181–187
Sinogeikin SV, Schilling FR, Bass JD (2000) Single crystal elasticity of lawsonite. Am Miner 85:1834–1837
Wang A, Han J, Guo L, Yu J, Zeng P (1994) Database of standard Raman spectra of minerals and related inorganic crystals. Appl Spectrosc 48:959–968
Winkler B, Milman V, Nobes RH (2001) A theoretical investigation of the relative stabilities of Fe-free clinozoisite and orthozoisite. Phys Chem Miner 28:471–474
Acknowledgments
This research was supported by the National Natural Science Foundation of China (U1232204 and 41473056) and in part through a scholarship to FQ by the Chinese Scholarship Council. SDJ acknowledges support from the US National Science Foundation through grant EAR-1452344 and the David and Lucile Packard Foundation. Use of the COMPRES/GSECARS gas-loading system was supported by COMPRES under NSF Cooperative Agreement No. EAR 11-57758 and by GSECARS through NSF Grant No. EAR-1128799 and DOE Grant No. DE-FG02-94ER14466. Portions of this work were performed at the Mineral Physics Laboratory of the University of Texas at Austin, and we would like to thank J.F. Lin for help with Raman spectroscopy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, F., Wu, X., Wang, Y. et al. High-pressure behavior of natural single-crystal epidote and clinozoisite up to 40 GPa. Phys Chem Minerals 43, 649–659 (2016). https://doi.org/10.1007/s00269-016-0824-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-016-0824-7